- Home
- »
- Pharmaceuticals
- »
-
Insulin Aspart Market Size, Share, Industry Report, 2033GVR Report cover
![Insulin Aspart Market Size, Share & Trends Report]()
Insulin Aspart Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Branded, Biosimilar), By Application (Type 1 Diabetes, Type 2 Diabetes), By Distribution Channel (Hospital Pharmacies, Online Pharmacies), By Region, And Segment Forecasts
- Report ID: GVR-4-68040-907-6
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Insulin Aspart Market Summary
The global insulin aspart market size was estimated at USD 3,520.00 million in 2025 and is projected to reach USD 5,019.85 million by 2033, growing at a CAGR of 4.60% from 2026 to 2033. The market is experiencing steady growth driven by the increasing global burden of Diabetes Mellitus, particularly in emerging economies where lifestyle changes and urbanization are accelerating disease prevalence.
Key Market Trends & Insights
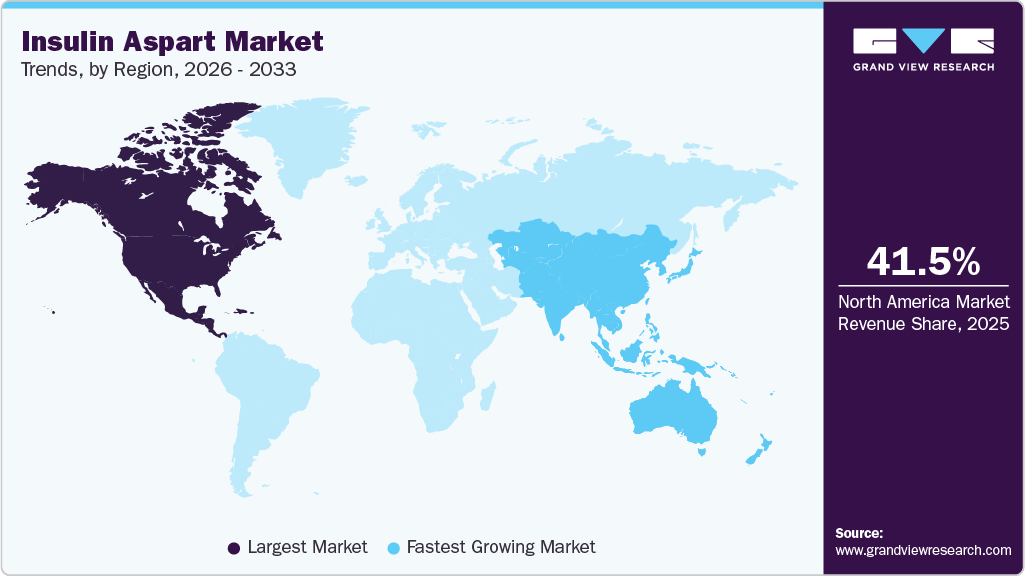
- North America insulin aspart market held the largest share of 41.50% of the global market in 2025.
- The insulin aspart industry in the U.S. is expected to grow significantly over the forecast period.
- By product, the branded segment held the largest market share of 73.42% in 2025.
- By application, the type 2 diabetes segment held the largest market share of 51.87% in 2025.
- By distribution channel, the hospital pharmacies segment held the largest market share of 47.01% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 3,520.00 Million
- 2033 Projected Market Size: USD 5,019.85 Million
- CAGR (2026-2033): 4.60%
- North America: Largest market in 2025
- Asia Pacific: Fastest-growing market
Rising incidences of obesity, sedentary habits, and aging populations are significantly contributing to higher insulin dependency rates. In addition, improved screening programs and early diagnosis initiatives are expanding the patient pool requiring insulin therapy. Government-led awareness campaigns and healthcare reforms are further supporting the adoption of treatment. The growing emphasis on effective glycemic control to prevent long-term complications is also strengthening demand. As a result, rapid-acting insulin analogs such as Insulin Aspart are increasingly preferred therapeutic options.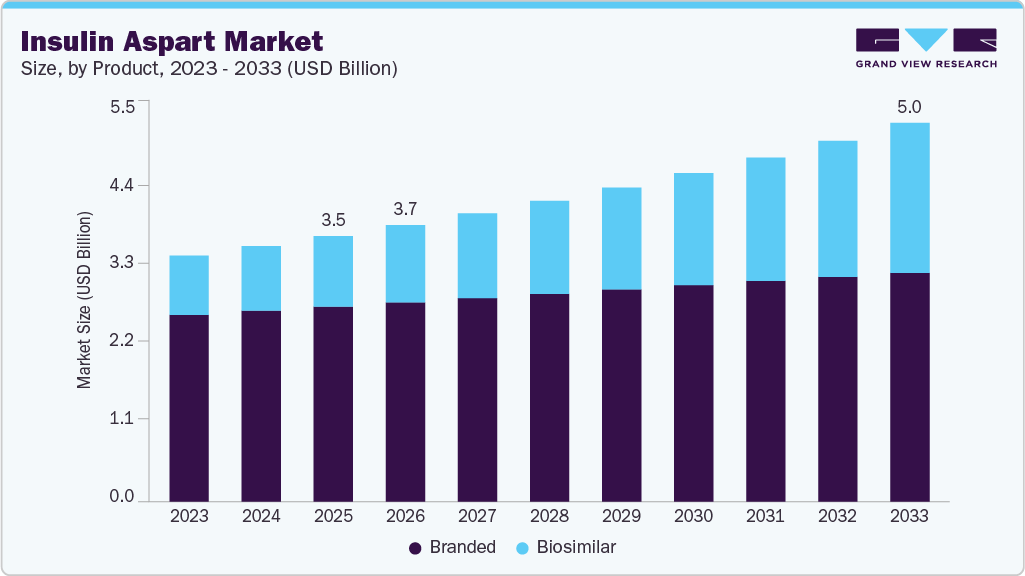
The continuous advancement in insulin delivery technologies, including insulin pens, pumps, and smart delivery systems, is further fueling the growth of the market. These innovations enhance patient convenience, dosing accuracy, and adherence to therapy, thereby improving clinical outcomes. The integration of digital health tools, such as continuous glucose monitoring (CGM) systems and connected insulin devices, is further supporting real-time diabetes management. Pharmaceutical companies are also focusing on developing biosimilars and cost-effective formulations to expand accessibility across price-sensitive markets. Increasing healthcare expenditure and insurance coverage for diabetes care are enabling broader access to advanced insulin therapies. Collectively, these factors are fostering strong market penetration of insulin aspart-based treatments.
Furthermore, the market is benefiting from favorable regulatory approvals and expanding product applications across different patient populations. Strategic collaborations, partnerships, and product launches by key industry players are enhancing global distribution networks and strengthening market presence. There is also a growing shift toward personalized medicine, where tailored insulin regimens improve patient-specific outcomes. Rising demand in hospital and homecare settings is further boosting product utilization. In addition, increased investments in research and development are driving formulation improvements and the development of next-generation insulin products. These combined dynamics are expected to sustain long-term growth momentum for the insulin aspart market worldwide.
Market Concentration & Characteristics
The insulin aspart market demonstrates a moderate to high degree of innovation, driven by continuous advancements in formulation technologies and delivery systems for Insulin Aspart. Innovations such as smart insulin pens, integration with continuous glucose monitoring systems, and improved pharmacokinetic profiles are enhancing treatment precision for Diabetes Mellitus. Besides, the development of biosimilars and next-generation ultra-fast insulin variants is increasing competition and accessibility. Ongoing R&D investments and digital health integration continue to push the market toward more patient-centric and technologically advanced solutions.
The industry presents high barriers to entry due to the complex manufacturing processes and stringent regulatory requirements associated with biologics like Insulin Aspart. Significant capital investment is required for advanced production facilities, clinical trials, and compliance with global quality standards. Established players benefit from strong brand recognition, extensive distribution networks, and long-standing relationships with healthcare providers treating Diabetes Mellitus. Additionally, intellectual property protections and the technical challenges of developing biosimilars further limit new entrants in the market.
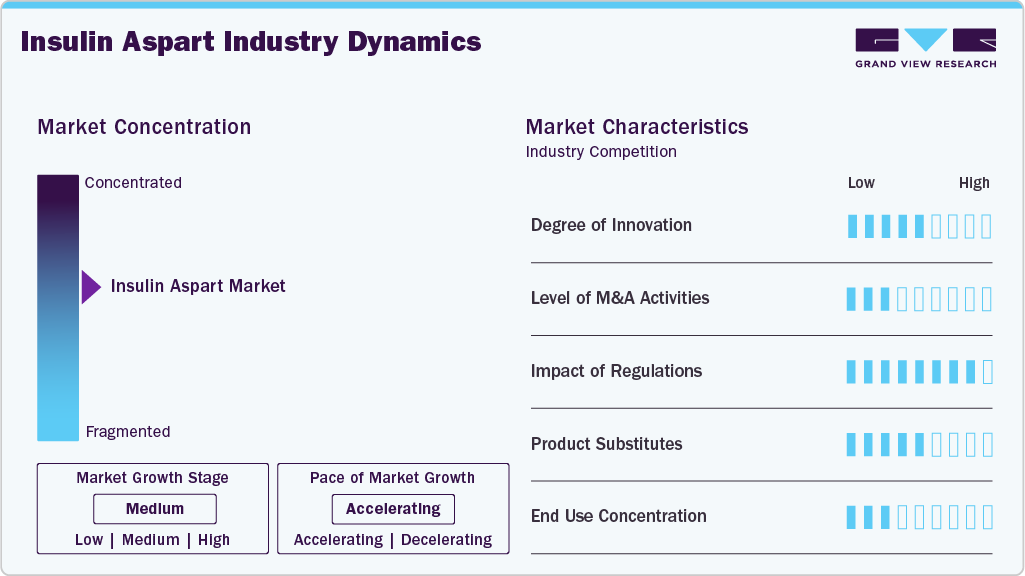
The market is significantly influenced by stringent regulatory frameworks that govern the approval, manufacturing, and distribution of biologics such as Insulin Aspart. Regulatory bodies enforce rigorous clinical evaluation, pharmacovigilance, and quality compliance standards, which increase development timelines but ensure high safety and efficacy for patients with Diabetes Mellitus. Additionally, evolving biosimilar approval pathways and pricing regulations are encouraging competitive market entry while improving affordability and access. Government-led reimbursement policies and the inclusion of insulin therapies on essential medicine lists are further driving market expansion by enhancing patient access.
The insulin aspart industry faces moderate competition from product substitutes, including other rapid-acting insulin analogs and non-insulin therapies used in the management of Diabetes Mellitus. Alternatives such as Insulin Lispro and Insulin Glulisine offer similar efficacy, while newer drug classes like GLP-1 receptor agonists are gaining traction in type 2 diabetes care. Despite this, Insulin Aspart maintains strong demand due to its rapid onset, flexible dosing, and widespread clinical acceptance. Continuous innovation and physician familiarity further help sustain its competitive positioning in the presence of substitutes.
The market is witnessing strong geographical expansion, particularly across emerging regions where the prevalence of Diabetes Mellitus is rising rapidly. Growing healthcare infrastructure, increasing urbanization, and improving access to diagnosis and treatment are driving demand in Asia-Pacific, Latin America, and the Middle East. Multinational companies are expanding their footprint through strategic partnerships, local manufacturing, and distribution agreements to enhance the availability of Insulin Aspart. Additionally, supportive government initiatives, rising healthcare spending, and broader insurance coverage are further accelerating market penetration in underserved and rural areas.
Product Insights
The branded segment dominated the market with the largest revenue share of 73.42% in 2025, primarily supported by strong physician preference for well-established rapid-acting insulin analogs such as NovoLog and NovoRapid. These products are widely regarded as clinical benchmarks due to their proven efficacy in postprandial glucose control, predictable pharmacokinetic profiles, and established long-term safety data across both Type 1 and Type 2 diabetes populations. The segment continues to benefit from high trust among endocrinologists and healthcare providers, particularly in hospital and specialty care settings where precise glycemic management is critical. Additionally, branded insulin aspart products are increasingly integrated with advanced delivery technologies, including prefilled insulin pens and connected smart injection systems, which enhance dosing accuracy, treatment adherence, and patient convenience. The sustained rise in global Diabetes Mellitus prevalence, coupled with increasing adoption of intensive insulin therapy regimens, continues to reinforce demand for branded insulin aspart therapies. Furthermore, ongoing innovations in digital diabetes management and insulin delivery devices further strengthen the competitive position of branded products as the standard of care in rapid-acting insulin treatment worldwide.
The biosimilar segment is projected to grow at a CAGR of 9.94% over the forecast period, driven by increasing demand for cost-effective insulin therapies and growing acceptance of biosimilars across healthcare systems. These products provide comparable efficacy, safety, and pharmacokinetic profiles to reference biologics, making them highly suitable for large patient populations and government-supported diabetes programs. The rising prevalence of Diabetes Mellitus, along with increasing healthcare cost pressures, is accelerating the shift toward biosimilar adoption globally. Regulatory authorities are also actively supporting biosimilars through streamlined approval pathways and interchangeability designations, further strengthening market penetration. For instance, in February 2025, the U.S. FDA approved Merilog (insulin-aspart-szjj), the first rapid-acting insulin aspart biosimilar referencing NovoLog, marking a significant milestone in expanding affordable access to prandial insulin therapy. Similarly, in July 2025, Biocon Biologics received approval from the U.S. Food and Drug Administration for Kirsty (insulin aspart-xjhz), the first interchangeable rapid-acting insulin aspart biosimilar to NovoLog in the U.S., significantly enhancing treatment accessibility and affordability. This milestone underscores the expanding commercialization and strong future potential of biosimilar insulin aspart products worldwide.
Application Insights
The type 2 diabetes segment dominated the market with the largest revenue share of 51.87% in 2025, driven by the rapidly increasing global prevalence of Type 2 Diabetes Mellitus, which accounts for nearly 90-95% of all diabetes cases as of 2025. Rising obesity, sedentary lifestyles, and aging populations continue to accelerate disease burden, thereby increasing reliance on rapid-acting insulin analogs such as insulin aspart for effective postprandial glucose control. Insulin therapy is widely adopted in advanced or poorly controlled cases, reinforcing strong, sustained demand in this segment. Additionally, healthcare systems are increasingly focusing on expanding access to advanced diabetes therapies through strategic partnerships and commercialization initiatives. For instance, in February 2026, Abbott partnered with Novo Nordisk in India to commercialize a semaglutide-based therapy for type 2 diabetes, aiming to expand access to evidence-based treatment through combined distribution and scientific expertise. This development highlights the growing emphasis on collaborative efforts to improve treatment accessibility and comprehensive diabetes management, indirectly supporting the broader insulin therapy ecosystem, including insulin aspart.
The type 1 diabetes segment is projected to grow at a significant CAGR during the forecast period. The segment represents a critical application area in the insulin aspart market, driven by the absolute dependence on insulin therapy for individuals with Type 1 Diabetes Mellitus, where the body produces little to no insulin due to autoimmune destruction of pancreatic beta cells. This condition accounts for approximately 5-10% of all diabetes cases globally as of 2026, with patients requiring lifelong insulin administration for survival and glycemic control. Rapid-acting insulin analogs, such as insulin aspart, are essential in intensive insulin regimens, particularly for mealtime glucose management and for use in insulin pumps. The increasing adoption of advanced insulin delivery systems and pediatric diabetes care programs is further strengthening demand in this segment. For instance, in March 2026, the Karnataka government in India announced the provision of free insulin pens for children with type 1 diabetes to ensure continuous access to insulin therapy and reduce financial burden on families. This development highlights the growing focus on improving accessibility and long-term management of type 1 diabetes, directly supporting the demand for insulin aspart therapies.
Distribution Channel Insights
The hospital pharmacies segment dominated the market with the largest revenue share of 47.01% in 2025. The hospital pharmacies segment plays a vital role in the insulin aspart market, driven by the high volume of inpatient diabetes management, emergency care, and specialized endocrinology services delivered within hospital settings. These pharmacies ensure the availability of rapid-acting insulin, such as insulin aspart, for critically ill patients, perioperative care, and those requiring intensive insulin therapies under medical supervision. Hospitals also support the initiation and titration of insulin regimens, making them a key distribution channel for advanced insulin analogs. The growing burden of diabetes and the increasing number of hospital admissions related to its complications further strengthen demand in this segment. Furthermore, hospital pharmacies maintain strict cold-chain logistics and clinical oversight to ensure safe and effective insulin delivery. For instance, in October 2025, the state of California launched the CalRx program in collaboration with Civica Rx to supply low-cost insulin through healthcare systems and pharmacy networks, including hospitals, improving affordability and access to essential insulin therapies. This development highlights the expanding role of institutional distribution channels, including hospital pharmacies, in ensuring consistent insulin availability and cost-effective diabetes care.
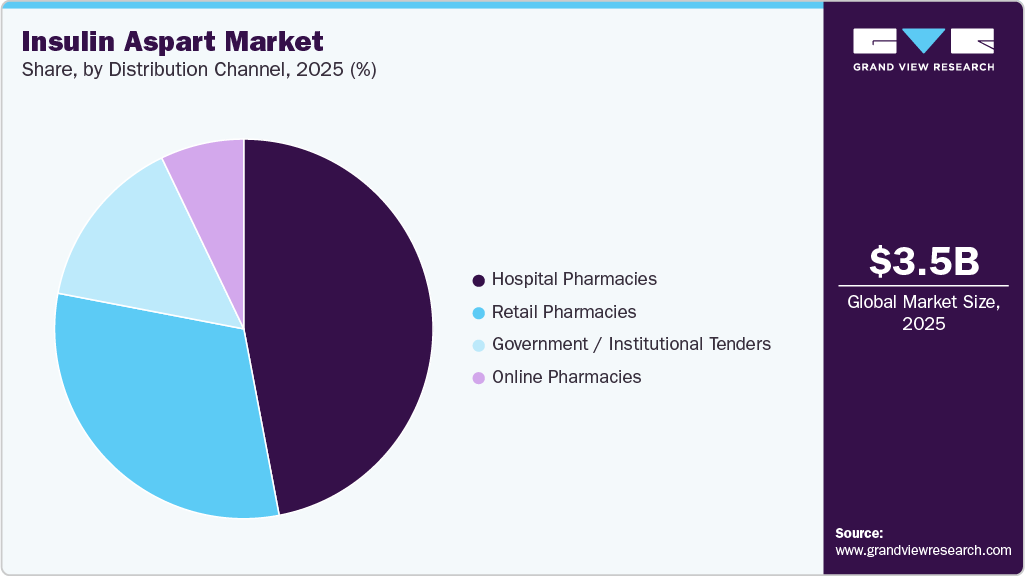
The online pharmacies segment is projected to grow at a CAGR of 14.63% over the forecast period. The segment is gaining significant traction in the insulin aspart market, driven by the increasing digitalization of healthcare and growing patient preference for convenient access to chronic medications. Online pharmacies enable patients with Diabetes Mellitus to order insulin aspart through e-prescriptions with doorstep delivery, improving adherence and continuity of care. The expansion of telehealth services and direct-to-consumer pharmaceutical platforms is further accelerating the adoption of online pharmacy channels. These platforms also enhance pricing transparency and accessibility, particularly for long-term insulin users managing chronic conditions. For instance, in September 2025, AstraZeneca launched “AstraZeneca Direct,” an online platform enabling patients to access prescribed medications for chronic diseases, including diabetes, with home delivery and transparent pricing. This development highlights the growing role of digital pharmacy ecosystems in expanding access to essential therapies and supporting the distribution of insulin products such as insulin aspart.
Regional Insights
The North America insulin aspart industry dominated with a share of 41.50% in 2025, driven by the high prevalence of Diabetes Mellitus, particularly in the United States and Canada, supported by widespread screening and early diagnosis rates. Advanced healthcare infrastructure and strong access to endocrinology care enable consistent adoption of intensive insulin therapies, including Insulin Aspart. Favorable reimbursement frameworks and insurance coverage for insulin analogs further enhance patient access and adherence. The region also benefits from high adoption of advanced delivery devices such as insulin pumps and smart pens integrated with digital monitoring systems. Additionally, the strong presence of leading pharmaceutical companies and continuous investment in diabetes management technologies are accelerating product uptake. Increasing patient awareness regarding glycemic control and prevention of complications further supports sustained market growth in North America.
U.S. Insulin Aspart Market Trends
The insulin aspart industry in the U.S. is driven by the high and growing burden of Diabetes Mellitus, with over 40 million people living with the condition (diagnosed and undiagnosed) and a rapidly expanding prediabetic population that continues to widen the treatment base. Strong healthcare infrastructure, widespread insurance coverage, and high adoption of intensive insulin regimens are accelerating the use of Insulin Aspart across clinical settings. The market is also supported by increasing uptake of biosimilars and interchangeable insulin products, improving affordability and accessibility. For instance, in July 2025, Biocon Biologics received FDA approval for Kirsty, an interchangeable insulin aspart, further strengthening product availability and innovation in the segment. The market is further propelled by the increasing adoption of digital diabetes management solutions, including insulin pumps and continuous glucose monitoring systems, which enhance treatment adherence and support optimized glycemic control.
Europe Insulin Aspart Market Trends
The insulin aspart industry in Europe is driven by the rising prevalence of Diabetes Mellitus, particularly across countries such as Germany, the UK, and France, supported by aging populations and lifestyle-related risk factors. Well-established public healthcare systems and favorable reimbursement policies enable broad patient access to advanced insulin therapies, including Insulin Aspart. The region is also witnessing increased adoption of biosimilar insulin products, contributing to cost containment and improved treatment accessibility. Growing emphasis on early diagnosis and disease management programs is further strengthening demand for rapid-acting insulin analogs. Additionally, integration of digital health technologies such as continuous glucose monitoring and insulin delivery devices is enhancing treatment outcomes. Strong regulatory support and ongoing innovation in diabetes care continue to sustain steady market growth across Europe.
The UK insulin aspart industry is driven by the increasing prevalence of Diabetes Mellitus, supported by a well-established public healthcare system under the NHS that ensures broad access to insulin therapies. A strong government focus on cost containment has accelerated the adoption of biosimilar insulin products, improving the affordability of Insulin Aspart. The market also benefits from structured diabetes management programs, early diagnosis initiatives, and widespread use of advanced delivery devices such as insulin pens and pumps. Additionally, growing emphasis on switching from originator biologics to biosimilars is enhancing market competition and accessibility. For instance, in July 2023, insights published in The Pharmaceutical Journal highlighted the increasing importance of biosimilar insulin adoption in the UK, driven by healthcare system cost pressures and the need to expand patient access. Furthermore, continued regulatory support and integration of digital diabetes care solutions are strengthening long-term market growth across the UK.
The insulin aspart industry in Germany maintains a strong position within Europe. The market is driven by the high prevalence of Diabetes Mellitus and a well-structured statutory health insurance system that ensures broad patient access to advanced therapies. The country’s strong focus on cost-efficiency has accelerated the adoption of biosimilar insulins, enhancing the uptake of Insulin Aspart across both hospital and outpatient settings. Germany is also one of the leading European markets for biosimilar penetration, supported by physician acceptance and favorable substitution policies. Additionally, increasing integration of digital diabetes management tools and insulin delivery technologies is improving treatment adherence and outcomes. For instance, in March 2025, a study published in Diabetes Technology & Therapeutics highlighted the growing clinical adoption of insulin aspart biosimilar SAR341402 across Europe, including Germany, demonstrating comparable efficacy and safety to originator products and supporting wider market penetration. Furthermore, the continuous expansion of biosimilar pipelines and pricing pressure from healthcare payers are fostering competition and driving sustained growth in the industry.
The France insulin aspart industry is driven by the rising burden of Diabetes Mellitus and a strong public healthcare system that ensures widespread access to advanced insulin therapies. The country’s focus on cost containment and efficiency has accelerated the adoption of biosimilar products, significantly boosting the uptake of Insulin Aspart. National and regional health authorities are actively promoting biosimilar prescribing through policy measures and physician incentives, enhancing market penetration. Additionally, increasing integration of digital diabetes management tools and structured care pathways is improving treatment adherence and outcomes. Furthermore, the ongoing expansion of biosimilar pipelines and strong regulatory support continue to drive innovation and accessibility in the market.
Asia Pacific Insulin Aspart Market Trends
The insulin aspart industry in the Asia Pacific is expected to register the fastest CAGR of 6.94% over the forecast period. The market is driven by the rapidly increasing prevalence of Diabetes Mellitus, supported by large patient populations in countries such as China and India. Rising urbanization, changing dietary patterns, and sedentary lifestyles are significantly contributing to the growing diabetes burden across the region. Expanding healthcare infrastructure and improving access to insulin therapies are further supporting market growth, particularly in emerging economies. Additionally, increasing adoption of biosimilar insulin aspart and government initiatives to enhance affordability are accelerating market penetration. The integration of digital health tools and growing awareness regarding early diagnosis and effective disease management are also improving treatment adherence. Furthermore, the strong presence of regional and global pharmaceutical players, along with ongoing product accessibility initiatives, continues to drive the growth.
The Japan insulin aspart industry is driven by the rising prevalence of Diabetes Mellitus, particularly among the country’s rapidly aging population, where the majority of individuals are aged 65 and above, increasing the need for long-term insulin therapy. The high burden of type 2 diabetes and unique disease characteristics in Japanese patients, such as lower insulin secretion capacity, further support the demand for rapid-acting insulin analogs like insulin aspart for effective postprandial glucose control. Japan’s universal healthcare system ensures broad reimbursement coverage for insulin therapies and delivery devices, significantly improving patient access and adherence. Additionally, strong adoption of advanced insulin delivery formats such as prefilled pens and connected devices enhances treatment convenience and precision. The presence of well-established pharmaceutical companies and continuous focus on patient-centric diabetes management further contribute to market growth. Furthermore, increasing awareness, early diagnosis, and structured disease management programs continue to support the expansion of the market in Japan.
The insulin aspart industry in China is driven by the rapidly increasing prevalence of diabetes mellitus, supported by one of the largest diabetic populations globally and a rapidly aging demographic. Urbanization, changing dietary habits, and sedentary lifestyles are significantly contributing to disease burden, thereby increasing the demand for rapid-acting insulin analogs such as insulin aspart for effective postprandial glucose control. Government-led healthcare reforms, particularly centralized procurement programs, are improving affordability and accelerating the adoption of insulin analogs and biosimilars across the country. Moreover, the strong presence of domestic manufacturers and increasing focus on local production are enhancing supply capabilities and reducing reliance on imports. The expansion of hospital infrastructure and broader insurance coverage are further supporting patient access to advanced diabetes therapies. Overall, sustained policy support and rising treatment demand continue to drive the growth.
Latin America Insulin Aspart Market Trends
The insulin aspart industry in Latin America is driven by the rising prevalence of diabetes mellitus, particularly in countries such as Brazil, Mexico, and Argentina. Increasing urbanization, unhealthy dietary patterns, and growing obesity rates are significantly contributing to disease burden, thereby driving demand for rapid-acting insulin analogs such as insulin aspart. Public healthcare systems across the region are actively expanding access to insulin therapies through subsidy programs and partnerships with pharmaceutical companies, improving affordability and treatment uptake. Additionally, the growing adoption of biosimilar insulin products is enhancing cost efficiency and broadening patient access to advanced therapies. The expansion of retail and specialty pharmacy networks, along with improving cold-chain infrastructure, is further strengthening insulin distribution across the region. Furthermore, increasing awareness initiatives and advancements in diabetes management technologies are supporting better diagnosis, adherence, and long-term disease control, thereby driving the growth of the market in Latin America.
The Brazil insulin aspart industry is influenced by the country’s large-scale public health infrastructure, where diabetes treatment is primarily delivered through the Unified Health System (SUS), ensuring broad population coverage. The government’s strong role in medicine distribution, including centralized insulin procurement and free or subsidized access programs, significantly shapes market dynamics and pricing structures. Unlike more privatized markets, Brazil emphasizes volume-driven supply and cost optimization, which has accelerated the inclusion of biosimilar and analog insulin products in public formularies. At the same time, regional disparities in healthcare access between urban and rural areas continue to affect consistent treatment delivery. The growing presence of domestic pharmaceutical manufacturing and technology transfer initiatives is strengthening supply continuity and reducing reliance on imports. For instance, in July 2025, Brazil resumed domestic insulin production after more than 20 years through a government-led technology transfer program, aiming to supply a significant share of national demand and enhance self-sufficiency in insulin availability. This development highlights the country’s strategic shift toward local production and long-term sustainability of insulin supply, directly supporting the market.
Middle East & Africa Insulin Aspart Market Trends
The insulin aspart industry in the Middle East & Africa is driven by the rising prevalence of Diabetes Mellitus, particularly in countries such as Saudi Arabia, the United Arab Emirates, and South Africa. High rates of obesity, sedentary lifestyles, and genetic predisposition are significantly contributing to the increasing diabetes burden across the region, thereby driving demand for rapid-acting insulin analogs such as insulin aspart. Governments in the Gulf Cooperation Council (GCC) countries are heavily investing in healthcare infrastructure and expanding access to advanced diabetes treatments, while public-private partnerships are improving insulin availability in parts of Africa. Additionally, the growing adoption of biosimilar insulin products is enhancing affordability and supporting wider patient access in cost-sensitive markets. However, disparities in healthcare infrastructure and limited access in rural and low-income regions continue to pose challenges to market growth. Furthermore, increasing awareness programs, screening initiatives, and gradual improvements in healthcare systems are supporting better diagnosis and long-term diabetes management, driving the growth of the market in the Middle East & Africa.
The Saudi Arabia insulin aspart industry is characterized by a strong institutional procurement framework and high reliance on government-led healthcare delivery, where the majority of diabetes treatment is managed through public hospitals and specialized care centers. The country’s centralized purchasing system enables bulk acquisition of insulin therapies, improving pricing efficiency and ensuring consistent availability of rapid-acting insulin analogs such as insulin aspart. A notable shift toward preventive care and early intervention programs is reshaping diabetes management, with increased screening initiatives identifying patients at earlier stages of the disease. Additionally, Saudi Arabia’s high adoption of advanced medical technologies, including insulin pens and digital monitoring systems, reflects a preference for precision-based treatment approaches. The expansion of health insurance coverage for expatriates and private sector employees is further diversifying access channels beyond public institutions. Moreover, the country’s focus on localizing pharmaceutical production and reducing import dependence is gradually strengthening domestic supply capabilities for insulin products.
Key Insulin Aspart Company Insights
Novo Nordisk and Sanofi continue to strengthen their presence in the insulin aspart market through advanced insulin analog formulations and innovative delivery devices such as prefilled pens and connected insulin systems. Biocon and Tonghua Dongbao Pharmaceutical Co., Ltd. are expanding their presence with biosimilar insulin aspart offerings, supported by increasing regulatory approvals and efforts to improve affordability across emerging markets. Gan & Lee Pharmaceuticals and Julphar (Gulf Pharmaceutical Industries) are focusing on scaling production capacities and enhancing regional distribution networks to strengthen insulin accessibility. United Laboratories International Holdings Limited and Geropharm are advancing their portfolios through formulation improvements and localized manufacturing strategies. Wockhardt and Sedico Pharmaceutical Company are contributing by developing cost-effective insulin products and expanding their footprint in price-sensitive markets. The market is characterized by intensifying competition driven by biosimilar adoption, pricing pressures, technological advancements in insulin delivery, and increasing emphasis on improving patient access to rapid-acting insulin therapies.
Key Insulin Aspart Companies:
The following key companies have been profiled for this study on the insulin aspart market.
- Novo Nordisk
- Sanofi
- Biocon
- Wockhardt
- Tonghua Dongbao Pharmaceutical Co., Ltd.
- Gan & Lee Pharmaceuticals
- Julphar (Gulf Pharmaceutical Industries)
- Geropharm
- United Laboratories International Holdings Limited
- Sedico Pharmaceutical Company
Recent Developments
-
In April 2025, Julphar officially launched its locally manufactured full portfolio of insulin analogues, which includes insulin aspart (rapid-acting insulin) as part of its diabetes care expansion strategy in the MENA region. This initiative strengthens local production capabilities and improves access to affordable insulin therapies across regional markets.
-
In March 2025, Biocon Biologics entered a strategic collaboration with Civica to expand the availability of insulin aspart in the United States, focusing on improving affordability and addressing recurring insulin shortages through large-scale, not-for-profit manufacturing and distribution channels.
-
In February 2025, Sanofi received U.S. FDA approval for MERILOG (insulin-aspart-szjj), the first rapid-acting insulin aspart biosimilar referencing NovoLog. This milestone increases competition in the insulin aspart market, enhances affordability, and improves patient access, strengthening biosimilar adoption and intensifying competitive dynamics in the global rapid-acting insulin segment.
Insulin Aspart Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 3,664.11 million
Revenue forecast in 2033
USD 5,019.85 million
Growth rate
CAGR of 4.60% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Product, application, distribution channel, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key company profiled
Novo Nordisk; Sanofi; Biocon; Wockhardt; Tonghua Dongbao Pharmaceutical Co., Ltd.; Gan & Lee Pharmaceuticals; Julphar (Gulf Pharmaceutical Industries); Geropharm; United Laboratories International Holdings Limited;Sedico Pharmaceutical Company.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Insulin Aspart Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global insulin aspart market report based on product, application, distribution channel, and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Branded
-
Biosimilar
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Type 1 Diabetes
-
Type 2 Diabetes
-
Gestational Diabetes
-
-
Distribution Channel Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospital Pharmacies
-
Retail Pharmacies
-
Online Pharmacies
-
Government / Institutional Tenders
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. Based on product, the branded segment dominated the market with the largest revenue share of 73.42% in 2025, primarily supported by strong physician preference for well-established rapid-acting insulin analogs such as NovoLog and NovoRapid. .
b. The Insulin Aspart market is experiencing steady growth driven by the increasing global burden of Diabetes Mellitus, particularly in emerging economies where lifestyle changes and urbanization are accelerating disease prevalence.
b. The global Insulin Aspart market size was valued at USD 3,520.00 million in 2025 and is anticipated to reach USD 3,664.11 million in 2026.
b. The global Insulin Aspart market is expected to witness a compound annual growth rate of 4.6% from 2026 to 2033 to reach USD 5,019.85 million by 2033.
b. Some of the key players in Insulin Aspart market are Novo Nordisk; Sanofi; Biocon; Wockhardt; Tonghua Dongbao Pharmaceutical Co., Ltd.; Gan & Lee Pharmaceuticals; Julphar (Gulf Pharmaceutical Industries); Geropharm; United Laboratories International Holdings Limited;Sedico Pharmaceutical Company.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










