- Home
- »
- Pharmaceuticals
- »
-
Estrogen Replacement Therapy Market Size Report, 2033GVR Report cover
![Estrogen Replacement Therapy Market Size, Share & Trends Report]()
Estrogen Replacement Therapy Market (2026 - 2033) Size, Share & Trends Analysis Report By Therapy Type (ET, EPT), By Route Of Administration (Oral, Transdermal), By Indication, By Distribution Channel, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-896-2
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Estrogen Replacement Therapy Market Summary
The global estrogen replacement therapy market size was estimated at USD 11.75 billion in 2025 and is projected to reach USD 18.36 billion by 2033, growing at a CAGR of 5.80% from 2026 to 2033. The market is primarily driven by the rising global population of menopausal and postmenopausal women.
Key Market Trends & Insights
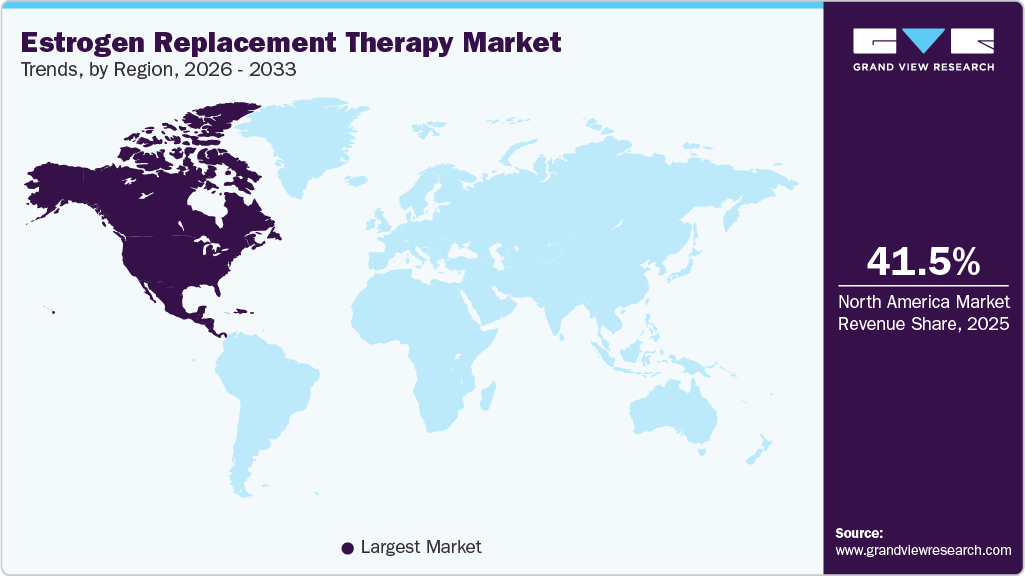
- North America estrogen replacement therapy market held the largest share of 41.46% of the global market in 2025.
- The estrogen replacement therapy industry in the U.S. is expected to grow significantly over the forecast period.
- By therapy type, the combined estrogen + progestin therapy (EPT) segment held the highest market share of 47.43% in 2025.
- By route of administration, the oral segment held the highest market share in 2025.
- By indication, the menopause management segment held the highest market share in 2025.
- By distribution channel, the hospital pharmacies segment held the highest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 11.75 Billion
- 2033 Projected Market Size: USD 18.36 Billion
- CAGR (2026-2033): 5.80%
- North America: Largest market in 2025
- Asia Pacific: Fastest market in 2025
Increasing life expectancy has extended the duration women experience menopause-related symptoms, thereby boosting the demand for long-term hormone management solutions. In addition, greater awareness about menopause-associated health risks, such as osteoporosis and cardiovascular conditions, is encouraging early adoption of ERT. Growing patient inclination toward improving quality of life further supports market expansion. Estrogen replacement therapy continues to play a critical role in managing menopausal symptoms and improving overall quality of life, driving its widespread adoption among women globally. Rising demand for advanced estrogen replacement therapy formulations that offer improved safety profiles and personalized dosing options is boosting innovation and acceptance. Increasing awareness and physician recommendations are also encouraging the use of various delivery formats, including transdermal patches, gels, and oral therapies, to enhance patient adherence and convenience. Pharmaceutical companies are actively investing in R&D for next-generation estrogen replacement therapies, focusing on bioidentical hormones, low-dose regimens, and patient-friendly delivery systems. For instance, in September 2024, Pfizer launched a USD 600,000 competitive medical education grant program to improve clinician knowledge of MHT. This initiative, which accepted applications through October 2024, directly supports market adoption by addressing educational barriers and ensuring evidence-based accessibility for estrogen replacement therapies.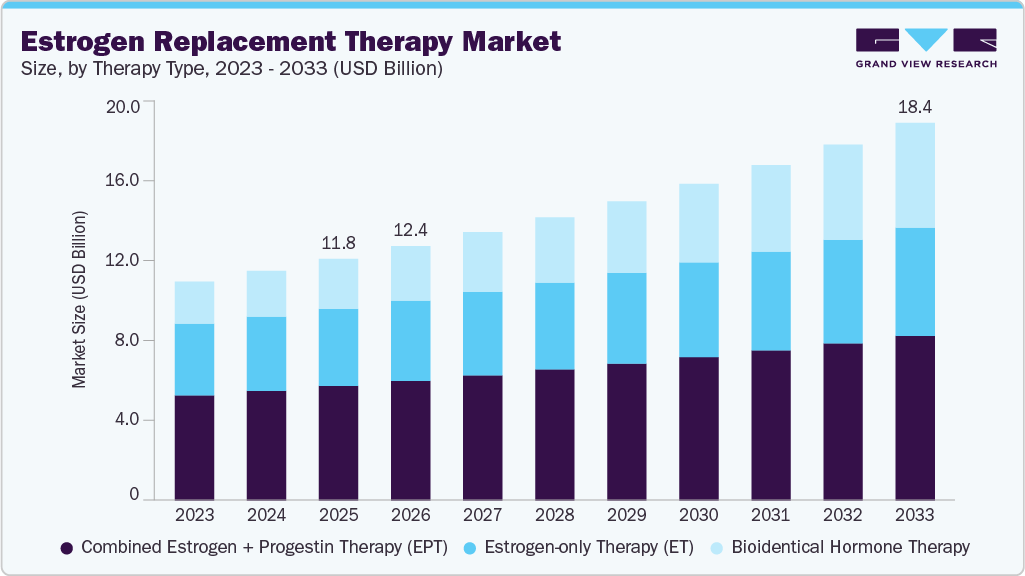
The market is also benefiting from continuous product innovation and strategic initiatives by pharmaceutical companies. New formulations with improved safety profiles and reduced side effects are addressing previous concerns associated with hormone therapy. Moreover, increasing regulatory approvals and ongoing research into bioidentical hormones are expanding treatment options. Companies are engaging in partnerships, product launches, and geographic expansions to strengthen their market presence. This ongoing innovation pipeline is expected to further accelerate the adoption of estrogen replacement therapy globally.
Industry Characteristics
The degree of innovation in the estrogen replacement therapy industry is steadily increasing, driven by advancements in drug formulation and delivery technologies. Companies are focusing on developing bioidentical hormones, transdermal systems, and low-dose therapies to improve safety and efficacy profiles. Continuous research into personalized hormone therapy and combination treatments is further enhancing therapeutic outcomes and patient adherence. Furthermore, innovation in sustained-release formulations and novel delivery routes is supporting the evolution of more convenient and patient-centric estrogen replacement therapy solutions.
The estrogen replacement therapy industry is characterized by stringent regulatory requirements, established pharmaceutical players, and a strong focus on product safety and efficacy. Barriers to market entry are high due to the need for extensive clinical trials, regulatory approvals, and compliance with evolving safety guidelines related to hormone therapies. Significant investment in R&D and manufacturing capabilities further limits the entry of new players. In addition, concerns regarding potential side effects and the need for strong physician trust and brand reputation create additional challenges for market penetration.
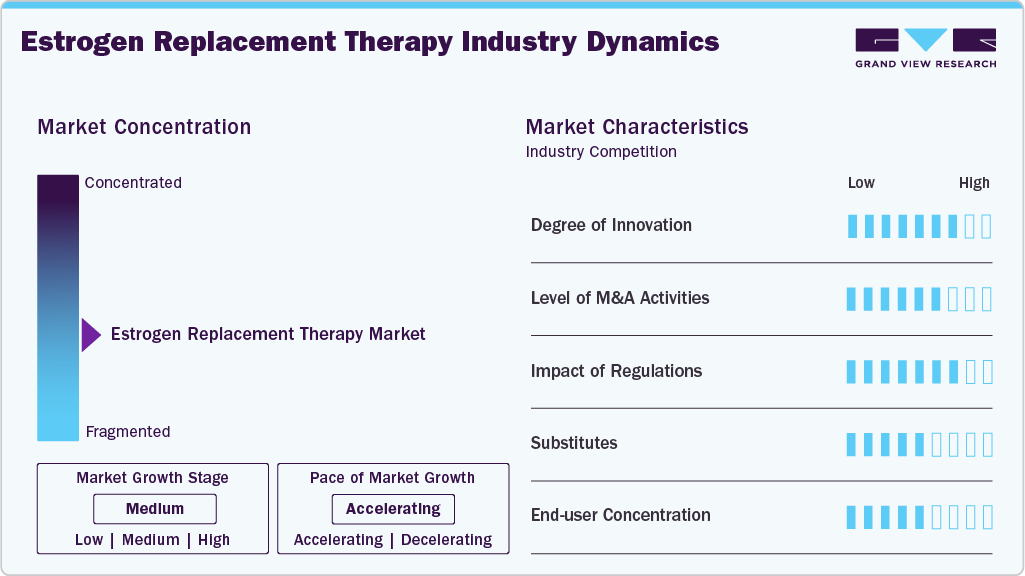
The estrogen replacement therapy industry is characterized by stringent regulatory oversight due to safety concerns associated with hormone-based treatments, particularly regarding risks such as breast cancer and cardiovascular disease. Regulatory authorities such as the U.S. Food and Drug Administration and the European Medicines Agency enforce strict clinical evaluation, labeling requirements, and post-market surveillance. These regulations increase development costs and time-to-market but ensure product safety and efficacy. As a result, companies focus on developing low-dose and safer formulations to comply with evolving regulatory standards while maintaining market competitiveness.
The estrogen replacement therapy industry is characterized by the availability of multiple treatment substitutes, including non-hormonal therapies, lifestyle modifications, and alternative medications for managing menopausal symptoms. Options such as selective serotonin reuptake inhibitors (SSRIs), herbal supplements, and phytoestrogens are increasingly used by patients seeking to avoid hormone-based treatments. Moreover, concerns regarding potential side effects of hormone therapy have encouraged the adoption of these substitutes. This competitive landscape of alternatives influences treatment choices and moderates the overall growth trajectory of the market.
The estrogen replacement therapy industry is characterized by a strong industry focus on geographical expansion to tap emerging opportunities and address unmet medical needs. Pharmaceutical companies are increasingly expanding their presence in regions such as Asia-Pacific, Latin America, and the Middle East & Africa, driven by improving healthcare infrastructure and rising awareness of menopause management. Strategic collaborations with regional distributors and healthcare providers are enabling broader product accessibility and market penetration. Furthermore, regulatory support and growing healthcare investments in these regions are further strengthening the global footprint of estrogen replacement therapy providers.
Therapy Type Insights
The combined estrogen + progestin therapy (EPT) segment dominated the market with the largest revenue share of 47.43% in 2025, driven by its proven effectiveness in managing menopausal symptoms while reducing the risk of endometrial hyperplasia in women with an intact uterus. The increasing prevalence of menopause-related conditions, including osteoporosis and vasomotor symptoms, continues to boost demand for combination therapies, particularly among aging female populations. Clinical evidence suggests hormone therapy can significantly reduce fracture risk and improve quality of life when initiated early in menopause, further supporting adoption. In addition, the availability of multiple combination formulations across oral and transdermal routes has enhanced treatment personalization and adherence. Recent product-level developments include continued utilization and expansion of combination therapies such as Bijuva and other estrogen-progestogen products in clinical practice, supported by evolving safety data. For instance, in February 2026, the U.S. Food and Drug Administration approved labeling updates for multiple menopausal hormone therapy products, including combination therapies, removing outdated boxed warnings to reflect updated scientific evidence and improve patient access.
The bioidentical hormone therapy segment is projected to grow at a CAGR of 9.77% over the forecast period, driven by increasing demand for personalized and physiologically compatible hormone treatments that closely mimic endogenous hormones. Rising awareness among women regarding menopause management and preference for “natural” or body-identical formulations are significantly supporting adoption trends. Furthermore, expanding the availability of regulated, FDA-approved bioidentical products is improving treatment reliability and addressing earlier safety concerns associated with compounded therapies. Growing investments in women’s health platforms and digital care models are further accelerating patient access and consultation for tailored hormone therapies. Regulatory developments are also playing a crucial role in shaping market growth by improving physician confidence and patient acceptance. For instance, in November 2025, the U.S. Food and Drug Administration announced plans to remove long‑standing boxed warnings from menopausal hormone therapy products to better align labeling with current scientific evidence and contemporary benefit-risk understanding. Building on that effort, in February 2026, the agency approved updated labeling for six such therapies that removes references to certain risk statements, reflecting a more nuanced interpretation of the evidence regarding these treatments’ safety profiles.
Route of Administration Insights
The oral segment accounted for the largest revenue share of 50.54% in 2025, driven by its high patient preference due to ease of administration, cost-effectiveness, and widespread availability across healthcare settings. Oral estrogen therapies remain the most prescribed option for menopause management, supported by strong clinical familiarity and established treatment guidelines. The segment is further benefiting from the growing prevalence of menopausal symptoms, with studies indicating that vasomotor symptoms such as hot flashes and night sweats affect approximately 70-80% of women during menopause, increasing demand for accessible treatment options. Moreover, advancements in oral formulations aimed at improving bioavailability and minimizing systemic side effects are enhancing patient adherence. Pharmaceutical companies are actively investing in next-generation oral hormone therapies with improved safety profiles and combination formulations. For instance, in March 2025, AbbVie Inc. expanded its women’s health portfolio through ongoing development and commercialization efforts focused on hormone therapies, reinforcing the continued relevance and growth of oral administration routes in estrogen replacement therapy.
The transdermal segment is projected to grow at a CAGR of 8.37% over the forecast period, driven by its ability to deliver estrogen directly through the skin while avoiding first-pass liver metabolism, thereby reducing the risk of thromboembolic events and improving safety profiles. Increasing clinical preference for transdermal patches and gels, particularly among women with cardiovascular risk factors, is significantly supporting segment growth. In addition, rising awareness regarding the benefits of steady hormone release and improved tolerability is enhancing patient adherence to transdermal therapies. Continuous innovation in patch technology and gel-based formulations is further expanding treatment options and convenience. Pharmaceutical companies are actively focusing on developing advanced transdermal delivery systems with optimized dosing and enhanced skin permeability. For instance, in February 2026, the U.S. Food and Drug Administration approved labeling updates for multiple menopausal hormone therapy products, reflecting evolving clinical evidence on safety profiles and supporting more informed use of estrogen therapies, including transdermal formulations, which are increasingly preferred due to their differentiated risk profile compared to oral alternatives.
Indication Insights
The menopause management segment dominated the market with the largest revenue share of 60.21% in 2025, driven by the high prevalence of menopausal symptoms such as hot flashes, night sweats, and osteoporosis among aging female populations worldwide. Increasing awareness regarding the long-term health impacts of untreated menopause, including bone density loss and cardiovascular risks, is encouraging early diagnosis and adoption of treatment. Moreover, improved clinical guidelines supporting the timely initiation of hormone therapy are enhancing physician confidence and patient uptake. The segment is further supported by expanding treatment options, including systemic and localized hormone therapies tailored to symptom severity and patient profile. Favorable regulatory developments and growing acceptance of hormone therapy are also accelerating market growth. For instance, in February 2026, the U.S. Food and Drug Administration approved updated labeling for multiple menopausal hormone therapy products, removing outdated risk warnings to better reflect current scientific evidence and improve patient access, thereby reinforcing the role of menopause management therapies in clinical practice.
The osteoporosis prevention/treatment segment is projected to grow at a CAGR of 7.79% over the forecast period, driven by the rising global prevalence of osteoporosis, particularly among postmenopausal women experiencing rapid bone density loss due to declining estrogen levels. Increasing awareness regarding fracture prevention and early screening initiatives is encouraging timely therapeutic intervention, thereby supporting segment growth. Estrogen-based therapies continue to play a crucial role in maintaining bone mineral density and reducing fracture risk, especially when initiated during early menopause. Furthermore, advancements in combination therapies and improved clinical guidelines are enhancing physicians' confidence in prescribing hormone-based treatments for bone health. Growing healthcare investments and emphasis on preventive care are further accelerating adoption. For instance, in May 2024, Pfizer Inc. reintroduced DUAVEE (conjugated estrogens/bazedoxifene) in the U.S. market following manufacturing updates, strengthening its portfolio in menopause and osteoporosis prevention, and highlighting continued industry focus on estrogen-based therapies for bone health management.
Distribution Channel Insights
The hospital pharmacies segment accounted for the largest revenue share of 45.70% in 2025, driven by the high volume of hormone therapy prescriptions initiated and monitored within hospital settings, particularly for patients requiring specialized gynecological and endocrine care. Hospitals serve as primary points for diagnosis, treatment initiation, and management of menopause-related conditions, thereby ensuring consistent demand through in-house pharmacy channels. In addition, the availability of trained healthcare professionals and integrated care pathways enhances patient trust and adherence to prescribed estrogen therapies. Increasing hospitalization rates for age-related conditions and improved access to specialist consultations are further supporting segment dominance. The segment also benefits from strong reimbursement frameworks and institutional procurement systems that favor hospital-based dispensing. For instance, in February 2026, the U.S. Food and Drug Administration approved labeling changes for multiple hormone therapy products, prompting widespread updates across hospital formularies and prescribing practices, thereby reinforcing the critical role of hospital pharmacies in the distribution and administration of estrogen replacement therapies.
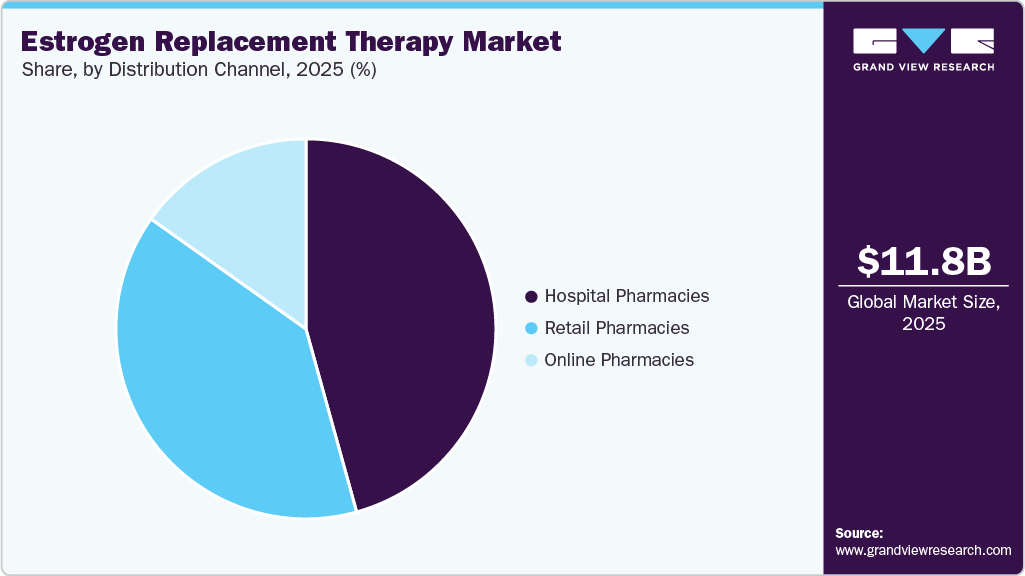
The online pharmacies segment is projected to grow at the fastest CAGR during the forecast period, driven by the increasing adoption of digital healthcare platforms and the growing preference for convenient, home-based access to medications. Rising penetration of telemedicine and e-prescriptions is enabling patients, particularly menopausal women, to access hormone therapies without frequent hospital visits, thereby improving treatment continuity and adherence. Moreover, the demand for discreet and stigma-free access to menopause-related treatments is accelerating the shift toward online pharmacy channels. Integration of digital consultation services with doorstep delivery is further enhancing patient engagement and expanding market reach. The segment is also benefiting from the rapid expansion of direct-to-consumer women’s health platforms. For instance, in January 2024, Evernow expanded its online hormone therapy services across multiple U.S. states, offering personalized prescriptions and home delivery, highlighting the growing role of digital platforms in improving access to menopause treatments.
Regional Insights
North America held the largest share in the market in 2025 due to the high prevalence of menopause-related conditions and a well-established healthcare infrastructure. The region benefits from strong awareness regarding hormone replacement therapies and early diagnosis of menopausal symptoms, driving higher treatment adoption rates. Furthermore, favorable reimbursement policies and widespread access to advanced healthcare services support the consistent use of estrogen therapies. The presence of leading pharmaceutical companies and ongoing research in women’s health further contributes to market growth. Increasing focus on personalized medicine and the availability of multiple therapy options are also strengthening the region’s market position. Moreover, supportive clinical guidelines and endorsements from medical organizations are enhancing physician confidence in prescribing hormone therapies. Rising healthcare expenditure and continuous innovation in drug formulations are further accelerating the adoption of estrogen replacement therapy across the region.
U.S. Estrogen Replacement Therapy Market Trends
The estrogen replacement therapy market in the U.S. is experiencing steady growth, driven by the rising prevalence of menopausal symptoms and increasing awareness regarding women’s health management. High healthcare spending and strong access to advanced treatment options are supporting widespread adoption of hormone replacement therapies across the country. In addition, growing acceptance of personalized and bioidentical hormone therapies is further enhancing patient demand. The presence of well-established pharmaceutical companies and expanding digital health platforms is improving accessibility and treatment adherence. Favorable clinical guidelines and ongoing research in menopause care are also contributing to market expansion. Moreover, increasing investments in women’s health and strategic portfolio expansions by key players are strengthening the availability of innovative hormone therapies across the U.S. For instance, in August 2024, Organon & Co. announced continued expansion of its women’s health portfolio and menopause-focused initiatives, reinforcing its commitment to improving access to hormone therapies and strengthening its position in the U.S. market. This development aligns with the growing demand for advanced and accessible menopause treatments in the country, highlighting how strategic industry initiatives are directly supporting overall market growth.
Europe Estrogen Replacement Therapy Market Trends
The estrogen replacement therapy market in Europe held a significant share, driven by the region’s rapidly aging female population and increasing life expectancy, which extends the duration of menopause-related healthcare needs. Strong government initiatives promoting women’s health screening programs are encouraging early diagnosis and treatment of hormonal imbalances. Furthermore, the widespread presence of universal healthcare systems across several European countries ensures equitable access to hormone therapies. The region is also witnessing a growing preference for low-dose and safer hormone formulations, aligning with evolving clinical practices. Increasing adoption of transdermal and localized therapies is further supporting market growth, particularly among patients seeking alternatives to oral treatments. Moreover, rising focus on preventive healthcare and long-term disease management is driving consistent demand for estrogen therapies. Continuous advancements in pharmaceutical manufacturing standards and regulatory harmonization across the European Union are also strengthening market expansion.
The UK estrogen replacement therapy market is developing as a result of increasing government focus on improving menopause care and addressing gaps in women’s health services. Rising awareness campaigns and national guidelines are encouraging women to seek timely treatment for menopause-related symptoms, thereby supporting therapy adoption. The country is also witnessing growing demand for safer and more accessible hormone replacement options, particularly amid ongoing discussions around HRT availability and supply stability. Expansion of primary care services and pharmacist-led consultations is further improving patient access to estrogen therapies. Moreover, increasing emphasis on workplace policies supporting menopausal women is contributing to higher diagnosis and treatment rates. For instance, in February 2024, the National Health Service expanded menopause support initiatives, including improved access to hormone replacement therapy through primary care channels, highlighting the country’s commitment to strengthening menopause management and driving market growth.
The estrogen replacement therapy market in Germany is witnessing steady growth, driven by the country’s strong emphasis on preventive healthcare and early disease management among aging female populations. Increasing awareness of menopause-related health risks, particularly bone health and cardiovascular conditions, is encouraging the timely adoption of hormone therapies. Germany’s well-established statutory health insurance system ensures broad access to prescription treatments, including hormone replacement therapies, supporting consistent demand. In addition, the country is seeing a shift toward individualized treatment approaches, including low-dose and transdermal estrogen therapies. Growing physician preference for evidence-based prescribing and adherence to updated clinical guidelines are further strengthening market growth. The presence of advanced pharmaceutical manufacturing capabilities and ongoing clinical research in women’s health also contributes to innovation and the availability of treatment options. For instance, in June 2024, Bayer AG reinforced its commitment to women's health through sustained R&D and portfolio management. As a German-headquartered leader, Bayer’s initiatives highlight Germany’s role as a central hub for innovation and patient access within the European market.
France estrogen replacement therapy marketis expanding steadily, driven by the country’s strong reimbursement framework under the national healthcare system, which ensures affordability and access to hormone therapies. The French healthcare model, characterized by specialist-led care, particularly by gynecologists, supports higher diagnosis and treatment rates for menopausal conditions. Increasing emphasis on pharmacovigilance and strict regulatory oversight is enhancing patient confidence in approved hormone therapies. Furthermore, growing medical attention toward managing menopause as a clinical condition rather than a lifestyle phase is encouraging treatment adoption. The market is also benefiting from rising consultations for menopause-related symptoms and structured follow-up care. Furthermore, increasing focus on long-term women’s health outcomes, including bone and cardiovascular health, is sustaining demand for estrogen replacement therapies.
Asia Pacific Estrogen Replacement Therapy Market Trends
The estrogen replacement therapy market in the Asia Pacific is expected to register the fastest CAGR of 8.37% over the forecast period, driven by the large and rapidly aging female population across countries such as Japan, China, and India. Increasing awareness of menopause-related health issues, particularly in urban areas, is encouraging more women to seek medical consultation and hormone-based treatments. The region is also experiencing a gradual shift from traditional remedies toward evidence-based estrogen therapies, supported by improving healthcare infrastructure. Moreover, rising disposable incomes and expanding private healthcare sectors are enhancing access to prescription therapies, including ERT. Variability in regulatory frameworks across countries is influencing the availability and adoption of different estrogen formulations. Growing focus on women’s health initiatives and educational campaigns further supports early diagnosis and treatment uptake. Furthermore, the increasing presence of multinational pharmaceutical companies and localized manufacturing is improving product availability and affordability across the region.
Japan estrogen replacement therapy market is evolving steadily, driven by the country’s rapidly aging female population and increasing focus on extending healthy life expectancy. Unlike Western markets, Japan has historically shown cautious adoption of hormone therapies due to safety concerns following global clinical studies, but recent years have seen gradual acceptance of low-dose estrogen treatments. The market is characterized by a strong preference for physician-supervised therapies, with gynecologists playing a central role in prescribing ERT within hospital settings. In addition, increasing clinical emphasis on managing menopause-related osteoporosis and quality-of-life symptoms is supporting demand. Japan also shows higher adoption of transdermal and localized estrogen therapies, aligning with safety-focused treatment approaches. Furthermore, government initiatives promoting healthy aging are encouraging women to seek medical guidance for menopause management. For instance, in April 2024, Aska Pharmaceutical Holdings Co., Ltd. continued to strengthen its women’s health portfolio by advancing hormone therapy products tailored for the domestic market, reflecting Japan’s focus on safe, localized estrogen therapy solutions.
The estrogen replacement therapy market in Chinais expanding rapidly, driven by the country’s large and aging female population alongside increasing urbanization and healthcare access. Historically, menopause management in China relied more on traditional medicine, but there is a clear shift toward evidence-based estrogen therapies, particularly in Tier 1 and Tier 2 cities. The market is characterized by growing acceptance of hormone therapy among urban populations, supported by improved patient education and physician training. Moreover, government-led healthcare reforms and expansion of public insurance coverage are enhancing access to prescription therapies, including ERT. China is also witnessing rising demand for standardized and regulated hormone products over compounded alternatives, reflecting increasing focus on safety and quality. Preference for hospital-based treatment and specialist consultations continues to influence prescribing patterns in the country. Furthermore, increasing domestic pharmaceutical manufacturing and partnerships with global players are improving the availability and affordability of estrogen therapies. For instance, in March 2024, Bayer AG expanded its women’s health portfolio presence in China through strategic initiatives focused on hormone therapy access, highlighting the country’s growing importance in the market.
Latin America Estrogen Replacement Therapy Market Trends
The estrogen replacement therapy market in Latin America is gaining momentum, driven by improving access to women’s healthcare services and increasing recognition of menopause as a medically managed condition rather than a natural life stage. The region is witnessing a gradual shift toward hormone-based treatments as awareness grows among both patients and healthcare providers. Expanding private healthcare infrastructure, particularly in countries like Brazil and Mexico, is enhancing access to prescription estrogen therapies. Furthermore, rising medical tourism and cross-border healthcare services are contributing to the availability of advanced menopause treatments. The market is also influenced by increasing participation of women in the workforce, driving demand for therapies that improve quality of life and productivity. Government efforts to integrate women’s health into broader public health programs are further supporting diagnosis and treatment rates. Furthermore, the growing presence of regional pharmaceutical manufacturers is improving the affordability and distribution of estrogen replacement therapies across diverse economic segments.
Brazil estrogen replacement therapy marketis developing steadily, driven by the country’s large population of menopausal women and increasing emphasis on women’s health within both public and private healthcare systems. Brazil’s mixed healthcare model, combining the public Unified Health System (SUS) with a strong private sector, is enabling broader access to hormone therapies, particularly in urban centers. The market is characterized by growing awareness of menopause-related conditions such as vasomotor symptoms and bone health, supported by expanding gynecological care services. In addition, cultural shifts toward proactive health management are encouraging women to seek medical treatment rather than relying solely on traditional approaches. Brazil also shows a rising preference for affordable and generic estrogen formulations, reflecting cost sensitivity across segments of the population. The presence of local pharmaceutical manufacturers is further improving accessibility and distribution of ERT products nationwide. For instance, in May 2024, Eurofarma Laboratórios S.A. continued to expand its women’s health portfolio, including hormone therapy offerings, highlighting the growing focus on accessible estrogen treatments in the domestic market.
Middle East & Africa Estrogen Replacement Therapy Market Trends
The estrogen replacement therapy market in MEA is witnessing gradual growth, driven by increasing awareness of menopause-related health issues and rising demand for quality women’s healthcare services across the region. Expanding healthcare infrastructure in countries such as Saudi Arabia, United Arab Emirates, and South Africa is improving access to prescription hormone therapies, particularly in urban centers. The market is supported by growing public and private investment in women’s health programs, including educational campaigns on menopause management. Furthermore, the rising adoption of imported and regulated hormone therapy formulations is enhancing safety and patient confidence. Cultural shifts and increasing acceptance of medicalized menopause care are encouraging more women to seek professional treatment rather than traditional remedies. The region also shows a growing preference for non-oral and transdermal estrogen therapies due to convenience and safety considerations. For instance, in September 2024, Julphar expanded its women’s health portfolio with new hormone therapy products, reflecting the region’s focus on improving access to advanced estrogen replacement therapies.
Saudi Arabia estrogen replacement therapy marketis evolving as awareness of menopause and hormone therapy increases amid shifting cultural perceptions of women’s health. Historically, menopausal hormone therapy (including estrogen replacement) has been underutilized in the Kingdom, with studies showing that a large majority of women have limited knowledge about these therapies and only a small percentage report being prescribed them, indicating a significant opportunity for education and clinical uptake. Physician attitudes also play a key role, as surveys among Saudi clinicians reveal that while many agree HRT should be offered to symptomatic menopausal women, barriers such as limited treatment options and challenges in communicating risks and benefits still affect prescribing practices. Growing public health initiatives aimed at improving women’s health literacy are expected to reduce hesitation and enhance uptake of estrogen therapies in the coming years. Moreover, expanding healthcare access in urban centers supports better diagnosis and treatment initiation for menopausal conditions. Increasing incorporation of evidence‑based hormone therapy into clinical practice guidelines will also contribute to broader adoption of estrogen replacement therapy. These factors together are strengthening the domestic ERT market as Saudi Arabia diversifies its healthcare services and addresses unmet needs in menopause management.
Key Estrogen Replacement Therapy Company Insights
Pfizer Inc., AbbVie Inc., and Bayer AG are actively strengthening the market through continuous innovation in multi-indication formulations that enhance drug stability, solubility, and bioavailability. Companies such as Eli Lilly and Company and Novo Nordisk A/S are expanding their ERT portfolios by investing in advanced delivery technologies and personalized hormone therapy solutions tailored for menopausal and osteoporosis treatments. Novartis AG and Teva Pharmaceutical Industries Ltd. are focusing on oral, transdermal, and controlled-release estrogen replacement therapy options, supporting the development of next-generation dosage forms with improved patient adherence. Meanwhile, Merck & Co., Inc., leverages its expertise in specialty chemistries to enhance formulation consistency, bioavailability, and long-term efficacy of estrogen therapies. F. Hoffmann-La Roche Ltd. continues to expand its plant-based and sustainable excipient offerings, addressing the rising demand for natural components in women’s health products, while Viatris Inc. plays a critical role in modified-release and film-coated estrogen therapies that improve treatment convenience and patient compliance. Collectively, these companies are driving advancements in formulation innovation, scalability, and regulatory alignment, supporting the evolving needs of modern estrogen replacement therapy development.
Key Estrogen Replacement Therapy Companies:
The following key companies have been profiled for this study on the estrogen replacement therapy market.
- Pfizer Inc.
- AbbVie Inc.
- Bayer AG
- Eli Lilly and Company
- Novo Nordisk A/S
- Novartis AG
- Merck & Co., Inc.
- F. Hoffmann-La Roche Ltd.
- Viatris Inc.
- Teva Pharmaceutical Industries Ltd.
Recent Developments
-
In February 2026, Dr. Reddy’s Laboratories acquired the India trademarks and related assets of estrogen‑based HRT brands Progynova and Cyclo‑Progynova from UK‑based Mercury Pharma for about USD 32 million, marking its strategic entry and expansion into the hormone replacement therapy segment to strengthen its women’s health portfolio.
-
In December 2025, Viatris Inc. reported regulatory progress on several programs, notably a planned FDA 505(b)(2) NDA submission in 2026 for a low‑dose estrogen weekly patch. This investigational therapy aims to expand treatment choices beyond daily estrogen dosing, demonstrating the company’s effort to broaden women’s health offerings.
-
In February 2025, The HRT Club nationwide launch expanded direct access to hormone replacement therapies by partnering with a network of over 700 providers and establishing agreements with drug manufacturers and wholesalers, aiming to reduce costs and improve availability of estrogen and related therapies across the U.S. without traditional insurance barriers.
Estrogen Replacement Therapy Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 12.37 billion
Revenue forecast in 2033
USD 18.36 billion
Growth rate
CAGR of 5.80% from 2026 to 2033
Historical data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Therapy type, route of administration, indication, distribution channel, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key company profiled
Pfizer Inc.; AbbVie Inc.; Bayer AG; Eli Lilly and Company; Novo Nordisk A/S; Novartis AG; Merck & Co., Inc.; F. Hoffmann-La Roche Ltd.; Viatris Inc.; Teva Pharmaceutical Industries Ltd.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Estrogen Replacement Therapy Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global estrogen replacement therapy market report based on therapy type, route of administration, indication, distribution channel, and region:
-
Therapy Type Outlook (Revenue, USD Billion, 2021 - 2033)
-
Estrogen-only therapy (ET)
-
Combined estrogen + progestin therapy (EPT)
-
Bioidentical hormone therapy
-
-
Route of Administration Outlook (Revenue, USD Billion, 2021 - 2033)
-
Oral
-
Transdermal
-
Vaginal
-
Parenteral
-
-
Indication Outlook (Revenue, USD Billion, 2021 - 2033)
-
Menopause management
-
Osteoporosis prevention/treatment
-
Hypogonadism
-
Others
-
-
Distribution Channel Outlook (Revenue, USD Billion, 2021 - 2033)
-
Hospital pharmacies
-
Retail pharmacies
-
Online pharmacies
-
-
Regional Outlook (Revenue, USD Billion, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
The global estrogen replacement therapy market size was valued at USD 11.75 billion in 2025 and is anticipated to reach USD 12.37 billion in 2026.
The global estrogen replacement therapy market is expected to witness a compound annual growth rate of 5.8% from 2026 to 2033 to reach USD 18.36 billion by 2033.
Based on therapy type, the Combined estrogen + progestin therapy (EPT) segment dominated the market with the largest revenue share of 47.43% in 2025, driven by its proven effectiveness in managing menopausal symptoms while reducing the risk of endometrial hyperplasia in women with an intact uterus..
Some of the key players in estrogen replacement therapy market are Pfizer Inc., AbbVie Inc., Bayer AG, Eli Lilly and Company, and Novo Nordisk A/S
The estrogen replacement therapy market is driven by the rising global population of menopausal and postmenopausal women..
About the authors:
Author: GVR Pharmaceuticals Research Team | Last Updated:
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









