- Home
- »
- Medical Devices
- »
-
Drug-coated Balloon Catheter Market, Industry Report, 2033GVR Report cover
![Drug-coated Balloon Catheter Market Size, Share & Trends Report]()
Drug-coated Balloon Catheter Market (2026 - 2033) Size, Share & Trends Analysis Report By Type (Coronary DCB, Peripheral DCB), By End Use (Hospitals, Specialty Cardiac/Vascular Centers, Ambulatory Surgical Centers (ASCs)), By Region, And Segment Forecasts
- Report ID: GVR-4-68040-085-4
- Number of Report Pages: 200
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Drug-coated Balloon Catheter Market Summary
The global drug-coated balloon catheter market size was estimated at USD 1.2 billion in 2025 and is projected to reach USD 2.3 billion by 2033, growing at a CAGR of 8.3% from 2026 to 2033. The rising prevalence of cardiovascular diseases and increasing incidence of coronary and peripheral artery disease continue to boost the need for practical, minimally invasive vascular interventions, which are key drivers of market growth.
Key Market Trends & Insights
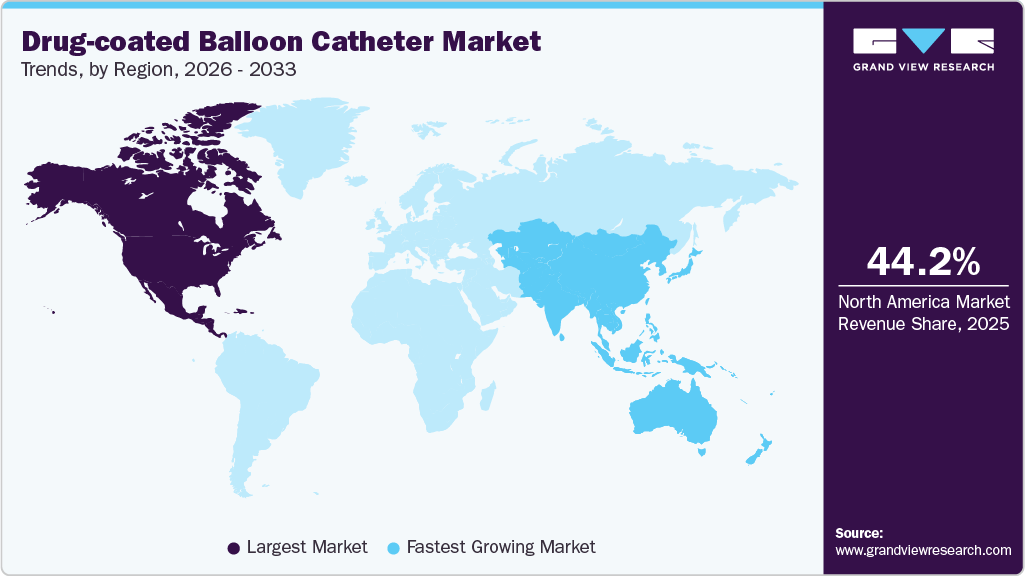
- North America drug-coated balloon catheter industry dominated the global market in 2025 with a revenue share of 44.2%.
- The U.S. market accounted for the largest share in North America in 2025.
- By type, the peripheral DCB segment accounted for the largest revenue share of over 71.4% in 2025.
- By end use, the hospitals segment accounted for the largest revenue share of 68.3% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 1.2 Billion
- 2033 Projected Market Size: USD 2.3 Billion
- CAGR (2026-2033): 8.3%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
According to Oxford Academic in January 2025, the global age-standardized prevalence of CVD is estimated at approximately 7,179 cases per 100,000 population, highlighting the persistent global burden of heart-related disorders. This high prevalence is driving demand for drug coated balloon catheters, which offer an effective, minimally invasive option for treating vascular narrowing and reducing restenosis in patients with coronary and peripheral artery disease, thereby supporting their growing adoption in interventional cardiology.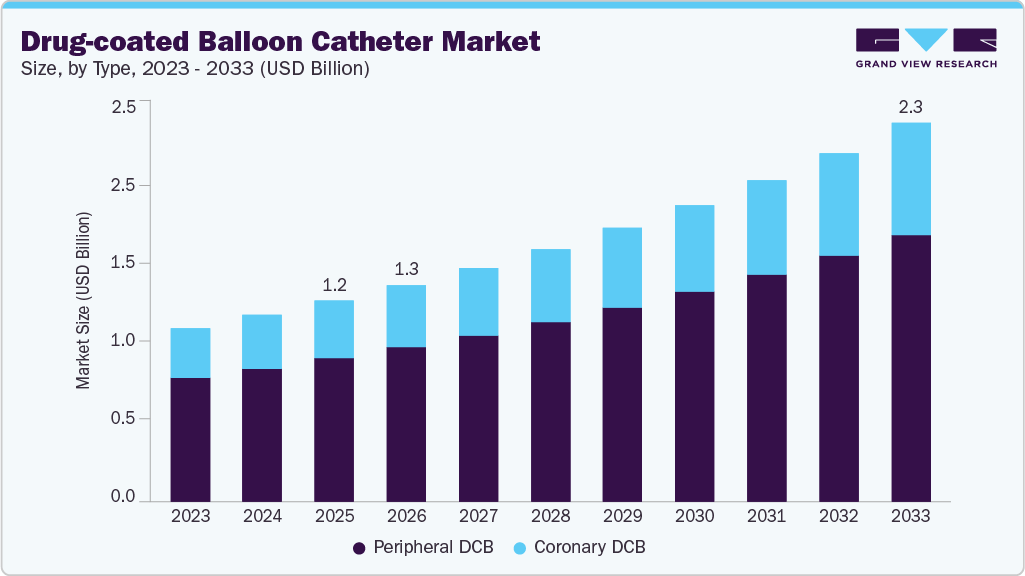
The market is witnessing strong growth driven by the rising prevalence of vascular diseases such as peripheral artery disease (PAD), increasing clinical adoption supported by superior outcomes over conventional balloons, and expanding use across hospitals and specialized cardiac centers. These devices offer effective revascularization without leaving permanent implants, making them a preferred minimally invasive treatment option. Advancements in imaging and the integration of AI-enabled procedural guidance are enhancing treatment precision and physician confidence, further accelerating the adoption of DCB technologies across interventional cardiology practices.
Increasing prevalence of Peripheral Artery Disease (PAD)
The increasing prevalence of Peripheral Artery Disease (PAD) remains a key driver of the drug-coated balloon (DCB) catheter industry. According to the American Limb Preservation Society. article published in July 2025, the projected 220% increase in peripheral artery disease (PAD) cases worldwide by 2050, with prevalence expected to rise from 113 million in 2021 to over 360 million, highlights a rapidly expanding patient population requiring vascular intervention. This rise is fuels demand for drug-coated balloon catheters, which are widely used in PAD treatment to restore vessel patency and reduce restenosis without leaving permanent implants, making them a preferred option for managing the growing PAD burden.
Growing adoption of drug-coated balloon catheters
The growing adoption of drug-coated balloon catheters is driven by strong clinical evidence demonstrating superior outcomes compared with conventional uncoated balloons in coronary interventions. According to a JAMA article published in March 2024, in a multicenter randomized U.S. trial involving 600 patients undergoing percutaneous coronary intervention for in-stent restenosis, treatment with a paclitaxel-coated balloon resulted in significantly lower 1 year target lesion failure (17.9%) compared with 28.6% in patients treated with uncoated balloons. The study also showed lower rates of ischemia-driven target-lesion revascularization (13.0% vs. 24.7%) and target vessel-related myocardial infarction (5.8% vs. 11.1%), emphasizing clinicians' confidence in paclitaxel-coated balloons as an effective and minimally invasive treatment strategy. This clear clinical advantage is accelerating the adoption of drug-coated balloon catheters in routine coronary practice, thereby fueling market growth.
Market growth is influenced by evolving hospital, outpatient, and specialty cardiac facility adoption, supported by streamlined distribution channels and regulatory approvals. Reimbursement frameworks, physician training, and integration of advanced devices into standard-of-care workflows are key factors facilitating wider accessibility and adoption across interventional cardiology settings. In June 2025, Infirmary Health revealed that Mobile Infirmary in Alabama became the first hospital in the region to use the newly FDA-approved AGENT Drug-Coated Balloon for treating coronary in-stent restenosis. The procedure delivers therapeutic drugs directly to the arterial wall without leaving a permanent implant, reducing complications and preserving future treatment options. This adoption enhances access to advanced cardiovascular care for patients across south Alabama and surrounding areas.
Drug-Eluting Balloon (DEB) Catheters: Innovation-Driven Evolution in Drug Delivery Technologies
Drug-eluting balloon (DEB) catheters represent a key technological evolution within the broader drug-coated balloon landscape, characterized by their ability to deliver antiproliferative drugs directly to the vessel wall during balloon inflation without leaving behind a permanent implant. While the terms DEB and DCB are often used interchangeably, the DEB nomenclature is increasingly associated with next-generation drug delivery approaches, particularly those involving controlled and sustained release mechanisms. This segment is gaining traction due to the growing shift from traditional Paclitaxel-based coatings toward Sirolimus-eluting technologies, which are designed to address safety concerns, enhance drug retention, and improve long-term clinical outcomes in both coronary and peripheral interventions.
Innovation within the DEB space is being driven by advancements in coating technologies, drug carrier matrices, and release kinetics. Companies such as Translumina have introduced sirolimus-based solutions such as the Protégé DEB, which focuses on localized drug delivery with no residual scaffold, while Cardionovum GmbH offers the Restore DEB, a paclitaxel-coated balloon widely used in coronary applications such as in-stent restenosis and small vessel disease. Similarly, Cordis has developed the Selution SLR DEB, which utilizes sustained limus release (SLR) technology to enable controlled drug dispersion over time, expanding its applicability across both coronary and peripheral vascular indications.
The increasing adoption of DEBs is supported by their clinical advantages, including reduced risk of chronic inflammation, elimination of long-term implant-related complications, and suitability for complex lesion subsets where stent placement may not be ideal. As a result, DEBs are being increasingly utilized in indications such as in-stent restenosis, small vessel disease, and peripheral artery disease. Furthermore, ongoing clinical studies and regulatory approvals are reinforcing confidence in sirolimus-based DEB platforms, positioning them as a strong alternative to conventional treatment modalities.
Technology Trend - AI Integration in Drug-Coated Balloon Catheter Procedures.
The industry is increasingly influenced by artificial intelligence enabled imaging and procedural support technologies that improve precision and decision-making during interventions. According to the NCBI article published in November 2025, AI algorithms are being integrated with advanced intravascular imaging tools such as intravascular ultrasound (IVUS) and optical coherence tomography (OCT) to provide real-time interpretation of vessel anatomy, plaque characteristics, and lesion morphology, enabling clinicians to better plan and execute balloon-based therapies with greater accuracy and safety. This trend reflects a broader shift toward data driven, personalized interventional cardiology practices that optimize device placement and predict potential complications based on patient specific imaging patterns.
For instance, AI-assisted imaging platforms powered by machine learning and deep learning models enhance real time guidance during catheter procedures by rapidly analyzing complex intravascular images to help physicians identify optimal treatment sites and adjust intervention strategies on the fly.
AI-enabled tool
Company
Modality
AI capability
Clinical implication
AVVIGO+ Multimodality Guidance System, Version: Not specified
Boston Scientific, Marlborough, MA, USA
IVUS
Lumen and vessel segmentation, plaque burden detection, length measurement
Stent/balloon sizing, lesion length assessment, landing site selection, post-PCI optimization
Ultreon 2.0, Version: 2.0
Abbott Vascular, Santa Clara, CA, USA
OCT
EEL detection, calcium arc/thickness/length recognition
Stent/balloon sizing, calcium modification planning
Market Concentration & Characteristics
The drug-coated balloon catheter industry exhibits a high degree of innovation, driven by advances in drug-delivery technologies, balloon design, and coating formulations that support a leave nothing behind treatment approach. For instance, in March 2024, Boston Scientific launched the AGENT paclitaxel-coated balloon catheter, the first FDA-approved coronary drug-coated balloon in the U.S. for the treatment of coronary in-stent restenosis. This regulatory approval and commercial launch expanded minimally invasive treatment options by enabling effective vessel therapy without the need for additional permanent stent implantation.
The drug-eluting balloon catheter industry is witnessing a moderate level of merger and acquisition (M&A) activity, as companies seek to enhance technological innovation and expand their global presence. For instance, in July 2025, Teleflex completed the acquisition of BIOTRONIK’s Vascular Intervention business, which includes drug-coated balloon catheters, such as the Pantera Lux DCB, as part of its portfolio. This strategic deal expands Teleflex’s presence in coronary and peripheral intervention devices and strengthens its global footprint in catheter-based therapies.

The drug-eluting balloon catheter market operates under a moderate regulatory environment, requiring approvals such as FDA clearance in the U.S. and CE marking in Europe, along with adherence to regional medical device regulations worldwide. While these requirements help ensure safety, effectiveness, and clinical reliability, they increase development expenses and extend approval timelines. Moreover, regulatory bodies are evolving their frameworks to support emerging digital and software enabled technologies, encouraging innovation while maintaining high patient safety standards.
There are no direct substitutes for drug-coated balloon catheters that offer the same combination of targeted drug delivery and leave nothing behind treatment approach; however, alternative therapies such as drug-eluting stents and conventional balloon angioplasty are used in similar clinical settings and may limit DCB adoption in some instances where permanent vessel support is preferred.
The drug-coated balloon catheter market is experiencing steady regional expansion as manufacturers strengthen their presence in high-growth regions such as Asia-Pacific, Latin America, and the Middle East. Companies are prioritizing local regulatory approvals, distribution partnerships, and physician training to improve access to advanced endovascular therapies. For instance, in 2025, MicroPort received market approval in China for its Firelimus sirolimus coated drug-coated balloon, supporting broader commercialization across the Asia Pacific region. Such regional expansions are improving the availability of minimally invasive vascular treatments in emerging healthcare systems and reinforcing long-term global market growth.
Type Insights
By type, the peripheral DCB segment accounted for the largest revenue share of over 71.4% in 2025, driven by the increasing prevalence of peripheral artery disease (PAD) and rising adoption of minimally invasive endovascular therapies, particularly for femoropopliteal lesions. According to the NCBI article published in November 2025, clinical evidence supports the effectiveness of newer sirolimus-coated technologies; for instance, the 2025 SELUTION SFA Japan study evaluated the SELUTION SLR sirolimus-coated balloon in 134 patients (136 lesions) and reported primary patency of 81.5% at 36 months along with 93.8% freedom from target lesion revascularization, representing sustained efficacy and safety in peripheral interventions. These outcomes are contributing to growing physician confidence and increasing use of DCBs in routine clinical practice.
The coronary DCB segment is expected to grow at the fastest CAGR over the forecast period, driven by the rising CVD cases and growing clinical adoption are driven by strong real-world evidence supporting its effectiveness in reducing restenosis while maintaining a leave-nothing-behind treatment strategy. For instance, in October 2025, the Transcatheter Cardiovascular Therapeutics (TCT) conference, where Boston Scientific reported findings from a multi-center observational registry involving more than 12,000 patients treated with the Agent paclitaxel-coated balloon catheter across over 700 U.S. sites. The data established strong safety outcomes and rapid therapy uptake in routine clinical practice, reflecting growing confidence among interventional cardiologists in the paclitaxel-coated DCB approach for managing in-stent restenosis and other coronary lesions.
End Use Insights
By end use, the hospitals segment accounted for the largest revenue share of 68.3% in 2025. Hospitals are increasingly driving adoption of drug-coated balloon catheters as they expand minimally invasive treatment options for complex coronary cases. For instance, in February 2025, UPMC Heart and Vascular Institute, where clinicians began routinely using the AGENT drug-coated balloon for the treatment of coronary in-stent restenosis following its FDA approval. The hospital integrated the device into clinical practice after participating in earlier clinical evaluations, using it as an alternative to repeat stenting to improve patient outcomes while avoiding additional metal implantation. This example highlights how large, specialized hospitals are accelerating market growth by rapidly translating regulatory approvals into real world clinical use.
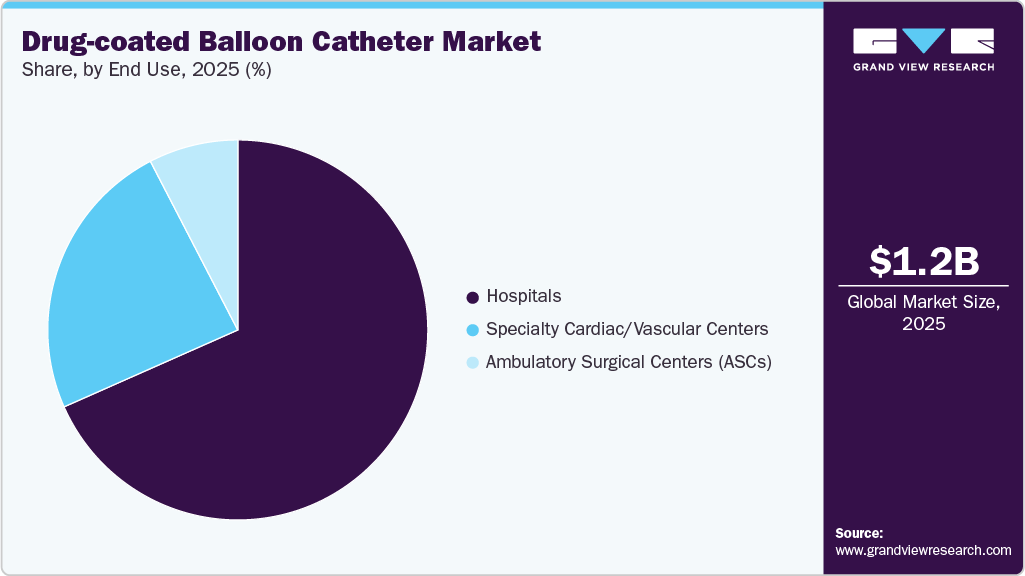
The ambulatory surgical centers segment is expected to grow at the fastest CAGR over the forecast period. The ambulatory surgical centers (ASCs) are increasingly performing minimally invasive vascular procedures as outpatient care expands. Facilities with catheterization labs and trained interventional teams are adopting drug-coated balloon (DCB) procedures, particularly for lower-risk femoropopliteal PAD cases, driven by benefits such as shorter hospital stays and faster patient recovery. For instance, in June 2024, OhioHealth Heart and Vascular services, which operate catheterization capabilities outside traditional inpatient hospital units and have been an early adopter of drug-coated balloon angioplasty procedures, reported being among the first to offer FDA-approved DCB therapy for in stent restenosis as part of their expanded outpatient cardiovascular care offerings. This reflects a growing trend of centers offering advanced DCB-based therapies outside the conventional inpatient setting as outpatient procedural volume increases.
Regional Insights
North America drug-coated balloon catheter industry dominated the global market with a share of 44.2% in 2025. Rising CVD cases drive market growth. According to the CDC article published in October 2024, in the U.S., cardiovascular disease remains a leading public health concern in the U.S., with one person dying from cardiovascular disease every 34 seconds. The report also notes that in 2023, about 919,032 people died from cardiovascular disease, underscoring the continued high burden of heart and circulatory conditions in the U.S. population. This sustained disease burden continues to drive demand for effective, minimally invasive treatments such as drug-coated balloon catheters to manage coronary and peripheral artery disease and reduce repeat vascular complications.
U.S. Drug-coated Balloon Catheter Market Trends
The U.S. drug-coated balloon catheter industry accounted for the largest market share in North America in 2025. Increasing CVD cases and the launch of technologically advanced products drive market growth. For instance, in March 2025, the Prevail drug-coated balloon was marketed in more than 80 countries worldwide, reflecting its broad international adoption. In the U.S., the device advanced into clinical development after receiving FDA approval for an Investigational Device Exemption in October 2024, allowing Medtronic to initiate the Prevail Global study, which has since enrolled its first patient.
Europe Drug-coated Balloon Catheter Market Trends
Europe drug-coated balloon catheter industry is growing over the forecast period. Rising CVD incidence and technologically advanced product launch drives the growth of the market. According to Oxford University Press article published in September 2025, cardiovascular disease remains the leading cause of death in Europe and globally, affecting more than 60 million people across Europe, and placing a substantial burden on healthcare systems. A large proportion of this population suffers from coronary and peripheral artery diseases that require ongoing vascular intervention to restore blood flow and prevent disease progression. This growing patient pool underscores the need for effective, minimally invasive treatment options to manage arterial narrowing and reduce repeat procedures.
UK drug-coated balloon catheter industry is growing over the forecast period. Increasing cases of coronary artery disease drive market growth. According to British Journal of Cardiology article published in August 2025, acute coronary syndrome continues to represent a significant cardiovascular burden in the UK, affecting approximately 93,500 individuals each year. This high incidence reflects the ongoing prevalence of coronary artery disease and the need for timely and effective interventional management to restore blood flow and prevent recurrent events. While healthcare providers seek to optimize outcomes while reducing procedural complexity and long-term complications, drug-coated balloon catheters are increasingly being considered in selected coronary interventions, particularly for managing in-stent restenosis and specific lesion types, supporting minimally invasive treatment strategies within the ACS care pathway.
Germany drug-coated balloon catheter industry is growing over the forecast period. The increasing incidence of peripheral vascular disease drives market growth. According to NCBI article published in August 2025, peripheral vascular disease represents a notable clinical burden in Germany, with an incidence of 145.6 cases per 100,000 person-years, highlighting the widespread presence of arterial disease within the population. This high disease incidence is driving demand for effective endovascular treatments, including drug coated balloon catheters, which are increasingly used to manage peripheral artery lesions by reducing restenosis and supporting minimally invasive intervention approaches.
Asia Pacific Drug-coated Balloon Catheter Market Trends
The Asia-Pacific drug-coated balloon catheter industry has witnessed a growing demand for drug-coated balloon catheters, driven by the increasing prevalence of cardiovascular and peripheral artery diseases across countries such as China, India, Japan, and South Korea. Growing adoption of minimally invasive endovascular procedures is being enabled by improving healthcare infrastructure, expansion of catheterization laboratories, and greater physician awareness of advanced vessel preserving treatment options. In parallel, leading global and regional players, including Medtronic, Bayer AG, B. Braun SE, and Eurocor GmbH, are strengthening their presence through hospital partnerships, clinical studies, and localized manufacturing or technology transfer initiatives. These efforts are improving access to drug-coated balloon therapies across both urban and semi-urban healthcare settings, supporting broader regional market growth.
China drug-coated balloon catheter industry accounted for the largest market share in the Asia Pacific region in 2025. Increasing CVD cases drive market growth. According to the Beijing Renhe Information Technology Co. Ltd article published in August 2025, in China, cardiovascular disease continues to pose a significant public health challenge, affecting an estimated 330 million people nationwide. Among this population, around 13 million individuals have experienced stroke, while approximately 11.4 million are living with coronary heart disease, underscoring the widespread burden of arterial and vascular disorders. This growing prevalence is increasing the number of patients requiring interventional management, thereby driving demand for effective, minimally invasive solutions such as drug-coated balloon catheters, which are increasingly used to treat coronary and peripheral artery disease and reduce repeat vessel narrowing in China’s expanding cardiovascular care landscape.
India drug-coated balloon catheter industry is expected to grow at a significant CAGR over the forecast period. The rising CVD burden in India is driving market growth. According to the Economic Times article published in February 2024, India is facing a steadily rising cardiovascular disease burden, reflected in an age-standardized mortality rate of about 272 deaths per 100,000 population, which is notably higher than the global average of 235 per 100,000. This elevated disease burden has led to an increasing number of patients requiring coronary and peripheral vascular interventions. Moreover, demand for drug-coated balloon catheters is growing, as these devices offer a minimally invasive approach to managing arterial disease, reducing restenosis, and improving long term vascular outcomes in India’s expanding cardiovascular care ecosystem.
Latin America Drug-coated Balloon Catheter Market Trends
The drug-coated balloon catheter industry in Latin America is expected to witness significant growth over the forecast period, due to the incidence of cardiovascular and peripheral artery diseases and the increasing adoption of minimally invasive endovascular procedures across countries such as Brazil and Argentina. Hospitals and specialized cardiac centers are expanding catheterization capabilities and integrating advanced vessel-preserving therapies to improve clinical outcomes.
Brazil drug-coated balloon catheter industry is expected to witness significant growth over the forecast period. Rising CVD incidence is driving market growth. According to an Elsevier Ltd. article published in July 2025, cardiovascular diseases continue to represent a significant public health challenge in Brazil, accounting for around 30% of all deaths and remaining the leading cause of mortality over the past several decades, except during the COVID-19 pandemic years of 2020 and 2021. This sustained burden of heart and vascular disease has led to a growing number of patients requiring coronary and peripheral interventions. Moreover, demand for drug-coated balloon catheters is increasing, as these devices offer effective, minimally invasive treatment options to manage arterial blockages, reduce restenosis, and improve long-term vascular outcomes within Brazil’s expanding cardiovascular care landscape.
MEA Drug-coated Balloon Catheter Market Trends
The drug-coated balloon catheter industry in the MEA is growing significantly over the forecast period, driven by the increasing incidence of cardiovascular and peripheral artery diseases and the widespread presence of risk factors such as obesity, diabetes, and hypertension. Countries including South Africa, Saudi Arabia, the UAE, and Kuwait are strengthening cardiac care infrastructure, with hospitals and specialized centers expanding endovascular treatment capabilities. In parallel, leading manufacturers such as Medtronic, Bayer AG, BIOTRONIK, and B. Braun SE are emphasizing their regional presence through the introduction of advanced drug-coated balloon technologies, physician training initiatives, and clinical support programs. These efforts are improving procedural expertise, expanding patient access to minimally invasive therapies, and supporting long-term market growth across the MEA region.
South Africa drug-coated balloon catheter industry is growing significantly over the forecast period. Increasing CVD incidence in South Africa is driving market growth. According to an NCBI article published in May 2024, in South Africa, cardiovascular disease remains a significant public health burden, responsible for about 17.3% of all deaths, or nearly one in every six fatalities. On average, more than 200 people die each day from heart disease or stroke, underscoring the sustained prevalence of vascular disorders in the country. This high disease burden is driving greater demand for effective, minimally invasive treatments such as drug-coated balloon catheters, which are increasingly used to manage coronary and peripheral artery disease and reduce repeat vascular complications within South Africa’s healthcare system.
Key Drug-coated Balloon Catheter Company Insights
Leading players in the drug-coated balloon catheter industry such as Boston Scientific Corporation, B. Braun SE, and Medtronic have strategically employed innovative approaches, including mergers and acquisitions, market penetration initiatives, partnerships, and distribution agreements. These strategies aim to enhance their revenue streams by leveraging collaborative efforts, expanding market reach, and fostering synergies within the dynamic landscape of drug-coated balloon catheters. Emerging market entrants such as iVascular S.L.U. and SMT are directing their efforts toward broadening their market presence, creating inventive technologies, and establishing strategic partnerships as part of their strategy to contend with established industry leaders.
Key Drug-coated Balloon Catheter Companies:
The following key companies have been profiled for this study on the drug-coated balloon catheter market.
- Boston Scientific Corporation
- Medtronic
- B. Braun SE
- Concept Medical
- Abbott
- Koninklijke Philips N.V.
- Teleflex Incorporated (BIOTRONIK)
- Translumina
- MicroPort Scientific Corporation
- Cardionovum GmbH
- iVascular S.L.U.
- Lepu Medical
- Meril Life Sciences Pvt. Ltd.
- Cordis
- Acotec Scientific (Acotec / Acotec Scientific AG)
- SMT
Recent Developments
-
In October 2025, an international study led by researchers at Mount Sinai demonstrated that a new sirolimus-eluting balloon performed comparably to conventional treatments for in-stent restenosis, highlighting the ongoing evolution of DCB technologies.
-
In October 2025, ANVISA approved Cardionovum’s Aperto over‑the‑wire (OTW) drug‑coated balloon for commercial use, enabling dialysis patients to access a therapy that prolongs arteriovenous fistula lifespan and improves treatment outcomes.
-
In July 2025, Teleflex Incorporated announced it has entered into a definitive agreement to acquire substantially all of the Vascular Intervention business of BIOTRONIK for an estimated cash payment of approximately EUR 760 million (USD 892.15 million).
-
In February 2025, BrosMed and Cordis have entered into a strategic collaboration to support the commercialization and market expansion of the VaSecure next-generation drug-coated balloon in China. Through this partnership, the companies aim to strengthen distribution, enhance clinical adoption, and improve access to advanced drug-coated balloon technology within the China market.
-
In October 2023, Cordis completed its acquisition of MedAlliance, enhancing its drug-eluting balloon (DEB) portfolio with SELUTION SLR sustained sirolimus technology.
Drug-coated Balloon Catheter Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 1.3 billion
Revenue forecast in 2033
USD 2.3 billion
Growth rate
CAGR of 8.3% from 2026 to 2033
Actual Data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD billion/million, volume in thousand units and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Vietnam; Malaysia; Indonesia; Japan; China; India; Australia; Thailand; South Korea; Brazil; Argentina; Colombia; Chile; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Boston Scientific Corporation; Medtronic; B. Braun SE; Concept Medical; Abbott; Koninklijke Philips N.V.; Teleflex Incorporated (BIOTRONIK); Translumina; MicroPort Scientific Corporation; Cardionovum GmbH; iVascular S.L.U.; Lepu Medical; Meril Life Sciences Pvt. Ltd.; Cordis; Acotec Scientific (Acotec / Acotec Scientific AG); SMT
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Drug-coated Balloon Catheter Market Report Segmentation
This report forecasts revenue growth at global, regional & country levels and provides an analysis of the industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global drug-coated balloon catheter market report based on type, end use, and region:

-
Type Outlook (Unit Volume, Thousand Units; Revenue, USD Million, 2021 - 2033)
-
Coronary DCB
-
By Indication
-
In-Stent Restenosis (ISR)
-
De-novo coronary lesions
-
Complex / Adjunct Coronary Lesions
-
-
By Drug Type
-
Paclitaxel
-
Sirolimus
-
Others
-
-
-
Peripheral DCB
-
By Indication
-
Femoropopliteal Artery Disease (PAD)
-
Below-the-Knee (BTK)
-
-
By Drug Type
-
Paclitaxel
-
Sirolimus
-
Others
-
-
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Specialty Cardiac/Vascular Centers
-
Ambulatory Surgical Centers (ASCs)
-
-
Regional Outlook (Unit Volume, Thousand Units; Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
South Korea
-
Australia
-
Thailand
-
Indonesia
-
Malaysia
-
Vietnam
-
-
Latin America
-
Brazil
-
Argentina
-
Colombia
-
Chile
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global drug-coated balloon catheter market size was estimated at USD 1.2 billion in 2025 and is expected to reach USD 2.3 billion in 2033.
b. Key factors that are driving the market growth include a growing elderly population, rising cardiovascular disease prevalence, and an increase in minimally invasive surgical techniques.
b. The global drug-coated balloon catheter market is expected to grow at a compound annual growth rate of 8.3% from 2026 to 2033 to reach USD 2.3 billion in 2033.
b. Some key players operating in the drug-coated balloon catheter market include Boston Scientific Corporation; Medtronic; B. Braun SE; Concept Medical; Abbott; Koninklijke Philips N.V.; Teleflex Incorporated (BIOTRONIK); Translumina; MicroPort Scientific Corporation; Cardionovum GmbH; iVascular S.L.U.; Lepu Medical; Meril Life Sciences Pvt. Ltd.; Cordis; Acotec Scientific (Acotec / Acotec Scientific AG); SMT.
b. North America dominated the drug-coated balloon catheter market with a share of 44.2% in 2025. This is attributable to rising healthcare awareness coupled with the presence of major market participants, rising cost of healthcare in the U.S. and constant research and development initiatives.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










