- Home
- »
- Medical Devices
- »
-
Nonclinical Pharmacology and Toxicology Regulatory Consulting Outsourcing Market Report, 2033GVR Report cover
![Nonclinical Pharmacology And Toxicology Regulatory Consulting Outsourcing Market Size, Share & Trends Report]()
Nonclinical Pharmacology And Toxicology Regulatory Consulting Outsourcing Market (2026 - 2033) Size, Share & Trends Analysis Report By Service (Regulatory Strategy & Nonclinical Development Planning, IND/CTA-Enabling Nonclinical Program Design), By Application, By Modality, By Therapeutic Area, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-891-7
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Summary
The global nonclinical pharmacology and toxicology regulatory consulting outsourcing market size was estimated at USD 0.965 billion in 2025 and is projected to reach USD 2.39 billion by 2033, growing at a CAGR of 12.2% from 2026 to 2033. The market growth is driven by the growing global drug development landscape, increasing drug discovery and development activities by pharmaceutical & biotechnology companies.
Key Market Trends & Insights
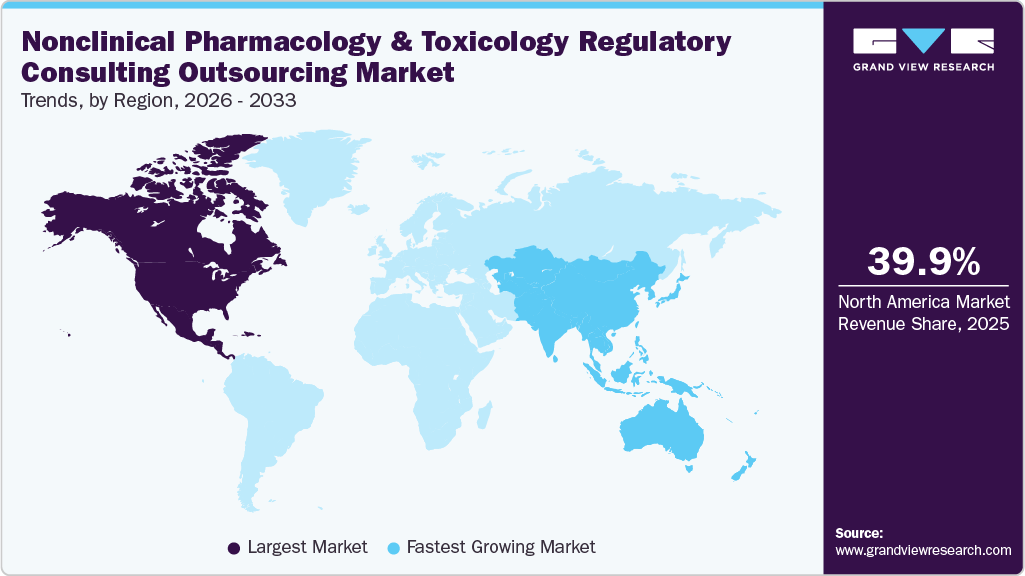
- The North America nonclinical pharmacology and toxicology regulatory consulting outsourcing market held the largest share of 39.9% of the global market in 2025.
- The nonclinical pharmacology and toxicology regulatory consulting outsourcing market in the U.S. is expected to grow significantly over the forecast period.
- Based on service, the regulatory strategy & nonclinical development planning segment held the largest market share in 2025.
- Based on application, the pre-IND stage segment held the highest market share in 2025.
- Based on therapeutic area, the oncology segment held the highest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 0.965 Billion
- 2033 Projected Market Size: USD 2.39 Billion
- CAGR (2026-2033): 12.2%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
In addition, increased demand for expert regulatory guidance during the preclinical stages and stringent regulatory requirements from authorities such as the U.S. FDA and the European Medicines Agency, which require comprehensive safety and pharmacology data before the clinical trials, are other key driving factors contributing to the market growth.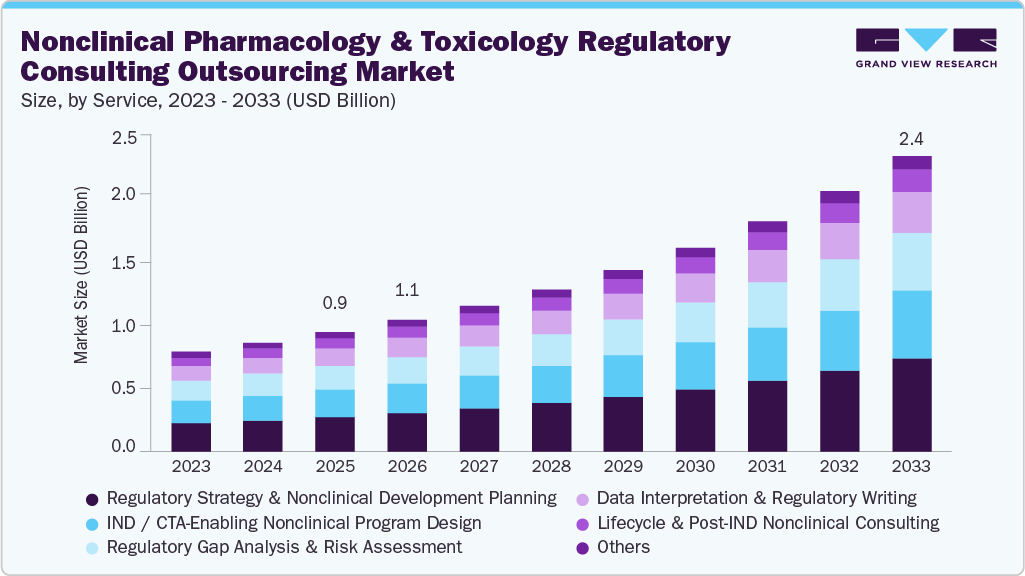
The market for nonclinical pharmacology and toxicology regulatory consulting outsourcing is driven by the growing complexity of biologics and advanced therapies, increasing global regulatory scrutiny and safety mandates, an increase in decentralized trials, and a paradigm shift toward new approach methodologies (NAMs).
In addition, increasing shift from conventional small-molecule drugs to complex biologics and advanced therapeutic modalities is a major factor driving the growth of the global nonclinical pharmacology and toxicology regulatory consulting outsourcing market. The biopharmaceutical pipeline is rapidly expanding, with modalities such as cell and gene therapies, multi-specific antibodies, RNA-based therapeutics, and gene-editing platforms that require highly specialized preclinical evaluation strategies.
According to the American Society of Gene & Cell Therapy (ASGCT), by Q4 2025, approximately 4,238 advanced therapies were under development, including 22% cell therapies, 29% RNA therapies, and 49% gene therapies. Unlike traditional small molecules, these complex biologics present unique scientific and regulatory challenges, including immunogenicity risks, vector biodistribution assessment, off-target gene-editing effects, and limited availability of relevant animal models. Thus, standard toxicology and pharmacology testing frameworks are often inadequate, requiring customized study designs, advanced bioanalytical approaches, and specialized regulatory interpretation. In addition, evolving global regulatory frameworks such as regenerative medicine pathways in the U.S. and advanced therapy medicinal product (ATMP) regulations in Europe require extensive expertise in preparing nonclinical regulatory packages. As a result, pharmaceutical and biotechnology companies increasingly depend on specialized regulatory consulting providers to design compliant nonclinical strategies, interpret complex datasets, and support IND-enabling submissions. This growing reliance on external expertise for advanced therapy development is expected to drive the market growth over the forecast period.
Furthermore, regulatory authorities across the globe are strengthening safety oversight and harmonizing standards to ensure the safety and quality of new therapeutics. Agencies such as the U.S. FDA and the EMA continue to update guidelines emphasizing data integrity, transparency, and lifecycle-based safety monitoring. Thus, the increasing stringency of global regulatory frameworks is a key factor driving the demand in the nonclinical pharmacology and toxicology regulatory consulting outsourcing market. In addition, increasing presence of biologics, gene therapies, and oligonucleotide-based therapeutics in decentralized trial pipelines is propelling the requirements for specialized studies such as tissue cross-reactivity assessments, immunotoxicology characterizations, and scientifically species selection.
Moreover, accelerated timelines associated with decentralized trials ideally require sponsors to conduct multiple nonclinical safety studies in parallel to support quick clinical trial initiation, leading to greater reliance on external expertise for study design and regulatory strategy. The increasing adoption of NAMs is emerging as a significant driver for the nonclinical pharmacology and toxicology regulatory consulting outsourcing market. NAMs include advanced testing strategies such as in vitro cell-based assays, organ-on-chip systems, high-throughput screening platforms, computational toxicology models, and artificial intelligence-powered predictive toxicology tools. Regulatory authorities around major markets are fostering the integration of these alternative approaches to supplement or replace traditional animal testing, while enhancing the accuracy of preclinical safety evaluations.
Opportunity Analysis
The nonclinical pharmacology and toxicology regulatory consulting outsourcing market is experiencing growth due to increasing complexity of modern therapeutic pipelines. Developing biologics, gene therapies, RNA therapeutics, antibody-drug conjugates, and multi-specific antibodies demands specialized nonclinical evaluation strategies that go beyond those used for traditional small molecules. These therapies involve complex mechanisms of action, distinct biodistribution patterns, and potential immunogenicity or long-term safety concerns, which requires customized toxicology and pharmacology studies. As a result, pharmaceutical and biotech companies are increasingly dependent on regulatory consulting companies to create modality-specific safety evaluation frameworks, justify species selection, support biodistribution and immunotoxicology testing, and prepare the necessary documentation for IND and CTA submissions.
In addition, the rapid growth of biotechnology startups and virtual drug development models presents substantial market expansion opportunities. Several emerging biotech companies operate with limited internal infrastructure, focusing mainly on innovation, intellectual property development, and clinical strategy. As these companies develop new therapies for clinical trials, they often lack in-house expertise in regulatory toxicology, global regulatory frameworks, and nonclinical safety program design. Thus, regulatory consulting providers play a strategic role by acting as external scientific advisors, supporting interactions with CROs, guiding nonclinical development strategies, and preparing regulatory submissions.
Similarly, the globalization of drug development and expansion into emerging markets offers a strong opportunity for regulatory consulting providers. The emergence of next-generation therapeutic modalities and first-in-class technologies provides opportunity for consulting providers. Innovative platforms such as Proteolysis Targeting Chimeras (PROTACs), gene-editing therapies, and advanced biologics often lack well-established regulatory guidelines, leading to uncertainty for sponsors during preclinical development. Thus, regulatory consulting firms with deep expertise in regulatory science collaborate with sponsors to develop customized safety evaluation strategies and proactively engage with regulatory authorities to establish acceptable development pathways. These customized strategies support sponsors secure enhance development pathways and reduce regulatory uncertainty for novel therapeutic technologies.
Impact of U.S. Tariffs on Nonclinical Pharmacology and Toxicology Regulatory Consulting Outsourcing Market
The tariffs imposed by the U.S. government on pharmaceuticals are affecting the nonclinical pharmacology and toxicology outsourcing services market, particularly by increasing input costs for early-stage pharmaceutical development, including comparator drugs, reagents, and assay kits. This, in turn, is affecting pharmaceutical companies' R&D spending, especially toxicology studies, which are being constrained by budget constraints. On the other hand, the tariffs had a favorable impact on the outsourcing services market, especially in the localization of nonclinical development services, which is impacting demand for domestic consulting services. This is creating a new outsourcing services scenario, wherein domestic-based regulatory toxicology consultants are benefiting from increased outsourcing, especially in contrast to their counterparts based in other countries. Supply chain disruptions in imported and proprietary biologic compounds are increasing the complexity of toxicology studies, thereby increasing the need for specialized services.
Moreover, companies are increasingly seeking consulting companies to redesign study protocols and sourcing strategies to address tariff risk, thereby making the role of a regulatory consultant even more integral to the business strategy process. However, increased development costs and pricing pressure could lead to a decline in outsourcing volumes from small biotech companies, as they may delay preclinical programs.
Technological Advancements
Technological advancements are creating new growth opportunities within the market. The integration of artificial intelligence (AI) and machine learning (ML), in silico modeling and simulation, organ-on-chip technology, high throughput screening (HTS), cloud-based regulatory information management systems (RIMS) is transforming the early stages of drug development.

AI and ML enhance data analysis, predictive toxicology, and regulatory decision-making by identifying patterns across large biological datasets. These technologies support automated report generation, risk assessment, and biomarker identification, improving submission accuracy, reducing timelines, and enabling more efficient interactions with regulatory authorities in nonclinical consulting workflows. In addition, AI-driven models enable researchers to analyze large volumes of historical safety data, identify early toxicity signals, and predict potential adverse effects before initiating costly in vivo studies. As regulatory agencies gradually explore frameworks for accepting AI-generated data in regulatory submissions, consulting companies with expertise in computational toxicology and digital safety modeling are likely to gain a competitive advantage. These companies assist sponsors in validating AI algorithms, integrating predictive models into regulatory strategies, and developing virtual control groups that may reduce reliance on animal testing. Thus, regulatory consultants have the opportunity to evolve from traditional advisory roles to strategic partners capable of integrating data science with regulatory compliance.
In silico tools use computational models to predict pharmacokinetics, toxicity, and drug interactions, minimizing reliance on animal studies. These simulations support dose selection, safety evaluation, and regulatory documentation. They enable faster, cost-effective decision-making while aligning with regulatory expectations for model-informed drug development strategies.
Organ-on-chip systems replicate human organ physiology on microfluidic chips, enabling precise toxicity and efficacy testing. These platforms improve translational accuracy compared to traditional models, supporting regulatory submissions with human-relevant data. They reduce animal testing and enhance safety assessment in nonclinical pharmacology and toxicology studies. This system is expected to create new growth opportunities for consulting providers. As regulators promote reduced reliance on traditional animal testing while ensuring thorough safety assessments, sponsors require guidance on integrating these innovative methods into regulatory submissions. Consulting companies that specialize in translational safety science and regulatory interpretation of NAM-generated data are therefore well-positioned to support the industry’s shift toward more predictive and efficient nonclinical testing frameworks. HTS technologies enable rapid testing of thousands of compounds for biological activity and toxicity. Automation, robotics, and advanced analytics accelerate lead identification and safety profiling. In regulatory consulting, HTS data supports early risk assessment, improves candidate selection, and strengthens nonclinical data packages for submissions.
Cloud-based RIMS centralize regulatory data, streamline document management, and ensure real-time collaboration across global teams. These systems enhance compliance, version control, and audit readiness. They facilitate efficient submission preparation, tracking, and lifecycle management. Regulatory consulting companies with deep expertise in regulatory science can collaborate with sponsors to develop customized safety evaluation strategies and proactively engage with regulatory authorities to establish acceptable development pathways. These customized strategies can help sponsors secure expedited development pathways and reduce regulatory uncertainty for novel therapeutic technologies.
Pricing Model Analysis
The costs of regulatory affairs are increasing by over 10% year-over-year due to expanding markets, rising complexity in global regulatory requirements, and greater compliance challenges. These trends are expected to persist in the near future. Life sciences companies need strategies to control regulatory costs and improve productivity and scalability to meet resource demands. Thus, the demand for outsourcing services is growing, which is also driving the need for various pricing strategies.
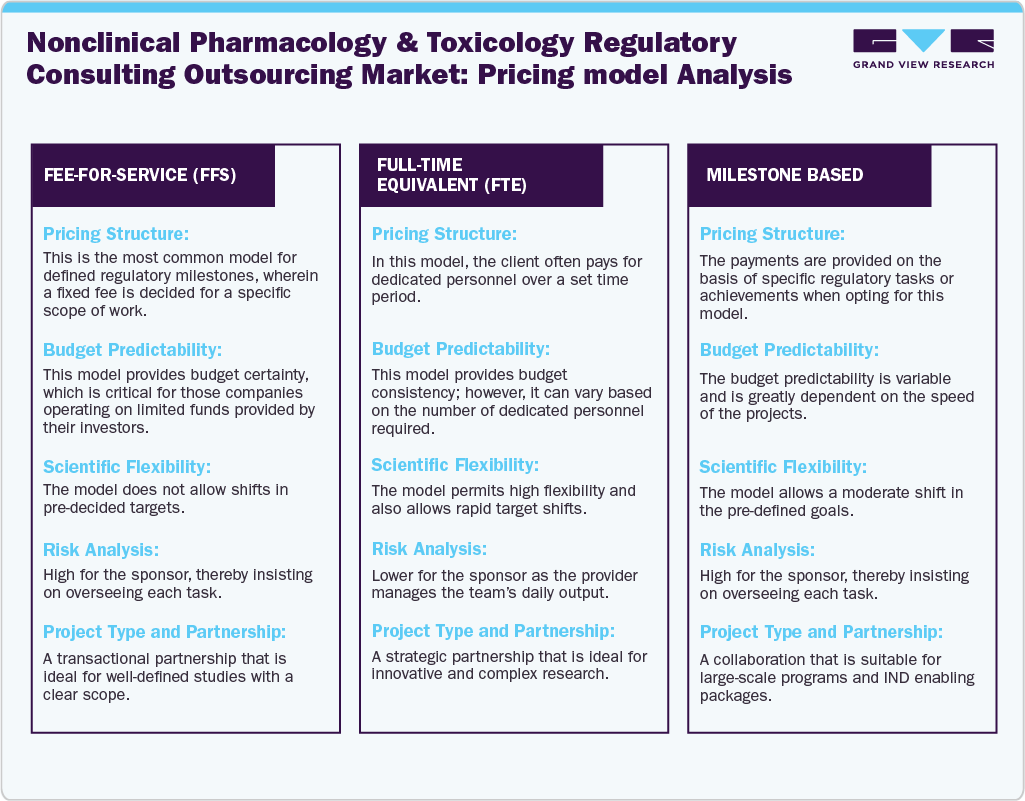
Pricing models in the nonclinical pharmacology and toxicology consulting outsourcing sector have shifted from traditional hourly rates to more value- and risk-based approaches, reflecting the needs of both physical and virtual firms driven by industry evolution. As drug development grows more complex, pricing increasingly depends on the scarcity of expertise rather than just labor costs. The standard pricing models mentioned earlier, the market has developed to include several complex arrangements that serve the manufacturer’s interests. For instance, the leased competence model involves external experts from consulting firms being integrated directly into the sponsor’s internal team for a specific period. They may use their own infrastructure or the sponsor’s facilities to meet particular milestones. Similarly, a program-based model provides an expanded version of FSOs, which manages the entire lifecycle of a drug, with the consultant overseeing all regulatory activities for that asset. Similarly, larger biopharmaceutical companies use a mix of these models as per their requirements.
Market Concentration & Characteristics
The nonclinical pharmacology and toxicology regulatory consulting outsourcing market growth stage is anticipated to be medium, and the pace of the market is accelerating. The market is characterized by the level of merger & acquisitions activities, the degree of innovation, regulatory impact, product expansion, and regional expansion.
The degree of innovation in this market is driven by service providers using new technologies, including digital tools, predictive toxicology, and AI-based data analysis. The use of in-silico science, high-throughput screening, and bioanalytical tools is increasing, in collaboration with biotechnology and pharmaceutical companies. The constant innovations in lab technologies are allowing for more efficient decision-making and submission of reports.

The evolving global regulations, including new requirements from the FDA and EMA, are impacting nonclinical studies worldwide. The new requirements are also making it essential for companies to maintain documentation, follow GLP, and ensure transparent reporting of data. This is also making it more favorable for companies to outsource their services to consultants who are well-versed in regulations. This is increasing complexity, but it also provides a competitive advantage for companies.
The market is experiencing a rise in merger and acquisition deals, with companies seeking to improve their scientific services and expand their global presence. Market players acquire niche consulting firms and toxicology service providers, which specialize in delivering services in areas like preclinical safety assessments and regulatory strategies. This companies enable companies to deliver more integrated services, which are helpful in addressing clients’ needs in a competitive outsourcing environment.
Service providers are expanding their services beyond traditional toxicology and pharmacology consulting services. They deliver a range of services, including study design, regulatory writing, risk assessments, IND-enabling services, and lifecycle management services. These service providers are supportive in making outsourcing easier for clients, as they are receiving a more holistic service from service providers.
The expansion of operations to regions such as North America, Europe, and the Asia Pacific remains an important strategy. The emerging regions of operation, especially in Asia, are gaining momentum due to reduced operational costs and an increase in pharmaceutical R&D activities. By establishing operations in these regions, consulting companies would be able to better align with local regulatory requirements. This would also be beneficial for global clients to execute nonclinical programs across regions.
Service Insights
On the basis of service segment, in 2025, the regulatory strategy and nonclinical development planning segment held the largest market share in the market, accounting for a revenue share of 29.11%. Regulatory strategy and nonclinical development planning are crucial drivers for this market, as pharmaceutical and biotechnology companies increasingly seek expert guidance to navigate complex global regulatory requirements. Early strategic planning helps optimize study design, reduce development risks, and align nonclinical programs to meet regulatory expectations from agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency. These factors are expected to support market growth over the forecast period.
The IND / CTA-enabling nonclinical program design segment is expected to grow significantly during the forecast period. IND and CTA-enabling nonclinical program design involves strategic planning and execution of preclinical studies required to support regulatory submissions for initiating human clinical trials. This segment is driven by the need to demonstrate the safety, pharmacology, and toxicological profiles of a drug candidate before beginning human trials. Pharmaceutical and biotechnology companies must generate solid nonclinical data to support regulatory filings such as an Investigational New Drug (IND) application in the U.S. and a Clinical Trial Application (CTA) in other global markets. Compliance with guidelines established by authorities like the U.S. FDA, European Medicines Agency, and the International Council for Harmonization is a key factor in guiding these programs. Such trends are expected to boost segment growth over the projected period.
Application Insights
On the basis of application, the pre-IND stage segment accounted for the largest share in 2025 during the forecast period. The pre-IND stage is a crucial phase in drug development, as it involves generating vital nonclinical pharmacology, toxicology, and safety data that are necessary for supporting discussions with regulatory bodies. During this stage, companies typically consult experts to design studies, assess risks, and prepare the necessary documentation to meet the requirements of agencies such as the U.S. FDA and the European Medicines Agency. Proper planning in this phase can effectively streamline future IND submissions and minimize delays in the development process.
The IND / CTA submission support segment is anticipated to grow at the fastest CAGR of 13.6% over the forecast period. The trend toward outsourcing pre-IND consulting is fueled by the growth of biotechnology startups and emerging pharmaceutical companies, many of which lack in-house regulatory expertise.The pre-IND stage outlines the nonclinical strategy essential for regulatory submissions. Besides, the outsourcing services offers stages that involves obtaining expert guidance to create pharmacology, toxicology, pharmacokinetics, and safety studies that fulfill regulatory expectations before submitting an IND application to the U.S. FDA. Consulting partners aid sponsors in establishing scientific development pathways, preparing regulatory briefing documents, and conducting gap assessments to ensure that nonclinical data packages comply with global regulatory standards, such as those set by the International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use. Such factors are expected to drive the market growth.
Modality Insights
On the basis of modality, the small molecules segment dominated the market with the largest revenue share in 2025. Small molecules remain a key modality in drug development due to their well-established manufacturing processes, predictable pharmacokinetics, and oral bioavailability. Companies rely on nonclinical pharmacology and toxicology studies aligned with guidance from the U.S. FDA and European Medicines Agency.
Nonclinical pharmacology and toxicology consulting services assist sponsors in designing and interpreting preclinical studies, including safety pharmacology, toxicokinetics, repeat-dose toxicity, genotoxicity, and reproductive toxicity assessments. These studies are essential to evaluate the safety profile and biological activity of small molecule compounds before they enter clinical trials. Consulting partners support the preparation of regulatory submissions, including nonclinical sections of IND, CTA, NDA, and other regulatory filings. Their role includes regulatory strategy development, data interpretation, gap analysis, and ensuring that study results comply with international regulatory guidelines and Good Laboratory Practice (GLP) standards.
The cell and gene therapies segment is anticipated to grow at the fastest CAGR over the forecast period. Cell & gene therapies are emerging as transformative treatment modalities, creating significant demand within the nonclinical pharmacology and toxicology regulatory consulting outsourcing market. These advanced therapies involve the modification or replacement of genetic material or the use of living cells to treat, prevent, or potentially cure complex diseases, including cancer, rare genetic disorders, and autoimmune conditions. Due to their innovative nature and complex biological mechanisms, cell & gene therapies require specialized nonclinical evaluation and regulatory expertise, making regulatory consulting outsourcing an essential component of development programs.
Therapeutic Area Insights
On the basis of therapeutic area, the oncology segment accounted for the largest share in 2025 during the forecast period. The segment growth is driven by increasing global cancer burden and strong drug development pipelines. Companies require extensive nonclinical pharmacology and toxicology evaluation aligned with regulatory guidance from the U.S. FDA and European Medicines Agency. The cardiovascular & metabolic segment is anticipated to grow at the fastest CAGR over the forecast period. Cardiovascular and metabolic diseases continue to be significant therapeutic areas because of the increasing prevalence of conditions such as cardiovascular disease and Type 2 Diabetes. Drug developers need comprehensive nonclinical pharmacology and toxicology studies to assess safety, efficacy, and metabolic effects before regulatory submissions and clinical trials.
The cardiovascular & metabolic segment is expected to grow significantly during the forecast period. Currently the cardiovascular diseases is witnessing rising requirement for nonclinical pharmacology and toxicology regulatory outsourcing driven by rising incidence of chronic conditions such as cardiovascular disease, type 2 diabetes, obesity, and hypertension is prompting pharmaceutical and biotechnology companies which is expected to drive the development of innovative therapeutics. In this context, outsourced regulatory consulting partners play a crucial role in designing robust nonclinical strategies that adhere to global regulatory expectations while mitigating development risks.
In the market, drug candidates are increasingly targeting cardiovascular and metabolic conditions that require comprehensive safety, pharmacology, and toxicology assessments, including cardiac electrophysiology studies, metabolic profiling, and long-term toxicity evaluations. Regulatory consultants aid sponsors in structuring nonclinical programs that comply with benchmarks set by agencies such as the U.S. FDA and the European Medicines Agency, thereby further contributing to market growth. Their expertise ensures that study designs align with regulatory requirements for Investigational New Drug (IND) or Clinical Trial Application (CTA) submissions, thereby bolstering scientific credibility and regulatory readiness.
End Use Insights
On the basis of end-use, the pharmaceutical and biopharmaceutical companies segment accounted for the largest share in 2025 during the forecast period. Pharmaceutical and biopharmaceutical companies are increasingly relying on outsourced nonclinical pharmacology and toxicology regulatory consulting services to streamline drug development and ensure compliance with evolving global regulatory requirements. As drug pipelines expand to include complex modalities such as biologics, cell and gene therapies, and targeted small molecules, the need for specialized expertise in nonclinical study design, data interpretation, and regulatory documentation has increased. Moreover, consulting partners support companies in preparing regulatory submissions, including IND and CTA packages, while ensuring alignment with international regulatory requirements.
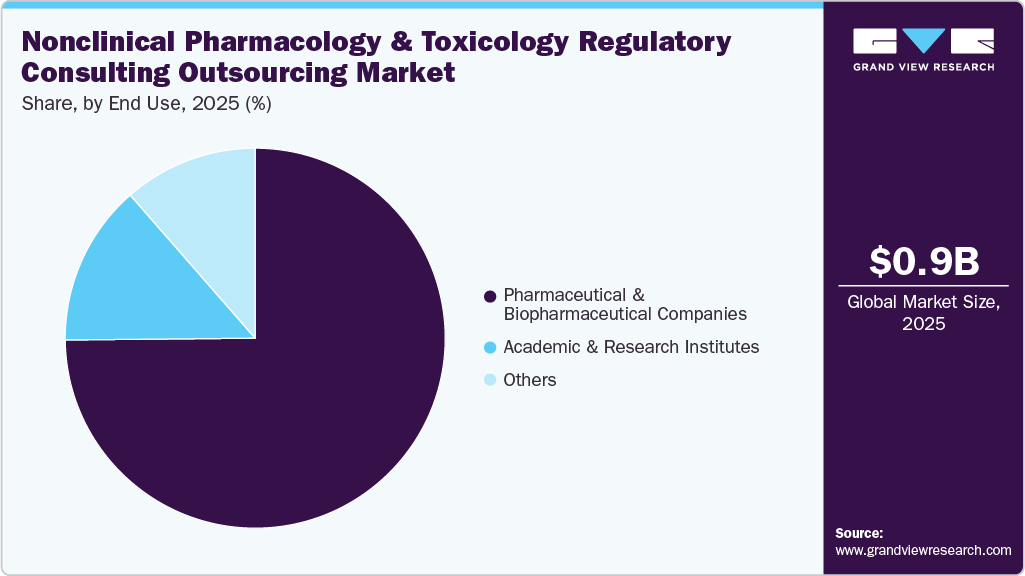
The academic and research institutes segment is anticipated to grow at a significant CAGR over the forecast period. Academic and research institutes play an important role in the nonclinical pharmacology and toxicology regulatory consulting outsourcing market, supporting pharmaceutical and biopharmaceutical companies with specialized scientific expertise and advanced research capabilities. These institutions often possess strong preclinical research infrastructure, experienced toxicologists, pharmacologists, and translational scientists who contribute to the design and evaluation of nonclinical studies required for regulatory submissions. Their involvement helps ensure that early-stage drug candidates are assessed using scientifically robust, regulatory-compliant methodologies.
Regional Insights
North America Nonclinical Pharmacology And Toxicology Regulatory Consulting Outsourcing Market Trends
North America's nonclinical pharmacology and toxicology regulatory consulting outsourcing market dominated the largest revenue share of 39.9% in 2025. The growth of this region is due to increasing biologics and advanced therapy research, complex regulatory requirements, demand for specialized toxicology expertise, and greater reliance on outsourced regulatory strategy services.
The region is witnessing the rapid expansion of biotechnology and advanced therapeutic research in the U.S. and Canada, driven by the increasing development of biologics, gene therapies, and cell-based treatments, which require comprehensive nonclinical pharmacology and toxicology evaluations before clinical trials. Besides, most pharmaceutical and emerging biotech companies are outsourcing regulatory consulting and safety assessment activities to specialized companies in large numbers to ensure compliance, accelerate development timelines, and manage complex scientific documentation for IND submissions. In addition, the regulatory environment in North America is governed by the U.S. FDA and Health Canada, requiring strict compliance with GLP standards and comprehensive nonclinical safety data before clinical trials, which further drives market growth.
U.S. Nonclinical Pharmacology And Toxicology Regulatory Consulting Outsourcing Market Trends
The nonclinical pharmacology and toxicology regulatory consulting outsourcing market in the U.S. accounted for the highest market share in the North American market. The market growth is driven by expanding biologics and gene therapy pipelines, rising drug development costs, and greater reliance on outsourced regulatory consulting and safety evaluation services. Some other factors contributing to market growth are expanding biologics pipelines in the U.S. and the rising need for regulatory approvals, which further contribute to the increased requirement of outsourcing regulatory activities to specialized consulting providers. According to a report of March 2026, CDER approved 46 novel drugs into the market that were not approved before. Similarly, in the past two years, the U.S. FDA’s Center for Drug Evaluation and Research (CDER) approved 96 novel drugs, of which biologics accounted for about 29% (28 approvals). In the same period, the Center for Biologics Evaluation and Research (CBER) approved an additional 24 biological products. Thus, a growing number of approvals in the market is expected to drive the market growth.
Moreover, strict regulatory oversight from the U.S. Food and Drug Administration (FDA), which requires comprehensive nonclinical safety data before clinical trials, further drives the U.S. market growth. For instance, in March 2026, the FDA placed a partial clinical hold on PepGen’s FREEDOM2 trial due to questions related to preclinical pharmacology and toxicology studies. Such regulatory scrutiny increases the need for experienced consultants who can support robust nonclinical data analysis, regulatory documentation, and risk mitigation strategies to meet evolving FDA expectations.
The Canada nonclinical pharmacology and toxicology regulatory consulting outsourcing market is driven by the rapid expansion of the biotechnology sector and rising investments in pharmaceutical R&D. In addition, supportive government funding initiatives and strong collaborations between academic institutions and industry players are propelling drug development innovations. Besides, growing regulatory complexity and the rising need for specialized expertise are expected to encourage pharmaceutical and biotechnology companies to increasingly rely on external regulatory consulting partners to streamline nonclinical development and ensure compliance with evolving regulatory requirements.
Furthermore, the companies are competing by offering integrated regulatory strategy, development planning, and scientific documentation services. In addition, smaller specialized consultancies are participating in the market by providing focused toxicology expertise, flexible engagement models, and customized regulatory advisory services for emerging biotech companies. Such factors are expected to drive the country’s growth over the estimated time period.
Europe Nonclinical Pharmacology And Toxicology Regulatory Consulting Outsourcing Market Trends
The market for nonclinical pharmacology and toxicology regulatory consulting outsourcing in Europe is driven by expanding biologics and advanced therapy development, rising pharmaceutical R&D investments, the growing number of biotech startups, and the increasing reliance on specialized regulatory consulting expertise. Currently, in the European market, increasing investments in pharmaceutical R&D are fueling demand for innovation, further expanding the drug development pipeline. Besides, funding from governments, private investors, and pharmaceutical companies to enhance research in biologics, advanced therapies, and precision medicine contributes to market growth. According to the European Federation of Pharmaceutical Industries and Associations' November 2024 report, in 2023, the pharmaceutical industry invested about USD 60,528 million in R&D in Europe.
In addition, according to the 2024 EU Industrial R&D Investment Scoreboard, health industries spent USD 298 billion globally, representing 20.5% of total business R&D expenditure worldwide. In addition, the growing number of biotechnology startups and innovation hubs across European countries is driving market growth. Many early-stage companies focus on discovery and translational research while relying on external regulatory consultants to support development strategies, scientific documentation, and interactions with regulatory authorities to advance therapeutic candidates efficiently. These factors are expected to drive market growth over the estimated time period.
The nonclinical pharmacology and toxicology regulatory consulting outsourcing market in Germany is expanding due to its strong government funding for life sciences, emerging focus on biosimilars development, and expansion of contract research infrastructure. In addition, Germany’s strong public funding and policy support for pharmaceutical innovation are expected to drive the market growth. For instance, some of the programs led by the Federal Ministry of Education and Research (BMBF) and national innovation clusters is providing funding for translational medicine and drug discovery projects, along with increasing the need for regulatory consulting services to support development planning and scientific documentation, further contributing to market growth.
Similarly, Germany’s leadership in biosimilars and the development of complex biologics is anticipated to contribute to market growth. As German pharmaceutical companies are actively developing biosimilar therapies and advanced biologics for global markets. These programs require specialized regulatory strategies and detailed regulatory documentation aligned with European requirements, prompting companies to outsource nonclinical pharmacology and toxicology consulting services to experienced regulatory experts. Such factors are expected to drive the market.
The UK nonclinical pharmacology and toxicology regulatory consulting outsourcing market is rising due to a strong biotechnology ecosystem, supportive government innovation programs, and increasing outsourcing by emerging biotech companies. Besides, major advancements in the UK are supported by leading universities, research institutes, and innovation clusters. Numerous early-stage biotech companies focus on drug discovery and translational research while relying on external consultants for regulatory strategy, development planning, and documentation support.
Moreover, increasing government initiatives and funding to promote pharmaceutical innovation and advanced therapeutic development are anticipated to drive the market growth. For instance, in January 2024, the UK strengthened its leadership in cell and gene therapy, securing 55% of Europe’s cell and gene therapy (CGT) venture capital in 2023, with 84 drugs in development and 23 therapies already approved for patient use. Such programs supporting cell and gene therapy research, along with collaboration between academia and industry, are encouraging companies to accelerate drug development, which increases demand for specialized regulatory consulting expertise. Such factors are anticipated to drive the market.
Asia Pacific Nonclinical Pharmacology And Toxicology Regulatory Consulting Outsourcing Market Trends
Asia Pacific is expected to grow at a significant CAGR over the forecast period. The region’s growth is driven by rising biotechnology innovation, growing government initiatives supporting drug development, cost advantages for outsourcing, and increasing regulatory complexity across emerging pharmaceutical markets. The cost-efficient research environment and strong CRO infrastructure across several Asia Pacific countries are advancing the region’s growth. Some countries, such as India, China, and South Korea, offer skilled scientific talent, advanced research facilities, and competitive operational costs. These advantages encourage global pharmaceutical and biotechnology companies to outsource development support activities, including regulatory consulting, to regional service providers that offer scientific expertise and operational efficiency.
China's nonclinical pharmacology and toxicology regulatory consulting outsourcing market is driven by expanding advanced biopharmaceutical research, the adoption of NAMs, the growing number of international collaborations, and the rising demand for specialized regulatory strategy support. The country’s expanding research into advanced therapeutics, including biologics, regenerative medicine, and precision therapies, is expected to drive market growth. These complex drug modalities require specialized regulatory planning and structured scientific documentation. Pharmaceutical and biotechnology companies, therefore, rely on regulatory consulting firms in large numbers to support development strategy, documentation preparation, and regulatory interactions with national authorities.
In addition, there is an increasing adoption of innovative research technologies and global collaborations in toxicology and drug development. For instance, in January 2026, VivoSim Labs expanded Asia-Pacific access to its NAMKind human-based toxicology services through distributor agreements in South Korea and China, reflecting the region’s growing demand for advanced methodologies and faster decision-making in drug development programs.
The nonclinical pharmacology and toxicology regulatory consulting outsourcing market in Japan is witnessing new growth opportunities attributed to expanding innovation landscape in advanced therapies, including regenerative medicine, biologics, and precision therapeutics. Most pharmaceutical and biotechnology companies developing these complex modalities ideally require specialized regulatory guidance for development planning, documentation, and interactions with regulatory authorities, thereby further contributing to market growth.
Besides, most consulting companies support companies in developing strategies, regulatory submissions, and conducting scientific consultation meetings with regulators, which has led to a rising demand for outsourcing regulatory expertise.
The nonclinical pharmacology and toxicology regulatory consulting outsourcing market in India is expected to experience significant growth at a significant CAGR during the forecast period. The country’s growth is driven by rising government initiatives supporting drug discovery, increasing contract research infrastructure, strong academic research networks, and growing biologics and biosimilars development.
In addition, the country is experiencing rapid expansion of the contract research ecosystem, with numerous CROs, research institutes, and biotechnology companies engaged in early-stage drug development. These organizations require regulatory consulting expertise to support development planning, scientific documentation preparation, and interactions with regulatory authorities.
Latin America Nonclinical Pharmacology And Toxicology Regulatory Consulting Outsourcing Market Trends
Latin America's nonclinical pharmacology and toxicology regulatory consulting outsourcing market is expected to grow at a steady rate over the forecast period, driven by expanding pharmaceutical manufacturing investments, increasing government support for life sciences, improving regulatory harmonization efforts, and the region’s cost-efficient research and development environment.
In the region, the growing presence of multinational pharmaceutical companies, including research collaborations and outsourcing regulatory support to navigate diverse regulatory requirements across countries is contributing to market growth. Besides, growing clinical development activities in Brazil, Argentina, and Chile are encouraging demand for specialized consulting expertise in regulatory documentation and submission strategies.
The nonclinical pharmacology and toxicology regulatory consulting outsourcing market in Brazil is expanding due to regulatory digitalization initiatives, modernization of regulatory submission systems, increasing participation of global biopharmaceutical companies, growing domestic biotech activity, and stronger alignment with international regulatory documentation standards.
The country is experiencing a rise in regulatory modernization led by the National Health Surveillance Agency (ANVISA). For instance, in November 2025, the partnership between Certara and ANVISA to implement eCTD 4.0 submissions via the GlobalSubmit software strengthens the digital regulatory infrastructure, improves submission workflows, and accelerates regulatory review processes for pharmaceutical products. Such initiatives propel market growth.
Middle East and Africa Nonclinical Pharmacology And Toxicology Regulatory Consulting Outsourcing Market Trends
The Middle East and Africa market is driven by growing biotechnology research initiatives, increasing regulatory modernization efforts, rising participation of multinational pharmaceutical companies, and strengthening regional healthcare innovation strategies. The growing focus on biosimilar and biopharmaceutical production in countries such as Saudi Arabia, the UAE, and South Africa is advancing the market growth.
Besides, the regional government is encouraging domestic biologics manufacturing through incentives and partnerships with international companies, increasing demand for regulatory consulting expertise to support complex biologics development documentation. For instance, in November 2025, Boston Oncology Arabia and Saudi Arabia’s Local Content & Government Procurement Authority signed an agreement to localize manufacturing of two biosimilar biologics, strengthening national drug security and supporting Vision 2030 healthcare localization goals.
The South African market is growing steadily, driven by expanding drug discovery initiatives, international research collaborations, rising pharmaceutical R&D investments, and expanding global partnerships that support South Africa’s life sciences innovation ecosystem. In addition, one of the major factors contributing to market growth is the strengthening of drug discovery capabilities through international partnerships. Moreover, increasing global investment to expand pharmaceutical and vaccine manufacturing capabilities across the continent is also driving market growth. For instance, in October 2025, EU’s €190 (USD 220) million Global Gateway investment under the Manufacturing and Access to Vaccines (MAV+) initiative supports local health product manufacturing in Africa, including South Africa, strengthening the regional life sciences ecosystem and creating greater demand for regulatory consulting services.
The UAE market is expanding due to increasing pharmaceutical innovation initiatives, the adoption of artificial intelligence in drug development, the establishment of the Emirates Drug Establishment, and rising collaborations with global biotechnology companies. Besides, the country's growth is driven by the growing adoption of artificial intelligence in pharmaceutical research. For instance, in February 2026, the launch of the InSilico Medicine AI project at the World Health Expo 2026 aimed to reduce research costs, accelerate drug discovery processes, and improve compound evaluation through advanced digital modeling. Such an initiative may enhance regulatory reliability, bolstering market growth.
Key Nonclinical Pharmacology And Toxicology Regulatory Consulting Outsourcing Company Insights
Market players are undertaking various strategic initiatives, such as the launch of new services, collaborations, partnerships, and mergers & acquisitions, to strengthen their service offering and provide a competitive advantage. For instance, in September 2025, Parexel partnered with Weave Bio to integrate AI-driven regulatory automation, improving preparation of regulatory submissions, including nonclinical IND documentation. Such initiatives are expected to boost the market’s growth.
Key Nonclinical Pharmacology and Toxicology Regulatory Consulting Outsourcing Companies:
The following key companies have been profiled for this study on the nonclinical pharmacology and toxicology regulatory consulting outsourcing market.
- Parexel International (MA) Corporation
- ToxStrategies
- Certara
- Dark Horse Consulting Group
- ELIQUENT Life Sciences
- SciLucent
- Latham BioPharm Group (Sia Partners)
- Salamandra
- Akkeri Inc.
- Aclairo Pharmaceutical Development Group, Inc.
- Biologics Consulting Group, Inc.
Recent Developments
-
In September 2025, Parexel partnered with Weave Bio to integrate AI-driven regulatory automation, improving preparation of regulatory submissions, including nonclinical IND documentation.
-
In June 2025, ToxStrategies strengthened regulatory toxicology expertise by appointing former U.S. FDA scientist. Such a move enhances regulatory insight in nonclinical safety assessments, supporting demand for specialized consulting services in the market.
-
In October 2024, Dark Horse Consulting Group acquired BioTechLogic to expand expertise in manufacturing, quality systems, and CMC regulatory consulting for advanced therapies, strengthening integrated development support and enhancing capabilities within the market.
Nonclinical Pharmacology And Toxicology Regulatory Consulting Outsourcing Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 1.06 billion
Revenue forecast in 2033
USD 2.39 billion
Growth rate
CAGR of 12.2% from 2026 to 2033
Actual data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD billion/million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Service, application, modality, therapeutic area, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Thailand; South Korea; Australia; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait; Oman; Qatar
Key companies profiled
Parexel International (MA) Corporation; ToxStrategies; Certara; Dark Horse Consulting Group; ELIQUENT Life Sciences; SciLucent; Latham BioPharm Group (Sia Partners); Salamandra; Akkeri Inc.; Aclairo Pharmaceutical Development Group, Inc.; Biologics Consulting Group, Inc.
Customization scope
Free report customization (equivalent to up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Nonclinical Pharmacology And Toxicology Regulatory Consulting Outsourcing Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global nonclinical pharmacology and toxicology regulatory consulting outsourcing market report based on service, application, modality, therapeutic area, end use, and region:
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Regulatory Strategy & Nonclinical Development Planning
-
IND / CTA-Enabling Nonclinical Program Design
-
Regulatory Gap Analysis & Risk Assessment
-
Data Interpretation & Regulatory Writing
-
Lifecycle & Post-IND Nonclinical Consulting
-
Others
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Pre-IND Stage
-
IND / CTA Submission Support
-
BLA / MAA
-
Others
-
-
Modality Outlook (Revenue, USD Million, 2021 - 2033)
-
Small Molecules
-
Biologics (mAbs, Proteins)
-
Cell and Gene Therapies
-
Others
-
-
Therapeutic Area Outlook (Revenue, USD Million, 2021 - 2033)
-
Oncology
-
Autoimmune & Inflammatory Diseases
-
CNS
-
Cardiovascular & Metabolic
-
Infectious Diseases
-
Rare Diseases
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical and Biopharmaceutical Companies
-
Academic and Research Institutes
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Thailand
-
South Korea
-
Australia
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
Qatar
-
Oman
-
-
Frequently Asked Questions About This Report
b. The global nonclinical pharmacology and toxicology regulatory consulting outsourcing market size was estimated at USD 0.97 billion in 2025 and is expected to reach USD 1.06 billion in 2026.
What is the nonclinical pharmacology and toxicology regulatory consulting outsourcing market growth?b. The global nonclinical pharmacology and toxicology regulatory consulting outsourcing market is expected to grow at a compound annual growth rate of 12.2% from 2026 to 2033 to reach USD 2.39 billion in 2033.
b. The regulatory strategy & nonclinical development planning segment dominated the nonclinical pharmacology and toxicology regulatory consulting outsourcing market with a share of 29.11% in 2025. The market growth is attributed to the increasing trend of outsourcing specialized regulatory expertise, the expansion of global clinical pipelines, and the need to minimize regulatory risks, support regulatory strategy, and nonclinical development planning services, driving growth for the market.
b. Some key players operating in the nonclinical pharmacology and toxicology regulatory consulting outsourcing market include Parexel International (MA) Corporation, ToxStrategies, Certara, Dark Horse Consulting Group, ELIQUENT Life Sciences, SciLucent, Latham BioPharm Group (Sia Partners), Salamandra, Akkeri Inc., Aclairo Pharmaceutical Development Group, Inc., Biologics Consulting Group, Inc., among others.
b. The market growth is driven by growing global drug development landscape, increasing drug discovery and development activities by pharmaceutical & biotechnology companies. In addition, increased demand for expert regulatory guidance during the preclinical stages and stringent regulatory requirements from authorities such as the U.S. FDA and the European Medicines Agency, which require comprehensive safety and pharmacology data before the clinical trials, are other key driving factors contributing to the market growth.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










