- Home
- »
- Medical Devices
- »
-
U.S. Ultrasound Probe Covers Market, Industry Report, 2033GVR Report cover
![U.S. Ultrasound Probe Covers Market Size, Share & Trends Report]()
U.S. Ultrasound Probe Covers Market (2025 - 2033) Size, Share & Trends Analysis Report By Fold (Accordion, Flat, Rolled), By Material (Latex, Polyethylene, Polyurethane), By Application, By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-768-8
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Ultrasound Probe Covers Market Summary
The U.S. ultrasound probe covers market size was estimated at USD 75.69 million in 2024 and is anticipated to reach USD 128.85 million by 2033, expanding at a CAGR of 6.13% from 2025 to 2033. This growth is attributed to the rising adoption of minimally invasive procedures, increasing emphasis on infection prevention and control, and the growing volume of diagnostic ultrasound examinations across hospitals, diagnostic centers, and outpatient facilities.
Key Market Trend & Insights
- By fold, the flat fold probe covers segment led the market with the largest revenue share in 2024.
- By application, the endo-cavity ultrasound segment led the market with the largest revenue share in 2024.
- By material, the polyethylene segments led the market with the largest revenue share in 2024.
- By end use, the hospital segment led the market with the largest revenue in 2024.
Market Size & Forecast
- 2024 Market Size: USD 75.69 Million
- 2033 Projected Market Size: USD 128.85 Million
- CAGR (2025-2033): 6.13%
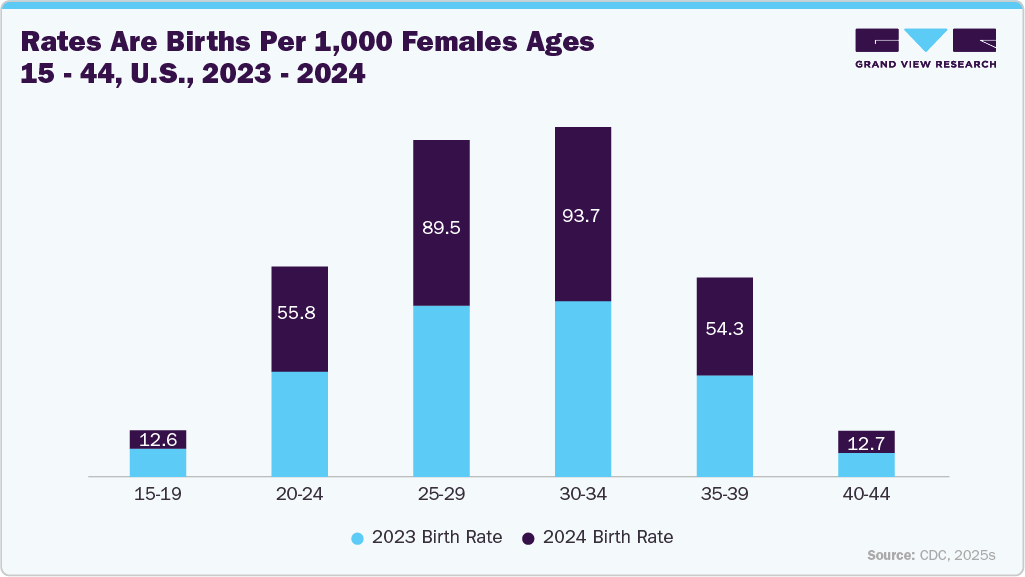
In addition, technological advancements in ultrasound systems, higher awareness of hygiene protocols among healthcare providers, and the increasing demand for single-use consumables are expected to drive market expansion further during the forecast period. Rising birth rates in the U.S. are among the strongest demand drivers for the ultrasound probe covers market because obstetric and gynecological imaging remains the largest application area for ultrasound. Every pregnancy involves multiple ultrasound scans for fetal health monitoring, and probe covers are essential for maintaining sterility, preventing cross-contamination, and ensuring infection control during these frequent procedures. The increase in live births, along with growing awareness around maternal and neonatal safety, has placed added emphasis on single-use, disposable covers in both hospitals and diagnostic centers.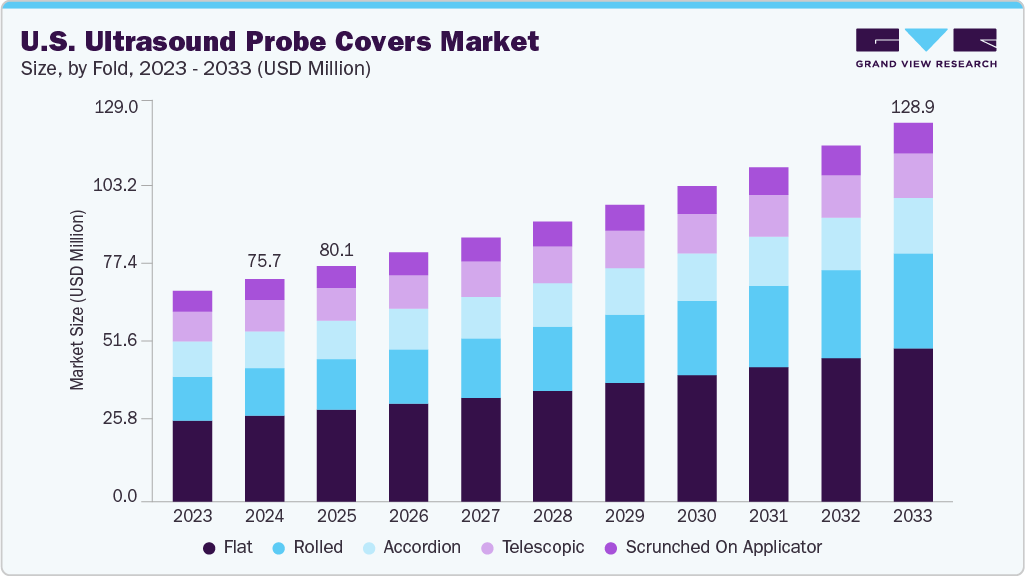
In addition, as more women are choosing to give birth in healthcare facilities equipped with advanced imaging, utilization of ultrasound has become almost universal in prenatal care. This patient flow translates to consistent, recurring consumption of probe covers, making birth rates a reliable underlying growth factor. Furthermore, U.S. policies that promote prenatal care access and insurance coverage ensure that nearly all pregnancies receive imaging support, supporting a stable and expanding market base for probe covers.
The prevalence and increasing incidence of various cancers significantly drive the demand for ultrasound probes and related consumables, including probe covers in the U.S. Ultrasound is widely used for early detection, diagnosis, treatment guidance, and follow-up monitoring in cancers such as breast, prostate, ovarian, thyroid, bladder, kidney, liver, and soft tissue malignancies. For instance, breast and thyroid cancers require frequent imaging for tumor detection and biopsy guidance, while prostate and ovarian cancers rely on transrectal and transvaginal ultrasound probes for accurate assessment. The rising cancer burden, coupled with the preference for non-invasive, real-time, and cost-effective imaging solutions, has increased the utilization of ultrasound systems in hospitals, diagnostic centers, and outpatient facilities.
Estimated Number of New Cancer Cases U.S., 2025
Cancer Type
Number of Cases
Breast
319,750
Uterine Corpus
69,120
Ovary
20,890
Prostate
313,780
Testis
9,720
Urinary Bladder
84,870
Kidney & Renal Pelvis
80,980
Liver & Intrahepatic bile duct
42,240
Gallbladder & biliary
12,610
Thyroid
44,020
Source: American Cancer Society
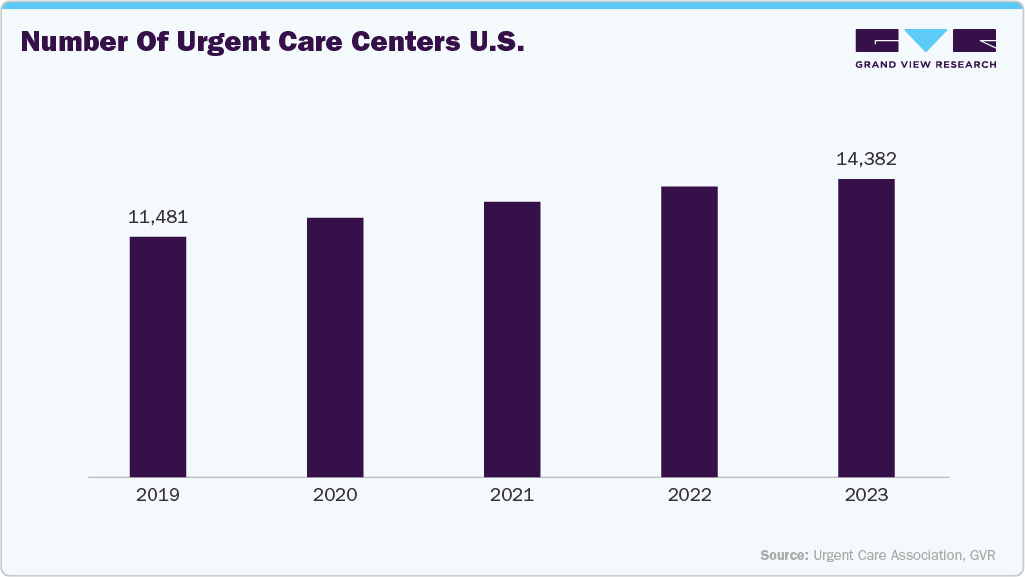
The rapid expansion of urgent care centers is a significant driver of the ultrasound probe covers market, as these facilities increasingly adopt diagnostic imaging, including point-of-care ultrasound, to deliver quick and accurate patient evaluations. Urgent care centers often serve as the first line of medical attention for injuries, pregnancy-related emergencies, and other acute conditions where ultrasound plays a vital role. With growing patient volumes and demand for cost-effective, immediate care, these centers rely on high-frequency ultrasound probes, necessitating strict infection control practices. As a result, the increased adoption of ultrasound services in urgent care settings directly boosts the consumption of disposable probe covers to ensure hygiene, prevent cross-contamination, and comply with infection control regulations.
Market Concentration & Characteristics
The U.S. ultrasound probe market is in a medium-growth stage, characterized by adoption across healthcare facilities, increasing emphasis on infection prevention protocols, and consistent demand from core applications such as obstetrics, gynecology, interventional radiology, and point-of-care diagnostics.
Innovation in the U.S. ultrasound probe covers market centers on advanced materials and functional improvements that balance infection control with imaging quality. Manufacturers are introducing thinner yet stronger barrier films, antimicrobial coatings, and eco-friendly materials that enhance acoustic transmission while reducing contamination risks. The push for disposable, single-use covers that perform reliably in high-volume hospital and outpatient settings has also improved fit and usability for various probe types. These innovations enhance patient safety and allow companies to differentiate products and command preference in competitive hospital procurement processes.
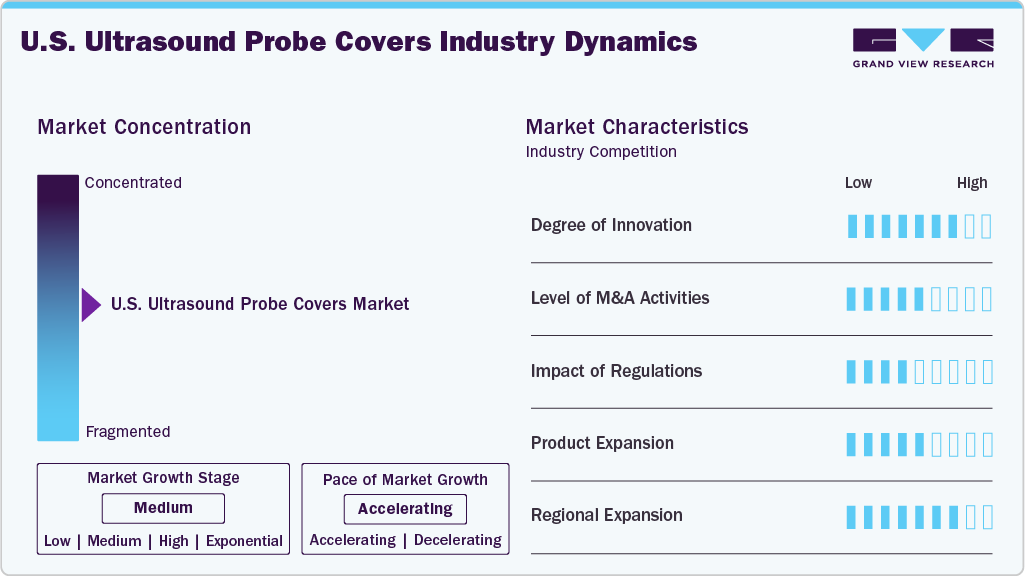
Mergers and acquisitions play a significant role in shaping the competitive landscape, as larger medical supply companies acquire niche cover manufacturers to expand their portfolios and secure distribution contracts with hospital systems. Acquisitions often focus on firms with proprietary polymer technologies, eco-friendly innovations, or strong regional market access. This consolidation increases economies of scale and strengthens bargaining power with healthcare providers, while smaller firms may position themselves as attractive acquisition targets. As a result, M&A activity accelerates market penetration, fosters portfolio expansion, and contributes to pricing competition across the sector.
Regulation is a critical market driver, as compliance with U.S. FDA device requirements and infection-control guidelines from the Centers for Disease Control and Prevention (CDC) is mandatory for probe covers. While probe covers are typically classified as consumable medical devices, regulatory scrutiny around sterility, quality assurance, and labeling directly impacts approval and hospital procurement. In addition, growing environmental concerns and potential regulations targeting single-use plastics may influence product design and materials. Strong regulatory compliance ensures market entry and builds trust with hospital infection-control committees, directly influencing purchasing decisions.
Product expansion in the market is being driven by growing demand for covers tailored to specialized probes, such as transesophageal, endocavity, and interventional ultrasound devices. Manufacturers are also developing bundled kits that combine probe covers with lubricants or disposal bags to simplify clinical workflow and improve convenience. Premium lines featuring antimicrobial coatings, acoustic-performance validation, or universal-fit designs are used as differentiators in competitive bids. This trend allows suppliers to capture greater value per procedure while meeting diverse customer needs across hospitals, diagnostic centers, and ambulatory care facilities.
Within the U.S., regional expansion is influenced by differences in healthcare infrastructure, procedure volumes, and population trends. High-birth-rate states, large metropolitan areas, and regions with strong outpatient diagnostic and OB/GYN clinic networks represent strong demand centers for probe covers. Suppliers pursue contracts with group purchasing organizations and integrated delivery networks to achieve scale, enabling access to multiple facilities under unified agreements. Regional penetration strategies, therefore, focus on balancing hospital procurement requirements with tailored offerings for ambulatory and specialty centers, ensuring wide product availability across the country.
Fold Insights
Flat fold probe covers dominate the U.S. ultrasound probe covers market 2024 primarily due to their ease of use, broad compatibility, and cost efficiency. Hospitals and diagnostic centers prefer flat-fold designs because they are simple to apply over a wide range of ultrasound probes, making them suitable for routine and specialized examinations. Their compact packaging also reduces storage space requirements and minimizes waste, which is especially important for high-volume healthcare settings. Furthermore, flat-fold covers are available in various materials, including polyethylene and latex-free options, aligning with infection prevention protocols and patient safety standards. These advantages, combined with their lower per-unit cost compared to more specialized formats such as telescopic or rolled covers, make flat-fold products the preferred choice across diverse clinical environments, driving their dominant share in 2024.
The rolled segment of the U.S. ultrasound probe covers is expected to grow at the fastest CAGR due to its convenience, efficiency, and time-saving benefits in clinical workflows. Rolled designs can be quickly unrolled directly onto the probe, reducing the risk of contamination and saving valuable preparation time during high-volume diagnostic or emergency procedures. This ease of application makes them particularly attractive in busy hospital settings, emergency rooms, and interventional radiology departments where rapid turnaround is critical. In addition, rolled covers ensure a smoother fit on the probe surface, improving image quality by minimizing air bubbles and wrinkles. As healthcare facilities prioritize efficiency and infection control, the adoption of rolled probe covers is accelerating, positioning this segment as the fastest-growing market.
Material Insights
Polyethylene ultrasound probe covers dominate the market in 2024 due to their cost-effectiveness, durability, and versatility across clinical applications, and they are also expected to register the fastest CAGR in the forecast years. These covers are widely preferred in hospitals, diagnostic centers, and ambulatory clinics because they offer reliable protection against contamination while being compatible with surface and endocavitary probes. Polyethylene’s ease of manufacturing allows it to be produced in various formats, including flat-fold and rolled designs, meeting the needs of high-volume healthcare settings. Growth is driven by increasing demand in emerging markets, growing awareness of infection control standards, and the healthcare industry’s shift toward disposable, single-use medical products. Innovations such as enhanced elasticity, tear resistance, and environmentally friendly variants further contribute to their rapid adoption, making polyethylene the material for established and expanding healthcare facilities.
Polyurethane ultrasound probe covers are expected to experience significant growth in the forecast years due to their superior elasticity, strength, and patient comfort compared to conventional materials. These covers provide a snug, wrinkle-free fit over probes, which enhances image quality and reduces the risk of air gaps during scanning. Their hypoallergenic properties make them particularly suitable for sensitive applications, such as endocavitary and fetal ultrasounds, where patient safety and comfort are critical. As healthcare facilities adopt advanced ultrasound technologies and focus on infection control, the demand for high-performance, durable probe covers such as polyurethane is rising. With ongoing innovations improving tear resistance and tactile sensitivity, polyurethane is poised to capture a larger market share, particularly in premium and specialized clinical settings.
Application Insights
Endo-cavity ultrasound dominates the U.S. ultrasound probe covers market in 2024 due to its high frequency of use in sensitive and specialized diagnostic procedures, particularly in gynecology, obstetrics, and urology. They are also expected to register the fastest CAGR in the forecast years. These procedures require endocavitary probes, which come into direct contact with mucous membranes, making strict infection control essential. Disposable probe covers are mandatory in such applications to prevent cross-contamination and maintain hygiene standards, driving consistent demand. The increasing number of prenatal screenings, fertility treatments, and minimally invasive interventions has further boosted the utilization of endo-cavity ultrasounds. The combination of clinical necessity, regulatory compliance, and growing procedural volumes ensures that endo-cavity applications remain the largest segment in the ultrasound probe covers market.
Interventional ultrasound holds a significant share of the U.S. ultrasound probe covers market due to its critical role in guiding minimally invasive procedures such as biopsies, catheter placements, and tumor ablations. These procedures require frequent and precise use of ultrasound probes in sterile environments, making disposable probe covers essential to prevent infection and maintain procedural safety. The growing adoption of image-guided interventions in hospitals and specialty clinics, coupled with an increasing preference for outpatient and ambulatory care settings, has contributed to the steady demand for probe covers in this segment. In addition, advancements in interventional ultrasound technology and rising clinician awareness of infection control protocols continue to support the segment’s strong market presence.
End Use Insights
Hospitals dominate the U.S. ultrasound probe covers market 2024 due to their high patient volumes, diverse diagnostic needs, and stringent infection control requirements. Hospitals perform various ultrasound procedures, including obstetrics, gynecology, interventional radiology, and emergency imaging, necessitating disposable probe covers to prevent cross-contamination. The presence of multiple departments and continuous patient turnover drives consistent and large-scale consumption of probe covers. Hospitals are more likely than smaller clinics or ambulatory centers to adopt advanced probe cover designs, such as rolled or polyurethane variants, to enhance efficiency and maintain compliance with regulatory standards.
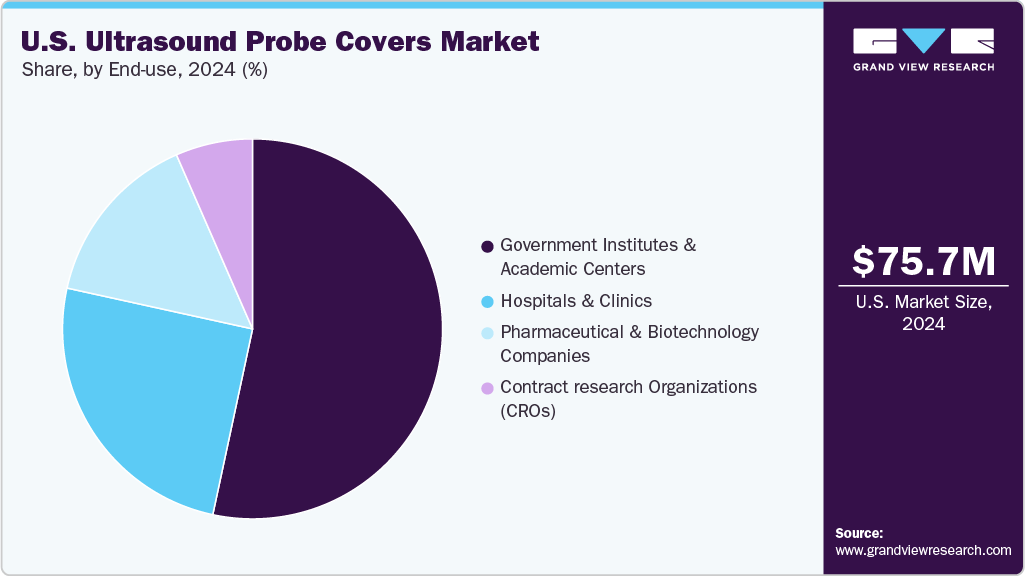
Diagnostic centers are expected to grow at the fastest CAGR in the U.S. ultrasound probe covers market due to their rapid expansion, increasing patient inflow, and rising adoption of advanced imaging services. These centers offer specialized and high-volume ultrasound diagnostics, including obstetric, gynecologic, vascular, and musculoskeletal imaging, requiring frequent probe covers to maintain hygiene and prevent cross-contamination. As healthcare delivery shifts toward outpatient and cost-efficient settings, diagnostic centers are increasingly preferred for routine and elective imaging procedures. Their focus on efficiency and patient safety drives demand for disposable probe covers, particularly easy-to-use designs like rolled and flat-fold covers, fueling accelerated growth in this segment.
Key U.S. Ultrasound Probe Covers Company Insights
The industry players are undertaking several strategic initiatives such as acquisitions, partnerships and collaborations. Moreover, the launch of novel products is anticipated to boost the competitive rivalry in the U.S. ultrasound probe covers market.
Key U.S. Ultrasound Probe Covers Companies:
- B. Braun SE
- Medline Industries, LP.
- Cardinal Health
- GE Healthcare Technologies
- Aspen Surgical Products, Inc.
- FITONE GROUP
- Boen Healthcare Co., Ltd.
- CIVCO Medical Solutions (Roper Technologies)
- Parker Laboratories, Inc.
- Ningbo Xinwell Medical Technology Co., LTD.
Recent Developments
-
In April 2025, B. Braun Medical Inc. introduced the EZCOVER Probe Cover Set in the U.S. market. This innovation enhances safety and efficiency during ultrasound-guided peripheral nerve block procedures. The EZCOVER Probe Cover Set addresses this concern by providing a sterile barrier that helps prevent cross-contamination and reduce the risk of healthcare-associated infections.
-
In February 2025, Aspen Surgical Products, Inc. announced Andau Medical as the exclusive distributor in Canada for Aspen’s full range of surgical products. This portfolio includes Aspen probe covers, needle guides, and patient positioning products previously supplied under a prior distribution agreement.
-
In September 2024, Medline is expanding its presence in the Chicagoland area by adding 110,000 square feet to its existing office space, strengthening its operational and distribution capabilities. This expansion reflects Medline’s strategy to enhance its market reach, improve supply chain efficiency, and better serve healthcare facilities across the region.
U.S. Ultrasound Probe Covers Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 80.07 million
Revenue forecast in 2033
USD 128.85 million
Growth rate
CAGR of 6.13% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Segments covered
Fold, application, material, end use
Regional scope
U.S.
Report coverage
Revenue, competitive landscape, growth factors, and trends
Key companies profiled
B. Braun SE; Medline Industries, LP.; Cardinal Health; GE Healthcare Technologies; Aspen Surgical Products, Inc.; FITONE GROUP; Boen Healthcare Co., Ltd.; CIVCO Medical Solutions (Roper Technologies); Parker Laboratories, Inc.; Ningbo Xinwell Medical Technology Co., LTD.
Customization scope
Free report customization (equivalent to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Ultrasound Probe Covers Market Report Segmentation
This report forecasts revenue growth at the country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For the purpose of this study, Grand View Research has segmented the U.S. ultrasound probe covers market report on the basis of fold, application, material, and end use:
-
Fold Outlook (Revenue, USD Million, 2021 - 2033)
-
Accordion
-
Flat
-
Rolled
-
Scrunched on applicator
-
Telescopic
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Surface Ultrasound
-
Endo-cavity Ultrasound
-
Interventional Ultrasound
-
Other Specialized Exams
-
-
Material Outlook (Revenue, USD Million, 2021 - 2033)
-
EMA
-
Latex
-
Polyethylene
-
Polyurethane
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Diagnostic Centers
-
Ambulatory / Clinics
-
Others
-
Frequently Asked Questions About This Report
b. The global U.S. ultrasound probe covers market size was estimated at USD 75.69 million in 2024.
b. The global U.S. ultrasound probe covers market is expected to grow at a compound annual growth rate of 6.13% from 2025 to 2033 to reach USD 128.85 million by 2033.
b. Flat fold probe covers dominated the U.S. ultrasound probe covers market in 2024.
b. Some key players operating in the U.S. ultrasound probe covers market include B. Braun SE, Medline Industries, LP, Cardinal Health, GE Healthcare Technologies, Aspen Surgical Products, Inc., FITONE GROUP, Boen Healthcare Co., Ltd., CIVCO Medical Solutions (Roper Technologies), Parker Laboratories, Inc., and Ningbo Xinwell Medical Technology Co., LTD.
b. The key factors driving the U.S. ultrasound probe covers market include a combination of clinical, technological, and regulatory influences. A primary driver is the rising number of ultrasound procedures across obstetrics, cardiology, urology, and point-of-care settings, which increases the demand for protective covers to maintain probe hygiene.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










