- Home
- »
- Medical Devices
- »
-
Botulinum Toxin CDMO Market Size, Industry Report, 2033GVR Report cover
![Botulinum Toxin CDMO Market Size, Share & Trends Report]()
Botulinum Toxin CDMO Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Type A Botulinum Toxin, Type B Botulinum Toxin), By Service (Packaging & Labelling, Regulatory Affairs, Supply Chain & Logistics), By Application, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-909-6
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Botulinum Toxin CDMO Market Summary
The global botulinum toxin CDMO market size was estimated at USD 2.5 billion in 2025 and is projected to reach USD 5.8 billion by 2033, growing at a CAGR of 11.5% from 2026 to 2033. The industry is expected to grow due to strong demand from end users, expanding therapeutic applications, and ongoing advancements in biopharmaceutical technologies.
Key Market Trends & Insights
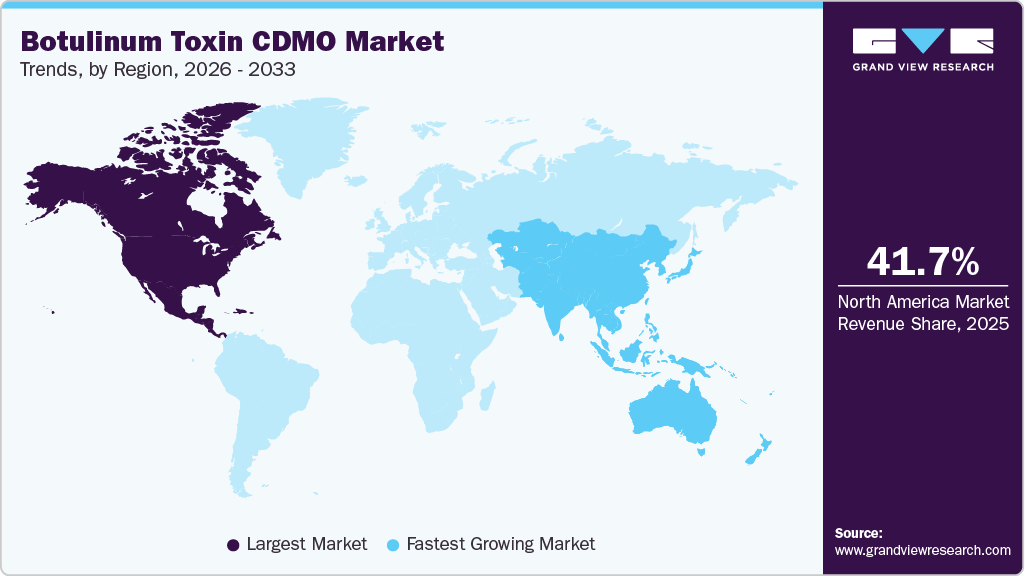
- North America botulinum toxin CDMO market held the largest share of 41.7% of the global market in 2025.
- The botulinum toxin CDMO industry in the U.S. is expected to grow significantly over the forecast period.
- Based on product, the type A botulinum toxin segment held the highest market share in 2025.
- Based on service, the contract manufacturing segment held the highest market share in 2025.
- Based on application, the aesthetic segment held the highest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 2.5 Billion
- 2033 Projected Market Size: USD 5.8 Billion
- CAGR (2026-2033): 11.5%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Besides, some other factors contributing to the market growth are the rising popularity of minimally invasive aesthetic procedures, growing consumer awareness, increased social acceptance, and an aging population seeking effective anti-aging solutions. In addition, the expanding range of therapeutic uses, including treatments for chronic migraine, spasticity, and overactive bladder, is expected to broaden the patient base and increase demand for specialized manufacturing capabilities.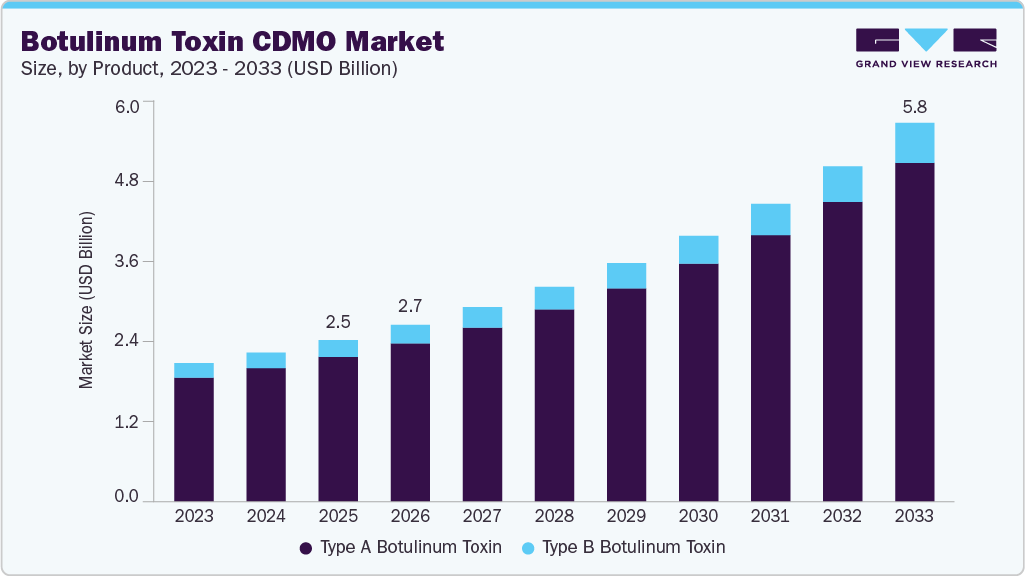
In addition, technological advancements are expected to drive the market growth by boosting efficiency, scalability, and product performance. Besides, developments in formulation, such as extended-duration botulinum toxin variants and protein-free compositions, are enhancing efficacy and minimizing immunogenicity risks. Advanced delivery systems, such as microneedles and precision injection devices, improve treatment outcomes and enhance patient safety. The rise of automation, digital manufacturing technologies, and single-use systems is facilitating more consistent processes and cost reductions.
Moreover, regulatory frameworks play a crucial role in shaping the market landscape. As botulinum toxin products are mostly considered biologics, they are required to adhere to strict compliance with quality, safety, and clinical standards. Increasing regulatory scrutiny and enhanced traceability requirements are prompting CDMOs to invest in robust quality management systems and meticulous documentation practices. The expansion of regulatory approvals for various indications supports broader commercialization and long-term market growth.
Furthermore, growing innovation driven by research aimed at extending product lifespans, enhancing safety profiles, and exploring emerging therapeutic applications, such as chronic pain and neuropsychiatric disorders. Besides, the emergence of biosimilars and next-generation neurotoxins following patent expirations is further intensifying competitive dynamics while supporting improved cost efficiency and broader market accessibility. In addition, the integration of advanced digital technologies, including AI-driven analytics and blockchain-enabled supply chain solutions, is enhancing operational transparency and effectively mitigating risks associated with counterfeit products.
Opportunity Analysis
The botulinum toxin CDMO industry is expected to witness new growth opportunities driven by geographic expansion, innovation in products, and a rising trend of outsourcing within the pharmaceutical and biotechnology sectors. Besides, regions such as Asia-Pacific and Latin America offer a range of opportunities driven by rising disposable incomes, heightened awareness of aesthetic procedures, and ongoing improvements in healthcare infrastructure. These factors are expected to drive the demand for affordable botulinum toxin treatments, prompting CDMOs to set up localized manufacturing and supply chains.
The advent of biosimilars and next-generation botulinum toxin formulations is driving market growth. As patents on leading products expire, new players are entering the market, facilitated by lowered entry barriers. These factors are expected to create strategic partnerships between pharmaceutical companies and CDMOs, enabling streamlined product development, scale-up, and commercialization. Besides, growing demand for integrated service offerings encompassing formulation development, analytical testing, clinical manufacturing, and regulatory guidance is expected to drive market growth.
Furthermore, advancements in manufacturing technologies such as continuous processing, automation, and digital quality management systems are boosting operational efficiency and ensuring regulatory compliance. These technologies help CDMOs improve scalability, shorten production timelines, and uphold high-quality standards, thereby enhancing their competitive edge. Thus, the increasing trend among pharmaceutical and biotech companies to outsource intricate biologics manufacturing, aimed at curbing capital expenditures and speeding up time-to-market, is likely to maintain strong demand for specialized CDMO services in the botulinum toxin sector.
Impact of U.S. Tariffs on the Global Botulinum Toxin CDMO Market
U.S. tariffs on pharmaceutical imports, including biologics and associated raw materials, are further influencing the global industry, reshaping supply chain dynamics, cost structures, and sourcing strategies. The increased tariffs on active pharmaceutical ingredients (APIs), intermediates, and specialized manufacturing equipment, especially those sourced from key supplier markets such as China and South Korea, are raising input costs for CDMOs that rely on imports. Besides, rising costs have reduced profit margins, forcing CDMOs to cover the costs or pass them on to clients, which, in turn, impacts pricing competitiveness in international markets. Thus, industry players are actively seeking to diversify their supply chains, bolster local sourcing capabilities, and form strategic partnerships to mitigate risks associated with tariffs.
Moreover, shifts toward manufacturing in North America are becoming a key area for domestic production investments aimed at reducing dependence on imports and strengthening supply chain resilience. While tariffs are driving innovation through the adoption of alternative sourcing models and advanced manufacturing technologies, they also add operational complexities and may constrain global collaboration. Thus, U.S. tariff policies are expected to drive the market for structural transformation in the industry, prompting realignment of supply chains, cost optimization efforts, and the growth of localized manufacturing ecosystems.
Technological Advancements
Technological advancements in botulinum toxin CDMO manufacturing are significantly fueling the production efficiency, quality, and scalability. Advanced fermentation and bioprocess optimization techniques enable high-yield toxin production with improved consistency through real-time monitoring and automation. Besides, precision purification technologies, such as advanced chromatography and filtration systems, ensure high purity while meeting stringent regulatory requirements, further contributing to market growth. In addition, aseptic fill-finish automation using robotics and isolator-based systems enhances sterility assurance and minimizes contamination risks, which is critical for drugs, further supporting the market.
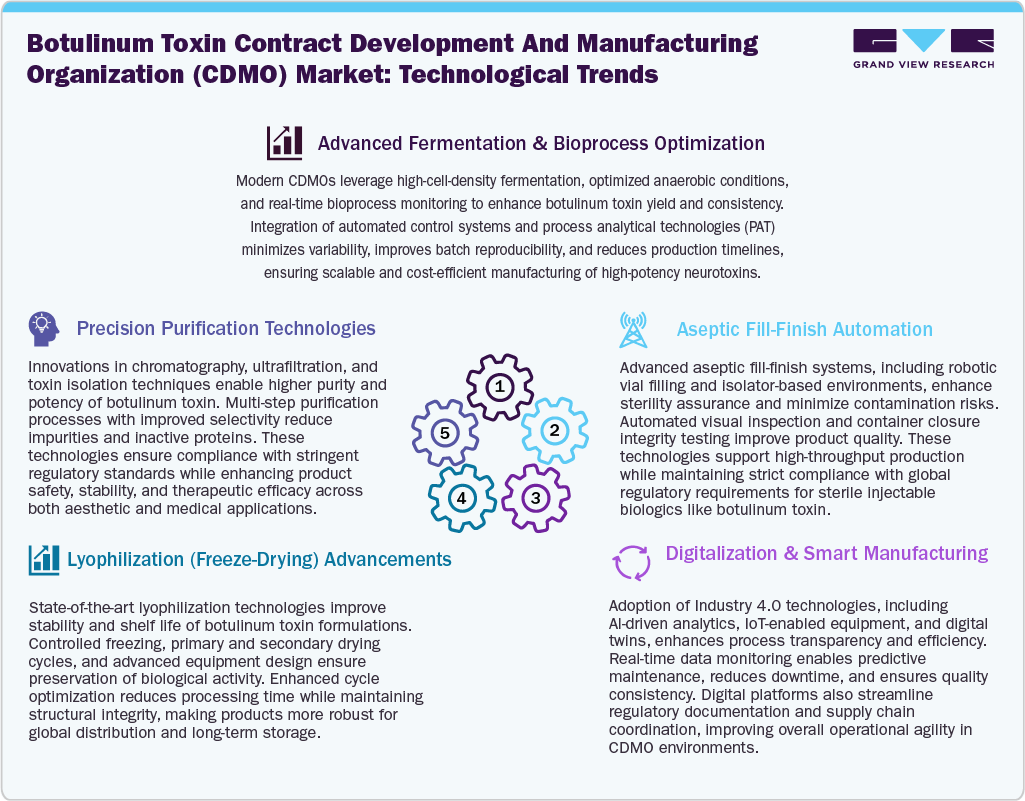
Moreover, innovations in lyophilization processes improve the stability and shelf life of botulinum toxin formulations by preserving their biological activity during storage and transportation. These developments are important for maintaining product efficacy across global supply chains. Furthermore, growing integration of digitalization and smart manufacturing technologies, including AI, IoT, and predictive analytics, is enabling CDMOs to achieve greater operational efficiency, reduce downtime, and enhance quality control. Thus, these technological advancements are expected to drive botulinum toxin CDMOs to deliver high-quality, scalable, and cost-effective products, supporting the growing demand for botulinum toxin in therapeutic and aesthetic applications worldwide.
Pricing Model Analysis
The industry features a variety of business models designed to accommodate different client needs throughout the development and commercialization process. The fee-for-service (FFS) model is widely used, allowing clients to contract with CDMOs for specific tasks such as formulation development, analytical testing, or batch manufacturing, offering high flexibility and cost transparency. Besides, the full-time equivalent (FTE) model promotes long-term partnerships by providing dedicated scientific and technical staff, which ensures continuity, efficient resource use, and predictable budgeting. In addition, for well-defined projects, the fixed-price model is favored, as it helps clients manage financial uncertainty through pre-agreed costs, though it requires clear deliverables and timelines.
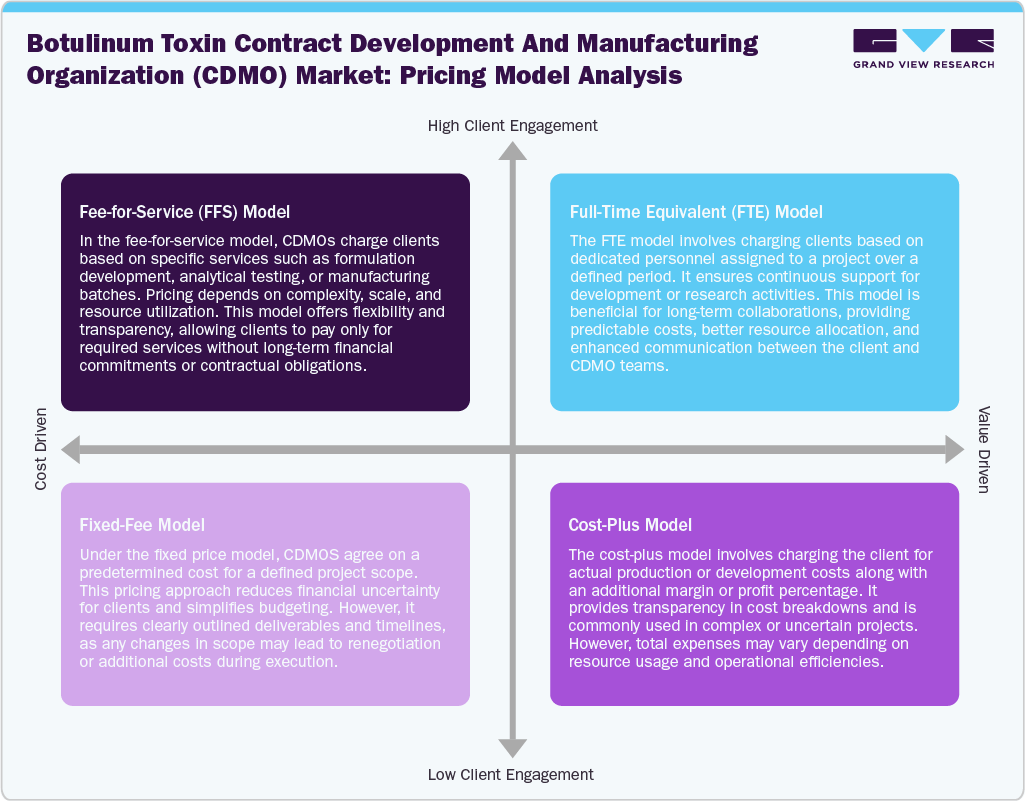
Furthermore, the cost-plus model offers transparency in complex projects, as pricing is based on actual incurred costs plus a negotiated margin, making it well-suited to handling variability in process development and manufacturing. Thus, these models are expected to drive the CDMOs and clients, promoting accountability while balancing financial risks, ultimately enhancing cooperation within the specialized market.
Market Concentration & Characteristics
The industry is at a high growth stage, with growth accelerating. The market is characterized by the degree of innovation, level of M&A activities, regulatory impact, service expansion, and regional expansion.
Innovation is crucial for competitiveness as CDMOs embrace advanced fermentation techniques, precision purification methods, and next-gen neurotoxin formulations. Innovations such as longer-lasting toxins, protein-free formulations, and enhanced delivery systems improve efficacy and safety, thereby enhancing productivity and enabling scalable solutions in this specialized field of biologics.

Mergers and acquisitions are on the rise as companies seek to bolster their biologics manufacturing capabilities and extend their geographic reach. These strategic acquisitions allow firms to gain access to niche expertise in toxin production, state-of-the-art facilities, and proprietary technologies, ultimately enhancing service offerings and accelerating entrance into new markets.
CDMO operations are significantly affected by stringent regulatory standards, given that botulinum toxin is considered a high-risk biologic. Adherence to good manufacturing practices (GMP), biosafety, and clinical guidelines can impact both development timelines and costs. CDMOs with robust regulatory knowledge help facilitate approvals, uphold product quality, and guide clients through the complexities of global regulatory environments.
To attract biopharma clients, CDMOs are broadening their service offerings across the entire value chain, including strain development, process optimization, analytical testing, fill-finish, and commercialization support. These comprehensive service packages simplify outsourcing for clients and foster enduring partnerships in both therapeutic and aesthetic markets.
In Asia-Pacific and North America, growth is accelerating to address rising demand while optimizing costs. Setting up local manufacturing sites strengthens supply chain resilience, lessens reliance on imports, and enhances regulatory compliance. This strategy enables CDMOs to engage with global clients and pursue new market opportunities efficiently.
Product Insights
On the basis of product, the type A botulinum toxin segment held the largest market share in the market, accounting for a revenue share of 89.6% in 2025. The segment is driven by strong clinical validation, extensive regulatory approvals, and widespread adoption across both aesthetic and therapeutic applications. Besides, type A botulinum offers various advantages, such as a longer duration of action, proven efficacy, and a favorable safety profile, making it the preferred option among healthcare providers and patients. In addition, the presence of established commercial brands, along with a robust pipeline of next-generation formulations, is expected to drive segment growth over the estimated time period. Moreover, rising demand for minimally invasive aesthetic procedures and the expansion of therapeutic applications, particularly in neurology and pain management, are expected to drive the requirement for Type A manufacturing and development services among CDMOs.
The type B botulinum toxin segment is expected to grow significantly during the forecast period. The segment's growth is driven by increasing adoption in therapeutic applications, growing clinical interest in alternative neurotoxins, and ongoing research into improved efficacy and targeted applications, which are further expanding the product pipeline. In addition, advancements in formulation technologies and efforts to enhance the duration of action are expected to drive the market growth.
Service Insights
On the basis of service, the contract manufacturing segment accounted for the largest share in 2025. The segment is driven by the increased complexity, stringent regulatory requirements, and capital-intensive nature of biologics production. Besides, most pharmaceutical & biotechnological companies rely on CDMOs with specialized infrastructure, containment capabilities, and expertise in large-scale sterile manufacturing. In addition, the rising need for consistent product quality, validated processes, and reliable global supply is expected to drive market growth. In addition, rising demand for botulinum toxin products and sustained demand across therapeutic and aesthetic applications are expected to drive market growth over the forecast period.
The contract development segment is expected to grow significantly during the forecast period. The segment growth is driven by the expanding pipeline of botulinum toxin products, including biosimilars and next-generation formulations. Besides, increased focus on product differentiation, enhanced efficacy, and novel therapeutic indications contribute to the development of services such as formulation optimization, process development, and analytical testing. In addition, early-stage outsourcing enables companies to accelerate timelines, manage development risks, and access advanced technical expertise. Furthermore, growing regulatory complexity and the need for integrated development solutions are prompting pharmaceutical and biotechnology companies to partner with CDMOs, thereby driving market growth.
Application Insights
On the basis of application, the aesthetic segment dominated the market with the largest revenue share in 2025. The segment is driven by growing demand for minimally invasive cosmetic procedures globally, increasing consumer awareness, rising disposable incomes, and the emerging influence of social media and beauty trends, which are boosting the procedure volumes. Besides, growing preference for non-surgical facial rejuvenation solutions with minimal downtime supports sustained demand for botulinum toxin products. In addition, continuous product innovation, including longer-lasting formulations and improved delivery techniques, is enhancing consumer adoption.
On the other hand, the therapeutic segment is projected to grow at a significant CAGR duringthe forecast period. The segment is driven by the expanding clinical applications and increasing acceptance of botulinum toxin in medical treatments. Some other factors contributing to the market growth are the growing prevalence of neurological and musculoskeletal disorders, along with conditions such as chronic migraine and spasticity. Besides, ongoing research into new indications, including pain management and urological disorders, is further broadening the treatment landscape. In addition, favorable clinical outcomes and increasing physician confidence are expected to offer new growth opportunities in the market.
End Use Insights
On the basis of end use, the pharmaceutical companies segment accounted for the largest share in 2025 during the forecast period. The segment growth is driven by established product portfolios, strong financial capabilities, and extensive global presence. These companies drive the large-scale demand for both development and commercial manufacturing services, particularly for approved and widely distributed botulinum toxin products. Besides, the focus on lifecycle management, regulatory compliance, and supply chain reliability has further increased the reliance on experienced CDMO partners. In addition, ongoing investments by pharmaceutical companies to expand therapeutic indications further drive market growth.
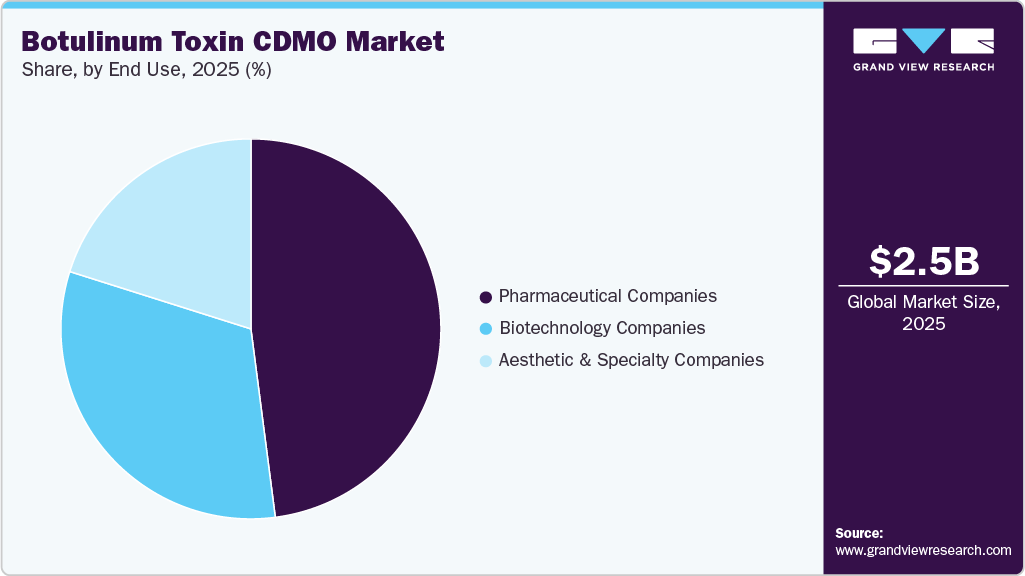
The aesthetic & specialty companies segment is expected to grow significantly during the forecast period. The growth is attributed to a strong focus on cosmetic applications and niche therapeutic areas, which has led companies to increasingly invest in innovative formulations, branding strategies, and targeted market expansion, driven by growing consumer demand for aesthetic treatments. Besides, limited in-house manufacturing capabilities and the rising need for operational flexibility have increased reliance on CDMOs for development, production, and packaging services. Furthermore, the rapid evolution of consumer preferences and competitive market dynamics is prompting these companies to accelerate product launches.
Regional Insights
North America botulinum toxin CDMO market led in 2025, accounting for a revenue share of 41.7%. The market growth is attributed to the presence of a well-established biopharmaceutical infrastructure, high R&D expenditure, and the increasing presence of biotech and pharmaceutical companies. Besides, most companies are actively outsourcing complex biologics development and manufacturing, which are expected to drive the regional growth over the estimated time period. In addition, advanced infrastructure, robust regulatory frameworks, and early adoption of innovative technologies, such as continuous bioprocessing and single-use systems, further enhance operational efficiency and support the market. Moreover, the increasing demand for aesthetic and therapeutic applications is anticipated to drive outsourcing in the region.
U.S. Botulinum Toxin CDMO Market Trends
The botulinum toxin CDMO market in the U.S. accounted for the highest market share in North America, owing to strong innovation capabilities and substantial R&D investments. Besides, the presence of leading pharmaceutical and biotechnology companies is expected to drive the demand for advanced development and manufacturing services. In addition, the growing adoption of cutting-edge technologies, including single-use systems and continuous bioprocessing, enhances operational efficiency and scalability, thereby contributing to market growth. Moreover, a well-defined regulatory framework and streamlined approval pathways support faster commercialization of biologics.
The Canada botulinum toxin CDMO market is expected to grow at a significant CAGR during the forecast period.The market is driven by the growing biotechnology sector, favorable government initiatives, and increasing investments in advanced manufacturing infrastructure. Besides, funding programs and regulatory support mechanisms are encouraging innovation and facilitating partnerships between biopharma companies and CDMOs. In addition, the growing emphasis on research-driven development and clinical advancement is expected to drive demand for both early-stage and commercial manufacturing services over the estimated time period.
Europe Botulinum Toxin CDMO Market Trends
The botulinum toxin CDMO market in Europe is driven by strong regulatory expertise, advanced manufacturing capabilities, and a well-integrated biopharmaceutical industry. Countries like Germany, the UK, and France further support the innovation and high-quality production. In addition, the increasing demand for biologics is expected to drive outsourcing to specialized CDMOs. Besides, growing initiatives between industry players and research institutions for clinical research and increased focus on adherence to stringent GMP standards are accelerating product development. Moreover, favorable conditions for clinical trials and GMP compliance further support the market growth.
Germany botulinum toxin CDMO market held the highest regional share in 2025. Germany is one of the leading botulinum toxin CDMO hubs, supported by advanced infrastructure, a skilled workforce, and strong biopharma R&D activities. Besides, increased demand for monoclonal antibodies, vaccines, and innovative therapies drives outsourcing to CDMOs, further supporting the market growth. Moreover, regulatory expertise and GMP-compliant facilities attract global clients, reinforcing Germany's critical position in European clinical and commercial biologics manufacturing.
The market's growth is driven by a strong industrial base, advanced manufacturing capabilities, and a well-established biopharmaceutical sector. Besides, the presence of a skilled workforce and robust research infrastructure benefits the country with continuous investments in process innovation. Moreover, the increasing demand for biologics, including monoclonal antibodies and advanced therapies, contributes to market growth. Moreover, stringent regulatory standards and growing strategic collaborations in the market are expected to drive the market over the estimated time period.
The botulinum toxin CDMO market in the UK is expected to grow significantly over the forecast period. The country's growth is fueled by an established biotechnology landscape, a strong academic research base, and a supportive regulatory environment. Besides, significant government initiatives and funding programs drive the market growth. In addition, the growing demand for complex biologics, including cell and gene therapies, is driving outsourcing. Moreover, innovation, combined with access to skilled professionals and advanced infrastructure, supports market growth.
Asia Pacific Botulinum Toxin CDMO Market Trends
Asia Pacific botulinum toxin CDMO market is expected to grow at a significant CAGR over the forecast period. The market is driven by growing cost advantages, expanding healthcare infrastructure, and increasing biopharmaceutical investments. Besides, countries such as China, India, and Japan are strengthening their manufacturing capabilities and regulatory frameworks to align with global standards. Moreover, the growing availability of skilled talent and production facilities is expected to boost the multinational companies to outsource development and manufacturing activities. In addition, government initiatives are enhancing regional competitiveness, positioning the region as a key hub for botulinum toxin outsourcing.
The botulinum toxin CDMO market in China is witnessing new growth opportunities due to strong government support, rapid expansion of the biotechnology sector, and increasing investments in research and development. Besides, with cost-effective manufacturing solutions and a growing base of advanced production facilities, the country has witnessed rising domestic demand for biologics, coupled with increased participation by global pharmaceutical companies, which have further accelerated outsourcing activities, supporting the market.
A well-established pharmaceutical industry, advanced technological capabilities, and a strong emphasis on quality and compliance characterize Japan botulinum toxin CDMO market. The country's robust regulatory framework and expertise in precision manufacturing ensure high standards in biologics production, further supporting the market growth. Moreover, collaborative efforts between industry and academia drive innovation, contributing to market growth.
The botulinum toxin CDMO market in India is experiencing rapid expansion, driven by a cost-efficient manufacturing environment, an expanding pharmaceutical sector, and the availability of a highly skilled workforce. The growing demand for scalable production capabilities and regulatory alignment with global standards is driving the country's development, strengthening its market.
Latin America Botulinum Toxin CDMO Market Trends
The botulinum toxin CDMO market in Latin America is expected to grow significantly over the forecast period. The market is driven by increasing healthcare investments, improving regulatory environments, and expanding biopharmaceutical capabilities. Countries such as Brazil and Argentina are focusing on strengthening domestic manufacturing to reduce reliance on imports and enhance regional supply chains, further supporting the market. In addition, government initiatives to promote local production and innovation are contributing to market development.
Brazil's botulinum toxin CDMO market is driven by growing investments in healthcare infrastructure, supportive government policies, and an increasing focus on local biopharmaceutical production. The country is witnessing rising demand for biologics, including vaccines and biosimilars, further supporting companies in driving development and expanding regional access. Such factors are expected to drive the market over the estimated time period.
Middle East & Africa Botulinum Toxin CDMO Market Trends
The botulinum toxin CDMO market in the MEA region is expected to grow steadily over the forecast period. The market is driven by government support, investments in advanced manufacturing, and its strategic location. Also, demand for vaccines, monoclonal antibodies, and innovative therapies is driving outsourcing. Moreover, scalable, compliant, and cost-effective production is enhancing the market growth.
The UAE botulinum toxin CDMO market is driven by increasing government investments in healthcare infrastructure, strategic initiatives to develop the life sciences sector, and its role as a regional hub for pharmaceutical distribution. The favorable business environment and regulatory advancements attract global biopharma companies to establish partnerships with CDMOs. In addition, the UAE's focus on innovation, coupled with access to modern manufacturing facilities and logistics capabilities, is expected to drive market growth.
Key Botulinum Toxin CDMO Companies Insights
The key players operating across the market are adopting strategic initiatives such as service launches, mergers & acquisitions, partnerships & agreements, and expansions to gain a competitive edge in the market. For instance, in April 2026, Chongqing Claruvis Pharmaceutical Co., Ltd. received approval from the National Medical Products Administration for its product Retoxin® (recombinant botulinum toxin type A, project code YY001). The therapy is indicated for the temporary reduction of moderate-to-severe glabellar lines in adult patients.
Key Botulinum Toxin CDMO Companies:
The following key companies have been profiled for this study on the botulinum toxin CDMO market.
- Lonza Group
- Thermo Fisher Scientific (Patheon)
- Vetter Pharma-Fertigung GmbH & Co. KG
- Recipharm AB
- Cenexi Group
- Grand River Aseptic Manufacturing
- Pharmaron
- WuXi Biologics
- Samsung Biologics
- Catalent, Inc.
- Boehringer Ingelheim BioXcellence
- Fujifilm Diosynth Biotechnologies
- Cambrex Corporation
- Apeloa Pharmaceutical Co., Ltd.
- Hikal Ltd
Recent Developments
- In September 2025, Rezon Bio launched as a European CDMO specializing in biologics. As a new brand, it leverages an established track record of supporting biologics development from gene-level research to global commercialization through partnerships with experienced industry collaborators.
Botulinum Toxin CDMO Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 2.7 billion
Revenue forecast in 2033
USD 5.8 billion
Growth rate
CAGR of 11.5% from 2026 to 2033
Actual data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, service, application, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Thailand; South Korea; Australia; Brazil; Argentina; South Africa; UAE; Saudi Arabia; Kuwait; Qatar; Oman
Key companies profiled
Lonza Group; Thermo Fisher Scientific (Patheon); Vetter Pharma-Fertigung GmbH & Co. KG; Recipharm AB; Cenexi Group; Grand River Aseptic Manufacturing; Pharmaron; WuXi Biologics; Samsung Biologics; Catalent, Inc.; Boehringer Ingelheim BioXcellence; Fujifilm Diosynth Biotechnologies; Cambrex Corporation; Apeloa Pharmaceutical Co., Ltd.; Hikal Ltd.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Botulinum Toxin CDMO Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global botulinum toxin CDMO market report based on product, service, application, end use, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Type A Botulinum Toxin
-
Type B Botulinum Toxin
-
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Contract Development
-
Formulation Development
-
Process Development and Scale-Up
-
Analytical and Testing Services
-
Others
-
-
Contract Manufacturing
-
Clinical Manufacturing
-
Commercial Manufacturing
-
-
Packaging & Labelling
-
Regulatory Affairs
-
Supply Chain & Logistics
-
Others
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Therapeutic
-
Aesthetic
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical Companies
-
Biotechnology Companies
-
Aesthetic & Specialty Companies
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
UAE
-
Saudi Arabia
-
Kuwait
-
Qatar
-
Oman
-
-
Frequently Asked Questions About This Report
b. The global botulinum toxin CDMO market size was estimated at USD 2.5 billion in 2025 and is expected to reach USD 2.7 billion in 2026.
b. Type A botulinum toxin segment held the largest market share of 89.6% share in 2025. The segment growth is attributed to strong clinical validation, extensive regulatory approvals, and widespread adoption across both aesthetic and therapeutic applications. Besides, it offers several advantages, including a longer duration of action, proven efficacy, and a favorable safety profile, making it the preferred option among healthcare providers and patients.
b. Some key players operating in the botulinum toxin CDMO market include Lonza Group, Thermo Fisher Scientific (Patheon), Vetter Pharma-Fertigung GmbH & Co. KG, Recipharm AB, Cenexi Group, Grand River Aseptic Manufacturing, Pharmaron, WuXi Biologics, Samsung Biologics, Catalent, Inc., Boehringer Ingelheim BioXcellence, Fujifilm Diosynth Biotechnologies,, Cambrex Corporation, Apeloa Pharmaceutical Co., Ltd., and Hikal Ltd., among others.
b. Some of the key factors driving market growth includes strong demand from end users, expanding therapeutic applications, and ongoing advancements in biopharmaceutical technologies. Besides, other factors contributing to market growth include the rising popularity of minimally invasive aesthetic procedures, growing consumer awareness, increased social acceptance, and an aging population seeking effective anti-aging solutions.
b. The global botulinum toxin CDMO market is expected to grow at a compound annual growth rate of 11.5% from 2026 to 2033 to reach USD 5.8 billion by 2033.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










