- Home
- »
- Medical Devices
- »
-
Clinical Trial Third Party Logistics Market Size Report, 2033GVR Report cover
![Clinical Trial Third Party Logistics Market Size, Share & Trends Report]()
Clinical Trial Third Party Logistics Market (2026 - 2033) Size, Share & Trends Analysis Report By Service (Logistics & Distribution, Storage & Retention, Packaging, Labeling & Blinding), By Phase, By Therapeutic Area, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-861-5
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Clinical Trial Third Party Logistics Market Summary
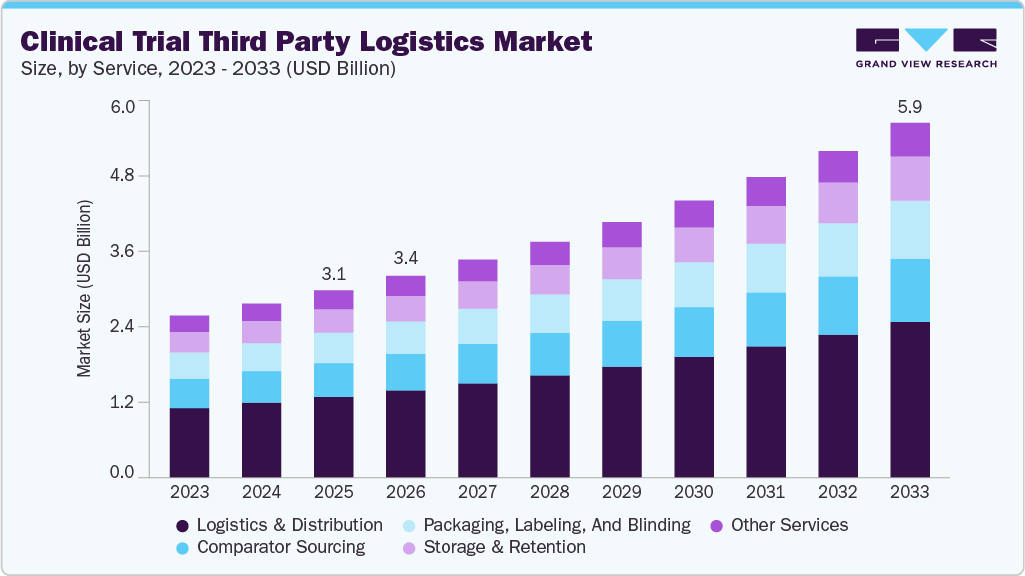
The global clinical trial third party logistics market size was estimated at USD 3.13 billion in 2025 and is projected to reach USD 5.93 billion by 2033, growing at a CAGR of 8.51% from 2026 to 2033. The growing globalization of clinical research, rising number of ongoing and planned clinical trials, and increasing R&D spending by pharmaceutical and biopharmaceutical companies are key factors expected to support market expansion.
Key Market Trends & Insights
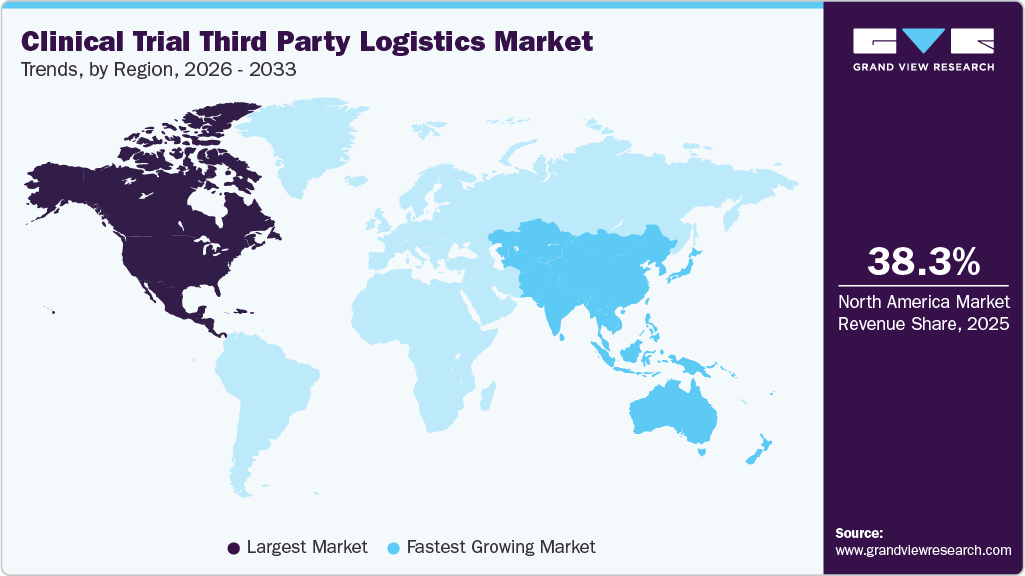
- The North America clinical trials third party logistics market held the largest global share of 38.26% in 2025.
- The U.S. clinical trials third party logistics industry is expected to grow significantly from 2026 to 2033.
- Based on Service, the logistics & distribution segment held the largest market share of 43.17% in 2025.
- Based on Phase, the phase III segment held the largest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 3.13 Billion
- 2033 Projected Market Size: USD 5.93 Billion
- CAGR (2026-2033): 8.51%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
In addition, ongoing advancements in supply chain technologies are enhancing operational efficiency, transparency, and reliability across clinical trial logistics. The increasing complexity of study designs, including multi-regional, decentralized, and advanced therapy trials, is further driving the adoption of digital tracking, real-time monitoring, and data-driven logistics solutions to enable faster, more compliant, and cost-effective clinical trial execution.The clinical trial 3PL market is experiencing strong growth due to the rising adoption of precision medicine, biologics, and decentralized clinical trial models, which have significantly increased the need for specialized outsourced logistics services. Advanced therapies require stringent temperature control, customized packaging, secure transportation, and real-time monitoring to maintain product integrity and regulatory compliance across global trial sites. As a result, pharmaceutical and biotechnology companies are increasingly relying on third-party logistics providers to manage complex distribution networks, cold chain requirements, and global site coordination, thereby supporting market expansion.
In addition, the growing complexity of multi-regional trials, expansion into emerging markets, and increasing regulatory scrutiny are accelerating the shift toward outsourced logistics models. Sponsors are focusing on core drug development activities while leveraging the expertise of 3PL providers in areas such as inventory management, risk mitigation, demand forecasting, and last-mile delivery. The integration of digital tracking platforms, IoT-enabled temperature monitoring, and just-in-time supply strategies is further enhancing operational efficiency and visibility across the clinical trial logistics ecosystem. Strategic partnerships and service expansion initiatives are also enabling logistics providers to broaden their global reach and strengthen service portfolios.
Furthermore, increasing R&D investments and the expanding pipeline of biologics, cell and gene therapies, and specialty injectables are expected to drive sustained demand for outsourced clinical trial logistics. Pharmaceutical and biotechnology companies are investing in global trial expansion and patient-centric approaches, which require flexible, scalable, and compliant logistics networks. Leading providers such as DHL and UPS Healthcare continue to expand temperature-controlled infrastructure, digital capabilities, and regional distribution hubs to address evolving sponsor requirements, reinforcing the long-term growth outlook of the clinical trial third party logistics industry.
Market Opportunities
The clinical trial 3PL market is expected to witness significant expansion driven by continued advancements in personalized medicine, decentralized clinical trial models, and increasing regulatory emphasis on compliant and resilient logistics operations. Rising R&D investments, the growing complexity of biologics and specialty therapies, and the integration of digital technologies are reshaping outsourcing strategies across the pharmaceutical and biotechnology sectors. Sponsors are increasingly partnering with specialized third-party logistics providers to enhance operational agility, ensure global regulatory compliance, and improve trial efficiency. Companies that invest in automation, digital visibility, and sustainable logistics infrastructure are likely to gain a competitive advantage by addressing evolving sponsor expectations while improving cost efficiency and patient outcomes.
One of the key growth opportunities lies in the rising demand for precision therapies, including cell and gene therapies and mRNA-based products, which require advanced cold chain capabilities and highly controlled transportation environments. This is driving investments in cryogenic storage, advanced temperature-controlled packaging, and real-time monitoring solutions to ensure product stability throughout distribution. In parallel, the growing adoption of patient-centric and decentralized clinical trial approaches is increasing the need for direct-to-patient (DTP) delivery, remote monitoring, and flexible last-mile logistics networks. These trends are encouraging 3PL providers to strengthen digital connectivity, develop hybrid distribution models, and enhance global reach. Leading logistics companies such as Marken and DHL are expanding temperature-controlled infrastructure and patient-focused logistics services to address these emerging requirements.
Pricing Model Analysis
Pricing in the clinical trial 3PL market is typically based on a combination of service complexity, shipment volume, temperature requirements, geographic coverage, and value-added capabilities such as real-time tracking and regulatory compliance. Providers generally adopt flexible and customized pricing models, including per-shipment charges, project-based contracts, and long-term strategic outsourcing agreements, depending on the scope and duration of clinical studies. Premium pricing is often associated with specialized services such as cryogenic transportation, decentralized trial logistics, and patient-centric delivery models. In addition, leading logistics players such as World Courier and UPS Healthcare increasingly offer bundled and technology-enabled solutions, which allow sponsors to optimize costs while ensuring service quality, compliance, and operational efficiency.
Service Insights
The logistics & distribution segment dominated the clinical trial 3PL industry, accounting for the largest share of 43.08% in 2025. This is due to increasing trial complexity, globalization, and stringent regulatory requirements. Rising demand for temperature-controlled transportation, just-in-time deliveries, and real-time shipment tracking drives investment in advanced cold chain logistics. Expanding clinical trials across emerging markets requires regional distribution hubs and optimizing supply chain efficiency. Additionally, regulatory compliance, including Good Distribution Practices (GDP), reinforces the need for specialized logistics providers. The shift toward direct-to-patient models and decentralized trials further fuels the demand for flexible, end-to-end distribution solutions.
Manufacturing is anticipated to grow significantly during the forecast period. The segment growth is driven by rising demand for biologics, gene therapies, and personalized medicine. Increased outsourcing to contract manufacturing organizations (CMOs) enhances scalability and cost efficiency. Regulatory-driven demand for GMP-compliant production and rapid clinical trial expansions accelerate market growth, fostering innovation in formulation and packaging solutions.
Phase Insights
The phase III segment accounted for the largest share of the clinical trial third party logistics market in 2025. The high segment growth is attributed to the rise in the number of clinical trial activities, its extensive patient enrollment, complex trial protocols, stringent regulatory requirements, and cost associated with this phase, and a high failure rate in this phase that leads to a loss in terms of both financial as well as human life. High demand for temperature-controlled storage, global distribution networks, and risk mitigation strategies drives investment in specialized logistics.
The phase II segment is expected to grow lucratively over the forecast period. Due to increased trial volumes, complex supply chain needs, and rising demand for temperature-sensitive biologics. Higher patient enrollment, stringent regulatory compliance, and the need for real-time monitoring drive logistics investments. Expanding decentralized trials and adaptive study designs strengthen demand for efficient supply chain solutions in this phase.
Therapeutic Area Insights
The cardiovascular diseases segment held the largest share of the clinical trial third party logistics industry in 2025. With the rise in disease burden, many companies focused on developing novel and innovative medicines to treat heart diseases and developing strategies to reduce the prevalence of heart attacks and strokes. According to the Center for Disease Control and Prevention, in the U.S., 702,880 people died due to heart disease in 2022.
The oncology segment is expected to grow lucratively over the forecast period. This is due to the rising demand for personalized cancer therapies, biologics, and cell & gene treatments. These trials require stringent cold chain logistics, real-time tracking, and regulatory compliance. Increasing global cancer prevalence and rapid drug development drive investment in specialized storage, handling, and distribution, fostering market expansion.
End Use Insights
The pharmaceutical and biotechnology companies segment held the largest share in 2025. The segment growth is owing to increasing R&D investments and complex trial designs. Stringent regulatory compliance, temperature-sensitive drug requirements, and global trial expansions necessitate advanced logistics solutions. The globalization of clinical trials, increased R&D by the pharmaceutical companies, and a large number of clinical trial studies have been conducted by them. For instance, ClinicalTrials.gov currently lists 20,739 active studies being performed in 208 countries.
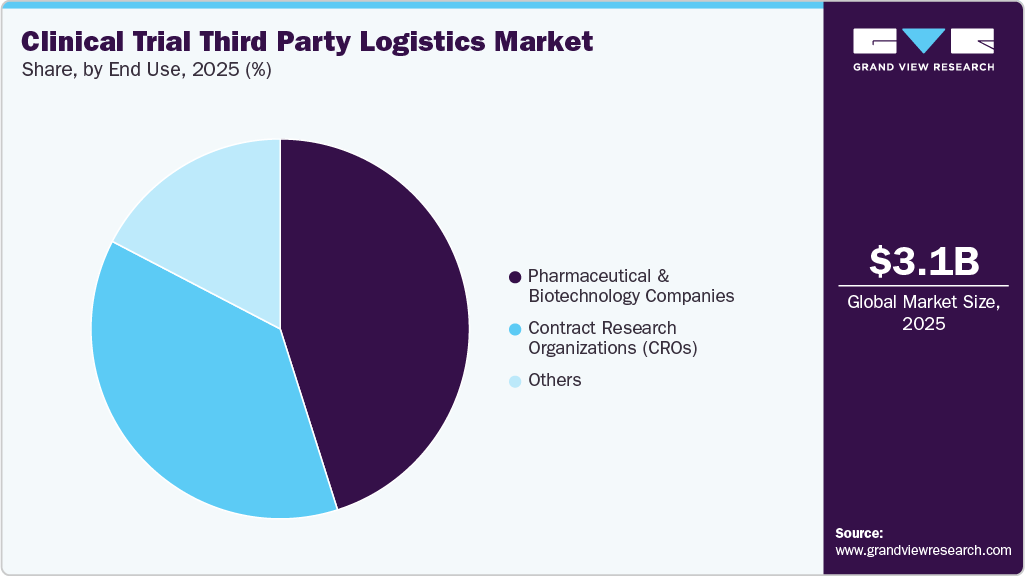
The Contract Research Organizations (CROs) segment is projected to expand at the fastest CAGR over the forecast period. The growth is driven due to the increasing outsourcing of clinical trial operations by pharmaceutical, biotechnology, and medical device companies to enhance cost efficiency, improve operational flexibility, and accelerate drug development timelines. CROs provide end-to-end capabilities, including study design, patient recruitment, site management, regulatory support, and data analytics, enabling sponsors to focus on core innovation and commercialization activities.
Regional Insights
North America dominated the clinical trial third party logistics industry, accounting for a revenue share of 38.26% in 2025. The region accounts for the highest number of clinical trials conducted among all regions, which is a major driver for the market growth. Significant increases in investments in clinical trials and a rise in the number of players in the clinical trial 3PL market are expected to contribute to growth.
U.S. Clinical Trial Third Party Logistics Market Trends
The U.S. clinical trial third party logistics industry dominated North America in 2025. The high revenue growth is primarily driven by high R&D investments, a strong regulatory framework, and the strong presence of key pharmaceutical and biotech players. Integration of advanced cold chain solutions, decentralized trials, and digital supply chain innovations further strengthened market growth. Increasing clinical trial volumes and stringent FDA compliance further accelerate demand for specialized logistics providers.
Europe Clinical Trial Third Party Logistics Market Trends
The growth of the Europe clinical trial third party logistics industry is driven by stringent EU GDP (Good Distribution Practice) regulations, a robust pharmaceutical landscape, and increasing cross-border trials. Strong government support for R&D, rising demand for biologics, and expansion in emerging markets further accelerate logistics innovation. The presence of significant logistics hubs, particularly in Germany, the UK, and Italy, strengthens market competitiveness and supply chain flexibility.
The Germany clinical trial third party logistics market growth is driven due to its strong pharmaceutical industry, advanced infrastructure, and strategic location as a logistics hub. Stringent GDP regulations and high investment in biologics and personalized medicine drive demand for specialized cold chain solutions. The country’s efficient regulatory framework and increasing decentralized trials and digital tracking adoption enhance supply chain transparency. Germany's focus on innovation and automation further strengthens its position as a key market in the clinical trial third party logistics industry.
The clinical trial third party logistics market in the UK is anticipated to grow at a significant CAGR over the forecast period. The market growth is due to a strong pharmaceutical ecosystem, government-funded R&D initiatives, and a well-regulated supply chain framework. Post-Brexit regulatory adaptations have increased demand for specialized logistics solutions to navigate new compliance requirements. Strategic investments in regional distribution hubs and partnerships with global logistics providers further position the UK as a key market in the clinical trial third party logistics industry.
Asia Pacific Clinical Trial Third Party Logistics Market Trends
The Asia Pacific clinical trial third party logistics industry is expected to grow at the fastest CAGR during the forecast period, due to the rising clinical trial activities, lower operational costs, and a growing pharmaceutical sector. Countries like China, India, and Japan are key hubs, attracting global sponsors with diverse patient populations and regulatory reforms. Increased demand for cold chain logistics, decentralized trials, and local regulatory expertise drives market growth.
The China clinical trial third party logistics market held the largest share of Asia Pacific in 2025. The market growth is owing to favorable regulatory reforms, growing biopharmaceutical investments, and a rising number of multinational trials. The China National Medical Products Administration (NMPA) has accelerated drug approval processes, boosting demand for specialized cold chain logistics and localized distribution networks. Rapid growth in cell and gene therapies, along with increasing domestic pharmaceutical innovation, drives the need for advanced temperature-controlled solutions. Expanding clinical research hubs in Beijing, Shanghai, and Guangzhou further strengthen market growth.
The clinical trial third party logistics market in India is expected to grow at the fastest CAGR over the forecast period. The market growth is driven by cost-efficient operations, a large patient pool, and favorable regulatory reforms. Growing investments in biologics, biosimilars, and vaccine research are fueling demand for cold chain logistics and real-time monitoring solutions. The rise of clinical research outsourcing (CROs) and government initiatives promoting clinical trials enhance supply chain efficiency.
The Japan clinical trial third party logistics market is expected to grow at the fastest CAGR over the forecast period, driven by advanced pharmaceutical R&D, stringent regulations, and strong demand for personalized medicine. The country’s focus on regenerative therapies, cell & gene treatments, and aging-related research necessitates specialized cold chain logistics and real-time tracking.
Latin America Clinical Trial Third Party Logistics Market Trends
The Latin America region is expected to grow at a significant CAGR over the estimated timeframe. Latin America is experiencing rapid growth in clinical trial sites due to its diverse patient pool, easy patient access, geographical proximity to North America, and cost-effective transportation. Key markets such as Colombia, Brazil, Peru, Argentina, and Chile accounted for 90% of regional trials. Regulatory reforms in Brazil, China, and Argentina aimed at expediting trial approvals are further driving the market expansion.
The Brazil clinical trial third party logistics market held the largest share of Latin America in 2025. The market growth is owing to high clinical trial activities, low operational cost, ease in patient recruitment, and presence of a diverse patient population. Rising demand for vaccine trials, biologics, and biosimilars fuels the need for cold chain logistics and regional distribution networks. Growth in public-private partnerships and expanding research hubs in Sao Paulo and Rio de Janeiro further drive market growth opportunities.
Key Clinical Trials Third Party Logistics Company Insights
Some of the players operating in the market includeThermo Fisher Scientific (Patheon), Catalent, Inc., Parexel, UPS Healthcare, Piramal Pharma Solutions, Almac Group, UDG Healthcare, FedEx, Movianto, DHL, and Packaging Coordinators Inc. The players are continuously involved in expanding their facilities, collaboration, and engaging in partnership, merger and acquisition of companies. These are key strategic initiatives that are influencing industry dynamics. For instance, in July 2024, Catalent, Inc. completed the expansion of its clinical supply facility in Schorndorf, Germany, strengthening its packaging, storage, and distribution capabilities. The facility enhanced clinical supply services to support global drug development and patient access to innovative treatments.
Key Clinical Trial Third Party Logistics Companies:
The following key companies have been profiled for this study on the clinical trial third party logistics market.
- Thermo Fisher Scientific Inc.
- UPS Healthcare
- Piramal Pharma Solutions
- DHL
- Parexel International
- Almac Group
- Movianto
- UDG Healthcare
- FedEx
- Catalent, Inc.
Recent Developments
-
In June 2024, Thermo Fisher Scientific launched a new ultra-cold facility in Bleiswijk, Netherlands. The cGMP-compliant site supports cell and gene therapies, biologics, antibodies, and vaccines, enhancing contract manufacturing and specialty logistics. It offers biorepository solutions and critical material storage, catering to biotech and pharmaceutical firms across all clinical trial phases.
-
In March 2024, Myonex announced the acquisition of Creapharm’s Clinical Packaging & Distribution, Commercial Packaging, and Bioservices divisions, expanding its global presence. This acquisition strengthens Myonex’s clinical packaging, labeling, and distribution capabilities, while Creapharm adds expertise in commercial packaging and ATMP supply chain management. The deal enhanced both companies' market position in clinical trial third party logistics market.
Clinical Trial Third Party Logistics Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 3.37 billion
Revenue forecast in 2033
USD 5.93 billion
Growth rate
CAGR of 8.51% from 2026 to 2033
Actual estimates/Historical data
2021 - 2025
Forecast period
2026 - 2033
Market representation
Revenue in USD million/billion and CAGR from 2026 to 2033
Segments covered
Service, phase, therapeutic area, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; South Korea; Thailand; Brazil; Argentina; Colombia; South Africa; Saudi Arabia; UAE; Kuwait, Oman, Qatar
Key companies profiled
Thermo Fisher Scientific Inc.; UPS Healthcare; Piramal Pharma Solutions; DHL; Parexel International; Almac Group; Movianto; UDG Healthcare; FedEx; Catalent, Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Clinical Trial Third Party Logistics Market Report Segmentation
This report forecasts revenue growth at the global, regional, and country level and provides an analysis on the latest trends and opportunities in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the clinical trial third party logistics market report based on service, phase, therapeutic area, end use, and region:
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Logistics & Distribution
-
Storage & Retention
-
Packaging, Labeling, and Blinding
-
Comparator Sourcing
-
Other Services
-
-
Phase Outlook (Revenue, USD Million, 2021 - 2033)
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Therapeutic Area Outlook (Revenue, USD Million, 2021 - 2033)
-
Oncology
-
Cardiovascular Diseases
-
Respiratory Diseases
-
CNS and Mental Disorders
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical and Biotechnology Companies
-
Contract Research Organizations (CROs)
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
Colombia
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
Oman
-
Qatar
-
-
Frequently Asked Questions About This Report
b. The global clinical trial third party logistics market size was estimated at USD 3.13 billion in 2025 and is expected to reach USD 3.37 billion in 2026.
b. The global clinical trial third party logistics market is expected to grow at a compound annual growth rate of 8.51% from 2026 to 2033 to reach USD 5.93 billion by 2033.
b. North America dominated the clinical trial third party logistics market with a share of 38.26% in 2025. This is attributable to significant increases in investments in clinical trials and a rise in the number of players in the clinical trial third party logistics market are expected to contribute to growth.
b. Some key players operating in the clinical trial third party logistics market include UPS Healthcare Piramal Pharma Solutions DHL Parexel International Almac Group Movianto UDG Healthcare FedEx Catalent, Inc.
b. Key factors that are driving the market growth include growing globalization of clinical research, rising number of ongoing and planned clinical trials, and increasing R&D spending by pharmaceutical and biopharmaceutical companies
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










