- Home
- »
- Pharmaceuticals
- »
-
U.S. Vaccines Market Size And Share, Industry Report, 2033GVR Report cover
![U.S. Vaccines Market Size, Share & Trends Report]()
U.S. Vaccines Market (2026 - 2033) Size, Share & Trends Analysis Report By Indication (Viral, Bacterial, Cancer Vaccines, Allergy), By Type (Subunit, Inactivated), By Payer Type, By Age Group, By Use Populations, By Usage/Administration Pattern, By Sales, And Segment Forecasts
- Report ID: GVR-4-68040-893-7
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
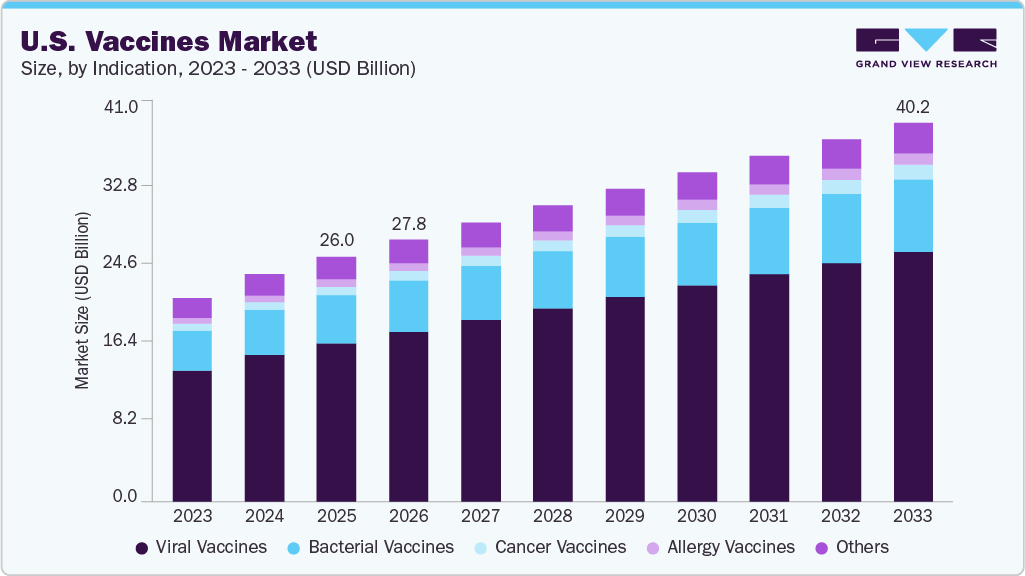
Market Size, 2025 (US$B)$26.0BForecast, 2033 (US$B)$40.2BCAGR, 2026 - 20335.4%U.S. Vaccines Market Summary
The U.S. vaccines market size was estimated at USD 26.00 billion in 2025 and is projected to reach USD 40.19 billion by 2033, growing at a CAGR of 5.40% from 2026 to 2033. The U.S. vaccines market is driven by rapid advancements in vaccine technologies and platform innovation.
Key Market Trends & Insights
- By indication, the viral vaccines segment held the highest market share of 64.67% in 2025.
- By type, the subunit vaccines segment held the highest market share in 2025.
- By payer type, the private payer segment held the highest market share in 2025.
- By Age Group, the adults (1864 years) segment held the highest market share in 2025.
- By Use Populations, the others segment held the highest market share in 2025.
- By Usage/Administration Pattern, the year-round vaccines segment held the highest market share in 2025.
- By Sales, the direct sales to providers segment held the highest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 26.00 Billion
- 2033 Projected Market Size: USD 40.19 Billion
- CAGR (2026-2033): 5.40%
The adoption of mRNA, recombinant, and vector-based approaches is accelerating the development of more effective and targeted vaccines across multiple disease areas. Continuous improvements in formulation, adjuvants, and delivery systems are enhancing immune response and overall product performance. Increasing focus on precision-based immunization is supporting the development of vaccines tailored to specific populations and disease risks.Advancements in manufacturing technologies are improving scalability, reducing production timelines, and ensuring consistent quality. Strong research pipelines and faster development cycles are enabling quicker responses to emerging health threats. These factors are strengthening innovation capabilities and supporting sustained growth across the U.S. vaccines market. For instance, in January 2026, the U.S. Department of Health and Human Services announced that the Centers for Disease Control and Prevention had accepted recommendations to revise the U.S. childhood immunization schedule following a Presidential Memorandum issued on December 5, 2025. The scientific assessment reviewed immunization practices across 20 peer-developed nations and found that, in 2024, the United States provided protection against 18 diseases, compared to Denmark, which immunized children against 10 diseases. The analysis also indicated that the U.S. recommended more childhood vaccines and more than twice as many doses as some European countries, while retaining three fully insured immunization categories without cost sharing.
Growing demand for adult immunization is a key factor supporting market expansion in the United States. Rising prevalence of infectious diseases among aging populations is increasing the need for vaccines across multiple indications and risk groups. Higher awareness of preventive healthcare is encouraging routine vaccination among adults, including those with chronic conditions. Expansion of vaccination access through pharmacies, clinics, and workplace programs is improving convenience and uptake. Increasing incidence of respiratory infections such as influenza and respiratory syncytial virus is driving seasonal vaccine demand across regions. A longer life expectancy reinforces the importance of ongoing immunization beyond early life stages. For instance, in October 2025, the Centers for Disease Control and Prevention released an updated adult immunization schedule for individuals aged 19 years and older, covering four age cohorts, 1926 years, 2749 years, 5064 years, and 65 years and above. The schedule recommended one or more doses of the COVID-19 vaccine, one influenza dose annually, a three-dose tetanus, diphtheria, and pertussis primary series followed by one booster every 10 years and one additional dose during each pregnancy, as well as one respiratory syncytial virus vaccine dose for adults aged 5074 years and those aged 75 years or older, while hepatitis B vaccination was recommended through 59 years of age. These structured dosing intervals reinforced sustained adult vaccine demand and long-term revenue stability.
The market is further driven by portfolio expansion and evolving vaccination strategies across disease areas. Increasing development of combination vaccines is improving convenience, reducing the number of doses, and enhancing patient compliance. Regular updates in vaccine formulations are addressing emerging variants and changing epidemiological patterns across populations. Expansion into new therapeutic areas, including oncology and personalized vaccines, is creating additional growth opportunities. Improvements in supply chain systems are supporting efficient distribution, storage, and availability across healthcare settings. Increased collaboration across research networks is accelerating innovation and clinical development. For instance, in January 2026, Health Policy Watch reported that U.S. health authorities implemented a major revision to the childhood immunization schedule, reducing the number of diseases covered by routine vaccination from 17 to 11 and removing universal recommendations for six vaccines, including those for rotavirus, influenza, meningococcal disease, hepatitis A, hepatitis B, and respiratory syncytial virus. The revision also reduced the human papillomavirus vaccination requirement from two doses to one and followed a December 5, 2025 directive to review national practices against peer nations such as Denmark, Japan, Australia, Canada, and the United Kingdom, which recommended vaccines for 10, 14, and 15 diseases, respectively. Previous guidance indicated that children in the United States could receive approximately 35 to 36 doses across 18 years, while respiratory syncytial virus alone accounted for about 80,000 pediatric hospitalizations annually, highlighting the significant public health and market implications of schedule changes.
Market Dynamics
The growth of the U.S. vaccines market was primarily driven by increasing government-supported immunization programs and expanding vaccination coverage across pediatric, adult, and elderly populations. Organizations such as the Centers for Disease Control and Prevention (CDC) and the U.S. Department of Health & Human Services (HHS) continued to promote routine and preventive vaccination through national awareness campaigns, funding support, and updated immunization schedules. In addition, rising awareness regarding infectious disease prevention, increasing focus on pandemic preparedness, and continuous advancements in vaccine development technologies contributed significantly to market expansion in the United States. For instance, in October 2024, the Centers for Disease Control and Prevention (CDC) endorsed the recommendation to lower the age for routine pneumococcal vaccination from 65 years to 50 years. The Advisory Committee on Immunization Practices (ACIP) voted 14 to 1 in favor of expanding pneumococcal vaccine recommendations to all adults aged 50 years and older in the U.S. The updated recommendation increased the eligible adult population for pneumococcal vaccination and aimed to improve protection against pneumonia, meningitis, and bloodstream infections among higher-risk populations. According to the CDC official announcement and , the revised guidance was expected to strengthen adult immunization coverage and support long-term growth in the U.S. vaccines market.
The growth of the U.S. vaccines market was restrained by increasing vaccine hesitancy and declining public confidence in immunization programs across certain population groups. The spread of misinformation regarding vaccine safety, concerns about side effects, and reduced trust in public health institutions negatively impacted vaccination uptake rates in the country. In addition, declining routine immunization coverage among children and adults created challenges for healthcare providers and government agencies in maintaining recommended vaccination levels. For instance, in October 2024, the Centers for Disease Control and Prevention (CDC) published its “Coverage with Selected Vaccines and Exemption Rates Among Children in Kindergarten, United States, 2023–24 School Year” report, which stated that national kindergarten coverage for the measles, mumps, and rubella (MMR) vaccine declined to 92.7%, below the recommended 95% coverage target. The report further highlighted that approximately 280,000 kindergarten children did not have documentation of completing the 2-dose MMR vaccine series during the 2023–2024 school year. Additionally, vaccine exemption rates increased to 3.3% from 3.0% in the previous school year, with exemptions exceeding 5% in 14 states. According to the CDC official MMWR report, declining vaccination coverage and rising exemption rates increased the risk of vaccine-preventable disease outbreaks and continued to act as a major restraint for the growth of the U.S. vaccines market.
The U.S. vaccines market witnessed significant growth opportunities due to the expanding adoption of adult immunization programs and the increasing commercialization of respiratory syncytial virus (RSV) vaccines. Rising awareness regarding preventive healthcare among aging populations, coupled with growing recommendations for adult vaccination against respiratory and infectious diseases, created substantial opportunities for vaccine manufacturers in the country. In addition, the introduction of new RSV vaccines for older adults and high-risk populations accelerated market expansion and product innovation. For instance, in June 2024, the Centers for Disease Control and Prevention (CDC) updated its recommendations for RSV vaccination among older adults in the United States. The agency recommended a single dose of RSV vaccine for all adults aged 75 years and older, as well as adults aged 60–74 years who were at increased risk of severe RSV disease. The CDC further stated that RSV leads to approximately 60,000 to 160,000 hospitalizations and 6,000 to 10,000 deaths annually among adults aged 65 years and older in the U.S. According to the CDC official RSV vaccination guidance for older adults, the expanded recommendations were expected to increase adult RSV vaccine uptake and create substantial commercial growth opportunities for vaccine manufacturers operating in the U.S. vaccines market.
Market Concentration & Characteristics
The U.S. vaccines market demonstrates a high and rapidly evolving level of innovation, driven by advanced technologies such as mRNA, recombinant platforms, and viral vectors. Companies like Moderna and Pfizer have accelerated vaccine development timelines significantly, especially after COVID-19. Innovation is now expanding into areas such as personalized cancer vaccines, combination vaccines, and next-generation boosters. Continuous R&D investment, supported by both private and public funding, sustains a strong pipeline of new products. Emerging tools like AI and genomic analysis are further enhancing vaccine discovery and trial efficiency. Overall, innovation is a key competitive differentiator and a major driver of long-term market growth.
Barriers to entry in the U.S. vaccines market are extremely high due to the capital-intensive nature of vaccine development and manufacturing. Firms must invest heavily in R&D, clinical trials, and specialized production facilities, which require advanced technical expertise. Regulatory approval processes governed by the U.S. Food and Drug Administration are stringent and time-consuming, often taking years to complete. Intellectual property protections and patents further restrict access to critical technologies. Additionally, established players benefit from strong distribution networks and government relationships. As a result, new entrants typically rely on partnerships or acquisitions to enter the market.
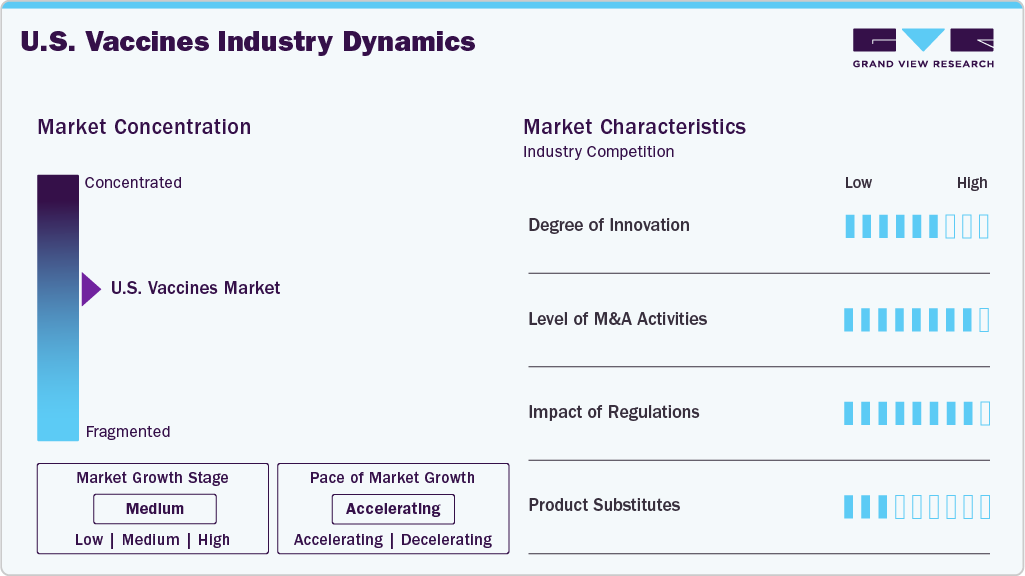
Regulations have a significant impact on the U.S. vaccines market, influencing every stage from development to commercialization. The U.S. Food and Drug Administration ensures safety and efficacy through rigorous clinical evaluation and approval standards. Meanwhile, the Centers for Disease Control and Prevention plays a key role in determining immunization schedules and vaccine recommendations. Government policies also affect pricing, reimbursement, and procurement, shaping demand dynamics. Regulatory decisions can accelerate or delay product launches, directly impacting company revenues. Overall, the regulatory environment creates both stability and operational complexity within the industry.
The threat of substitutes in the U.S. vaccines market is relatively low, as vaccines remain the most effective preventive solution for infectious diseases. Alternative treatments such as antiviral drugs or therapeutic interventions typically address symptoms rather than prevention. However, emerging options like monoclonal antibodies and gene-based therapies are beginning to act as partial substitutes in certain cases. Public health measures, including hygiene and behavioral interventions, can also reduce disease spread but are not direct replacements. Vaccine hesitancy may indirectly increase reliance on alternative solutions. Despite these factors, vaccines remain dominant in disease prevention.
Indication Insights
The viral vaccines segment dominated the market, accounting for the largest revenue share of 64.67% in 2025, driven by sustained demand for vaccines targeting influenza, COVID-19, RSV, and other viral diseases. High disease prevalence, recurring seasonal outbreaks, and the need for booster doses supported consistent uptake across both adult and pediatric populations. Continuous advancements in mRNA and recombinant vaccine technologies improved efficacy and accelerated product development timelines. Broad immunization coverage and increasing awareness of preventive healthcare further strengthened segment growth. Frequent updates in vaccine formulations to address emerging viral variants also contributed to market leadership. Strong pipeline activity in respiratory and infectious diseases continues to reinforce the segment's dominance. For instance, in January 2025, the Centers for Disease Control and Prevention stated that hepatitis A vaccination had been recommended for all children aged 12 to 23 months, with catch-up vaccination for those aged 2 to 18 years, and that complete protection required two doses administered at least 6 months apart, while a combination hepatitis A and B vaccine followed a three-dose schedule over 6 months.
The cancer vaccines segment is projected to grow at a CAGR of 7.31% over the forecast period, driven by increasing research in immuno-oncology and personalized medicine. The growing focus on therapeutic vaccines targeting specific tumor antigens is expanding treatment options across multiple cancer types. Advances in mRNA-based cancer vaccines and neoantigen-targeted therapies are accelerating clinical development. Growing clinical trial activity and encouraging early-stage results are supporting investor and industry interest. The increasing demand for targeted, less invasive treatment options is further driving adoption. Continuous innovation in biomarker-based therapies is expected to unlock new growth opportunities within this segment. For instance, between 2014 and 2024, a total of 757 cancer vaccine clinical trials were registered in the United States, compared with 89 in China, highlighting the scale of oncology vaccine development. The phase distribution showed that 52.1% of U.S. trials were in phase I, 36.3% in phase II, 7.9% in phase III, and 3.7% in phase IV, with 47.6% of trials conducted globally, 43.6% open, and 29.9% completed, and research spanning more than 20 cancer types compared with 5 in China, demonstrating a robust innovation pipeline supporting long-term market growth.
Type Insights
The subunit vaccines segment dominated the market with the largest revenue share of 42.95% in 2025 due to their strong safety profile and proven efficacy across multiple disease areas. These vaccines use specific antigens, which reduces the risk of adverse reactions and improves patient acceptance. Their stability and compatibility with established manufacturing processes support large-scale production. Subunit vaccines are widely used for influenza, hepatitis, and HPV, contributing to consistent demand. Advancements in adjuvant technologies have further enhanced immune response and product performance. Healthcare providers prefer these vaccines for routine immunization programs across diverse populations. Continuous improvements in formulation and delivery systems are reinforcing their leading position in the U.S. vaccines market. For instance, in September 2024, the Centers for Disease Control and Prevention reported that recombinant influenza vaccines such as Flublok were produced using synthetic technology that did not require influenza viruses or chicken eggs during manufacturing. The vaccine had first been licensed in the United States in 2013, with one trivalent recombinant influenza shot available for the season, and it was licensed for individuals aged 18 years and older and preferentially recommended among three vaccine options for adults aged 65 years and above.
The mRNA vaccines segment is projected to grow at a CAGR of 8.83% over the forecast period, owing to rapid technological advancements and expanding application areas. mRNA platforms enable faster vaccine development compared to traditional methods, supporting quicker responses to emerging diseases. High efficacy rates observed in recent vaccine launches have increased confidence in this technology. Ongoing research is expanding the use of mRNA vaccines into oncology and rare diseases. Improvements in storage and distribution requirements are enhancing accessibility. Increased investment in research pipelines is accelerating innovation and commercialization. These factors are driving strong growth potential for mRNA vaccines across the U.S. market.
Payer Type Insights
The private payer segment dominated the market, accounting for the largest revenue share of 49.85% in 2025, driven by widespread coverage through employer-sponsored insurance and private health plans. A large proportion of the U.S. population relies on private insurance for healthcare services, including vaccinations. Higher reimbursement rates and broader coverage policies support vaccine adoption in this segment. Private healthcare networks facilitate efficient access to vaccination services across urban and suburban areas. Increasing focus on preventive care is encouraging utilization of covered immunization services. Strong integration of vaccination programs within private healthcare systems enhances patient compliance. These factors contribute to the dominance of private payers in the vaccines market.
The self-pay / out-of-pocket segment is projected to grow at a CAGR of 6.40% over the forecast period owing to increasing demand for optional and travel-related vaccines. Rising awareness of preventive healthcare is encouraging individuals to invest in vaccinations beyond covered services. The expansion of retail clinics and pharmacy-based vaccination services is improving access for self-paying individuals. Growing international travel is driving demand for specialized vaccines such as yellow fever and typhoid. Higher disposable income levels are supporting out-of-pocket healthcare spending. The availability of a wider range of vaccines further encourages adoption. These factors are contributing to steady growth in this segment.
Age Group Insights
The adults (1864 years) segment dominated the market, accounting for the largest revenue share of 40.37% in 2025, driven by strong demand for routine and occupational vaccinations. This age group represents a large share of the working population and is therefore more exposed to infectious diseases. Increased awareness of preventive healthcare is driving vaccination uptake among adults. Workplace vaccination programs and healthcare initiatives are improving accessibility. Demand for vaccines such as influenza, HPV, and COVID-19 remains significant within this group. Active lifestyles and greater mobility increase vaccination needs. These factors support the leading position of adults in the U.S. vaccines market.
The elderly (65+) segment is projected to grow at a CAGR of 7.14% over the forecast period, owing to increased vulnerability to infectious diseases and age-related health conditions. Aging populations require regular immunization to prevent severe complications from infections. Higher incidence of diseases such as influenza, RSV, and pneumococcal infections is driving demand. Increased focus on healthy aging is encouraging vaccination uptake. The availability of vaccines specifically designed for older adults is supporting growth. Improved healthcare access and awareness are further enhancing adoption. These factors are contributing to the rapid expansion of this segment.
Use Populations Insights
The others segment dominated the market with the largest revenue share of 55.09% in 2025 due to broad coverage across the general population and routine immunization needs. This segment includes individuals receiving standard vaccinations outside specialized groups. Consistent demand for pediatric and adult vaccines supports strong revenue generation. Increasing awareness of preventive healthcare is encouraging vaccination across diverse populations. Availability of vaccines for multiple indications enhances segment coverage. Expansion of healthcare infrastructure is improving access to immunization services. These factors contribute to the dominance of this segment in the U.S. vaccines market.
The travelers segment is projected to grow at a CAGR of 8.10% over the forecast period, owing to increasing international travel and rising awareness of travel-related health risks. Demand for vaccines such as yellow fever, hepatitis A, and typhoid is increasing among international travelers. Growth in global mobility is driving the need for preventive immunization before travel. Expansion of travel clinics and specialized vaccination centers is improving access. Higher disposable income is supporting travel-related healthcare spending. Increased awareness of disease outbreaks in different regions is influencing vaccination decisions. These factors are driving growth in the traveler’s segment.
Usage/Administration Pattern Insights
The year-round vaccines segment dominated the market, accounting for the largest revenue share of 64.74% in 2025, driven by consistent demand for routine immunization across various disease areas. Vaccines for diseases such as HPV, hepatitis, and pneumococcal infections are administered throughout the year. Continuous need for preventive healthcare supports steady demand. Healthcare providers promote regular vaccination schedules to maintain immunity. Broad applicability across different age groups enhances utilization. Availability of established vaccination programs supports adoption. These factors reinforce the dominance of year-round vaccines in the market.
The seasonal vaccines segment is projected to grow at a CAGR of 6.26% over the forecast period owing to recurring outbreaks of seasonal diseases such as influenza. Annual vaccination campaigns drive consistent demand for seasonal vaccines. Increased awareness of disease prevention is encouraging higher uptake each year. Advances in strain selection and vaccine formulation are improving effectiveness. Expansion of vaccination services through pharmacies and clinics enhances accessibility. Rising incidence of seasonal infections supports growth. These factors contribute to the steady expansion of this segment.
Sales Insights
The direct sales to providers segment dominated the market with the largest revenue share of 64.85% in 2025, due to strong relationships between manufacturers and healthcare providers. Hospitals, clinics, and pharmacies serve as primary distribution channels for vaccines. Direct supply ensures efficient inventory management and timely availability. Established distribution networks support large-scale vaccine delivery. Healthcare providers prefer direct procurement for reliability and quality assurance. Integration with clinical workflows enhances administration efficiency. These factors contribute to the dominance of direct sales channels in the market.
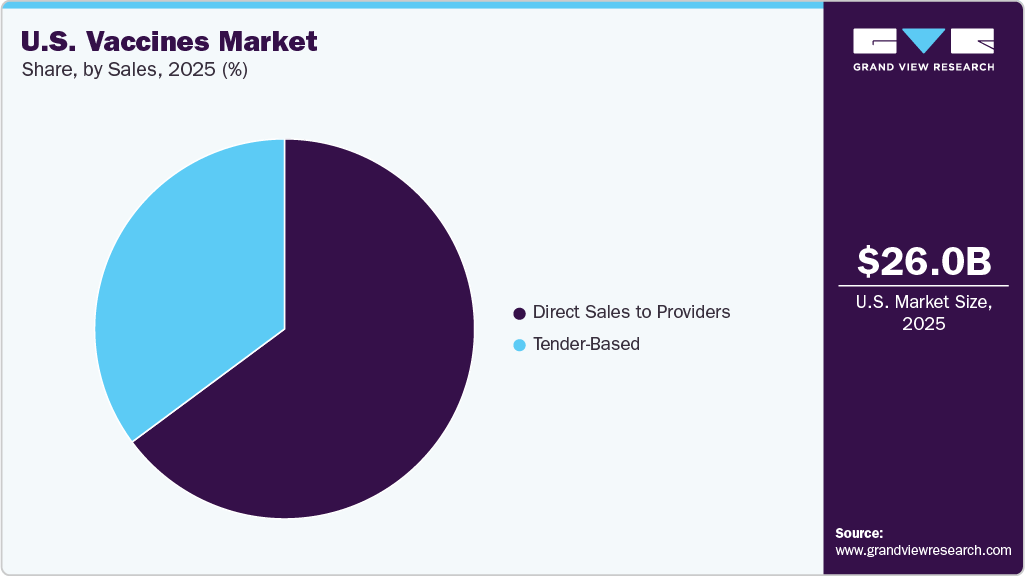
The tender-based segment is projected to grow significantly over the forecast period, driven by increasing bulk procurement practices and cost-optimization strategies. Large-scale purchasing agreements enable efficient vaccine distribution across multiple facilities. Competitive pricing structures attract buyers seeking cost-effective solutions. Expansion of organized healthcare networks is supporting tender-based procurement. Increased demand for high-volume vaccines is driving the adoption of this model. Streamlined procurement processes improve supply chain efficiency. These factors are supporting the growth of the tender-based segment.
Key U.S. Vaccines Company Insights
Pfizer and Moderna continue to dominate the U.S. vaccines market through their leadership in mRNA-based platforms and strong commercial portfolios in COVID-19 and next-generation vaccines. Merck & Co. and GSK maintain significant market share by focusing on established vaccines such as HPV, shingles, and respiratory diseases, supported by robust R&D pipelines. Sanofi and AstraZeneca are expanding their presence through innovative vaccine technologies and strategic collaborations targeting influenza, RSV, and combination vaccines. Novavax and CSL Limited are strengthening their positions with protein-based and specialty vaccines, particularly in influenza and adjuvant technologies. Serum Institute of India contributes to global supply and partnerships, enhancing accessibility and cost competitiveness in the U.S. and international markets. The competitive landscape is shaped by platform innovation, lifecycle management strategies, expanding adult immunization demand, and increasing focus on combination and next-generation vaccines.
Key U.S. Vaccines Companies:
- Pfizer
- GSK
- Merck & Co. (MSD)
- Sanofi
- Moderna
- Novavax
- CSL Limited
- AstraZeneca
- Serum Institute of India
Competitive Benchmarking
Operating Strategies
Competitive Edge
Weaknesses
Mature Players: Pfizer
- Pfizer focused on expanding its vaccine portfolio, strengthening manufacturing capacity, securing regulatory approvals, and collaborating with government agencies for immunization programs.
- The company also invested in advanced vaccine development, including RSV, influenza, pneumococcal, and combination vaccines, to maintain market leadership and strengthen its position in the U.S. vaccines market.
- Pfizer maintained competitive advantage through strong distribution networks, large-scale manufacturing infrastructure, diversified vaccine offerings, financial strength, and established healthcare partnerships.
- Its ability to support nationwide vaccination programs and continuously invest in vaccine innovation enabled the company to sustain a dominant position and strong commercial presence within the U.S. vaccines industry.
- Pfizer faced challenges including high operational and research costs, stringent regulatory compliance requirements, and pricing pressures from government-supported immunization programs.
- In addition, fluctuating demand for COVID-19 vaccines and large organizational structures occasionally reduced operational flexibility and slowed rapid responses to changing healthcare and vaccine market conditions in the U.S.
Emerging Players: Novavax
- Novavax prioritized innovation-driven strategies focused on recombinant and next-generation vaccine technologies. The company emphasized rapid clinical development, licensing agreements, and strategic collaborations to accelerate commercialization.
- It also invested in infectious disease vaccines and pandemic preparedness initiatives to strengthen market penetration and compete effectively with established pharmaceutical companies.
- Novavax gained competitive advantage through technological innovation, agility, and rapid vaccine development capabilities. Its expertise in recombinant protein-based vaccine technology enabled improved adaptability for emerging infectious diseases.
- The companys focus on next-generation vaccines and pandemic response strengthened its positioning within specialty and preventive vaccine markets across the United States.
- Novavax faced limitations including lower manufacturing capacity, funding dependency, restricted distribution infrastructure, and comparatively lower brand recognition.
- The company also experienced difficulties in scaling production, achieving widespread regulatory approvals, and competing with established pharmaceutical firms possessing stronger financial resources, larger commercial operations, and broader global vaccine distribution networks.
Recent Developments
-
In June 2025, The U.S. Food and Drug Administration expanded approval of Moderna’s respiratory syncytial virus vaccine, mRESVIA, to include adults aged 18 to 59 years at increased risk, extending its earlier indication limited to those aged 60 years and older. The vaccine had initially received approval in May 2024 as the first non-COVID messenger RNA vaccine in the United States. Moderna planned availability ahead of the 20252026 respiratory virus season, which typically begins in November, supporting broader adult immunization coverage and market growth.
-
In May 2025, The U.S. Food and Drug Administration approved Sanofi’s meningococcal vaccine, MenQuadfi, for infants as young as 6 weeks old, expanding its earlier approval for individuals aged 2 years and older. The vaccine provided protection against four serogroups, A, C, W, and Y, and the decision was supported by three late-stage clinical trials involving over 6,000 infants and toddlers aged 6 weeks to 19 months. In 2024, the United States recorded 503 confirmed or probable meningococcal cases, the highest since 2013, highlighting the need for pediatric immunization.
-
In September 2024, AstraZeneca announced that the U.S. Food and Drug Administration approved FluMist for self- or caregiver-administration, making it the first influenza vaccine in the United States that could be given without a healthcare provider. The vaccine was indicated for individuals aged 2 to 49 years, with adults aged 18 years and older eligible for self-administration. FluMist had originally received approval in 2003, with its use expanded to younger children in 2007, supporting broader influenza vaccination access and uptake in the U.S. vaccines market.
U.S. Vaccines Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 27.82 billion
Revenue forecast in 2033
USD 40.19 billion
Growth rate
CAGR of 5.40% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Indication, type, payer type, age group, use populations, usage/administration pattern, and sales
Key company profiled
Pfizer; GSK; Merck & Co. (MSD); Sanofi; Moderna; Novavax; CSL Limited; AstraZeneca; Serum Institute of India
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Vaccines Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. vaccines market report based on indication, type, payer type, age group, use populations, usage/administration pattern, and sales:
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Viral Vaccines
-
Hepatitis
-
Influenza
-
HPV
-
MMR
-
Rotavirus
-
Herpes Zoster
-
Japanese Encephalitis
-
RSV
-
Others
-
-
Bacterial Vaccines
-
Meningococcal Diseases
-
Pneumococcal Diseases
-
DPT
-
Others
-
-
Cancer Vaccines
-
Allergy Vaccines
-
Others
-
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Subunit Vaccines
-
Recombinant Vaccines
-
Conjugate Vaccines
-
Toxoid Vaccines
-
-
Inactivated
-
Live Attenuated
-
mRNA Vaccines
-
Viral Vector Vaccines
-
-
Payer Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Public Payer
-
Medicare
-
Medicaid
-
Federal Programs
-
-
Private Payers
-
Commercial Insurance Plans
-
Workplace Programs
-
-
Out-of-Pocket / Self-Pay
-
Others
-
-
Age Group Outlook (Revenue, USD Million, 2021 - 2033)
-
Pediatrics (0-17 years)
-
Adults (18-64 years)
-
Elderly (65+ years)
-
-
Use Populations Outlook (Revenue, USD Million, 2021 - 2033)
-
Workplace
-
Schools
-
Travelers
-
Military & Special Occupations
-
Others
-
-
Usage/Administration Pattern Outlook (Revenue, USD Million, 2021 - 2033)
-
Seasonal Vaccines
-
Year-Round Vaccines
-
Others
-
-
Sales Outlook (Revenue, USD Million, 2021 - 2033)
-
Tender-Based
-
Direct Sales to Providers
-
Delivered Customizations
This report has been delivered with the following In-depth customizations
Client Request
Customization Delivered
Value Adds
Adult RSV Vaccine Market Opportunity Assessment (U.S.)
Conducted a comprehensive assessment of the adult RSV vaccine opportunity across the U.S. market. The study evaluated target patient populations aged 60 years and above, hospitalization burden, CDC vaccination recommendations, pricing analysis of GSK and Pfizer RSV vaccines, retail pharmacy vaccination trends, payer reimbursement structures, and physician adoption patterns.
Helped the client identify high-growth adult immunization segments, estimate revenue opportunities, optimize pricing strategies, and evaluate competitive positioning against established RSV vaccine manufacturers operating within the U.S. vaccines market.
mRNA Vaccine Pipeline & Competitive Benchmarking
Delivered a detailed benchmarking analysis of mRNA vaccine developers focusing on Moderna, Pfizer-BioNTech, and emerging biotechnology companies. The assessment covered infectious disease and oncology vaccine pipelines, clinical trial progress, partnership activity, manufacturing scalability, intellectual property positioning, and commercialization strategies across the U.S. market.
Enabled the client to identify licensing and acquisition opportunities, evaluate white-space areas in mRNA vaccine development, and prioritize strategic investments in next-generation vaccine technologies with high commercial potential and long-term growth prospects.
U.S. Vaccine Manufacturing & Cold Chain Expansion Assessment
Conducted a nationwide feasibility assessment for vaccine manufacturing capacity and cold-chain infrastructure expansion across major U.S. healthcare and distribution networks. The study analyzed fill-finish capabilities, storage infrastructure, distribution bottlenecks, federal funding support, domestic manufacturing initiatives, and supply chain resilience for routine and pandemic-related vaccines.
Supported infrastructure investment planning by identifying high-demand vaccine categories, domestic production opportunities, supply chain optimization areas, and strategic partnerships to strengthen vaccine accessibility, distribution efficiency, and long-term procurement stability across the United States.
Frequently Asked Questions About This Report
The U.S. vaccines market size was valued at USD 26.0 billion in 2025 and is anticipated to reach USD 27.82 billion in 2026.
The U.S. vaccines market is expected to witness a compound annual growth rate of 5.4% from 2026 to 2033 to reach USD 40.19 billion by 2033.
Based on indication, the viral vaccines segment dominated the market with the largest revenue share of 64.67% in 2025, driven by sustained demand for vaccines targeting influenza, COVID-19, RSV, and other infectious viral diseases..
Some of the key players in U.S. vaccines market are Pfizer , GSK , Merck & Co. (MSD) , Sanofi , and Moderna
The U.S. vaccines market is driven by rapid advancements in vaccine technologies and platform innovation. The adoption of mRNA, recombinant, and vector-based approaches is accelerating the development of more effective and targeted vaccines across multiple disease areas..
About the authors:
Author: GVR Pharmaceuticals Research Team | Last Updated:
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









