- Home
- »
- Pharmaceuticals
- »
-
North America Active Pharmaceutical Ingredients Market 2033GVR Report cover
![North America Active Pharmaceutical Ingredients Market Size, Share & Trends Report]()
North America Active Pharmaceutical Ingredients Market (2025 - 2033) Size, Share & Trends Analysis Report By Type Of Synthesis (Biotech, Synthetic), By Type Of Manufacturer, By Type, By Application (Oncology, Endocrinology, Pulmonology), By Country, And Segment Forecasts
- Report ID: GVR-4-68040-221-1
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
North America Active Pharmaceutical Ingredients Market Summary
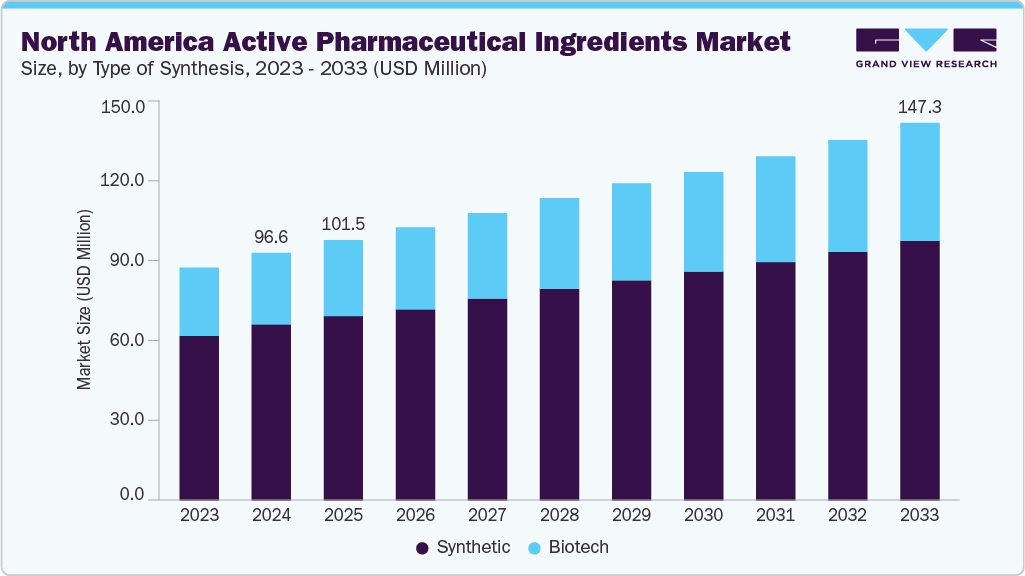
The North America active pharmaceutical ingredients market was estimated at USD 96.64 million in 2024 and is projected to reach USD 147.34 million by 2033, growing at a CAGR of 4.76% from 2025 to 2033. The increasing demand for generic drugs, advancements in biotechnology, a rising aging population, and favorable regulatory support drive this growth.
Key Market Trends & Insights
- By type of synthesis, the synthetic API segment holds the largest revenue share of 71.1% in 2024.
- By type of manufacturer, the captive APIs hold the largest revenue share of 50.40% in 2024.
- By type, the innovative APIs segment holds the largest revenue share of 52.46% and is expected to be the fastest-growing segment from 2024 to 2033.
Market Size & Forecast
- 2024 Market Size: USD 96.64 Million
- 2033 Projected Market Size: USD 147.34 Million
- CAGR (2025-2033): 4.76%
The presence of established pharmaceutical companies with strong manufacturing capabilities further supports the market’s expansion. The North America API market is undergoing a significant transformation, driven by various dynamic factors, including regulatory initiatives, technological advancements, and strategic investments to bolster domestic manufacturing capabilities. One of the most prominent developments in this market is the U.S. government's efforts to ensure a more resilient pharmaceutical supply chain. In August 2025, President Donald Trump signed an executive order to establish the Strategic Active Pharmaceutical Ingredients Reserve (SAPIR) to stockpile critical drug components necessary for national health and security. This strategic initiative, supported by the U.S. Department of Health and Human Services, highlights the growing recognition of the vulnerabilities in the global API supply chain, especially following the disruptions caused by the COVID-19 pandemic. This move is intended to reduce dependence on foreign suppliers, ensuring that essential medicines are readily available during emergencies and disruptions.
Simultaneously, major pharmaceutical companies have announced significant investments in domestic API manufacturing. In February 2025, Eli Lilly, one of the largest pharmaceutical companies in North America, revealed plans to invest USD 27 billion to construct four new manufacturing facilities in the United States, with three of these facilities dedicated to API production. The purpose of this expansion is twofold: to reduce reliance on foreign sources of APIs and to mitigate the impact of potential pharmaceutical import tariffs. This strategic shift reflects broader trends in the pharmaceutical industry, where companies are increasingly bringing production back to North America, driven by factors such as trade uncertainties and rising costs of overseas manufacturing. As part of this initiative, Eli Lilly also confirmed in August 2025 that it would sell a New Jersey plant, focusing on consolidating its manufacturing operations to align with its U.S.-centric production strategy. Such large-scale investments are expected to impact the market significantly, increasing local production capacity and enhancing supply chain resilience.
Alongside these developments, regulatory changes are shaping the landscape of API manufacturing in North America. In June 2024, the U.S. Food and Drug Administration (FDA) amended 21 CFR Part 211, enhancing Current Good Manufacturing Practices (CGMP) for drug products, including APIs. These revisions aim to improve quality assurance and manufacturing processes across the industry, ensuring that APIs meet the highest safety and efficacy standards. Such regulatory updates push pharmaceutical manufacturers to adopt state-of-the-art technologies and quality control measures. Furthermore, in January 2024, the FDA released new draft guidance clarifying that the human drug CGMP guidelines (ICH Q7) now apply to manufacturing veterinary APIs. This move reflects the FDA's commitment to enhancing oversight and aligning practices across various pharmaceutical industry segments. By expanding regulatory oversight to include veterinary APIs, the FDA ensures that APIs used in human and animal health meet the same rigorous standards, thereby improving the overall safety and effectiveness of medications.
In addition to regulatory and investment-driven changes, technological advancements are pivotal in shaping the API manufacturing sector. A significant trend is the shift toward continuous manufacturing, which allows for more efficient, scalable, and cost-effective production of APIs. In May 2023, Cambrex, an API manufacturer, completed a large-scale expansion of its API production plant in Massachusetts, increasing its capacity to meet the growing demand for high-quality, high-potency APIs. This expansion is part of a broader strategy by companies to invest in advanced manufacturing technologies that enhance production efficiency and enable faster response times to changing market demands. Continuous manufacturing, which involves automated, real-time monitoring systems, helps companies reduce production lead times, lower operational costs, and minimize the risk of human error. These technological innovations are expected to be a key factor in sustaining market growth, as they offer greater flexibility and responsiveness to shifts in the demand for pharmaceutical products.
These developments indicate a concerted effort to strengthen North America's domestic API manufacturing sector. By leveraging investments in manufacturing facilities, regulatory updates, and cutting-edge technologies, the region is positioning itself to become more self-reliant and resilient in the face of global disruptions. With increasing demand for generic and innovative drugs, the North American API market is set to continue evolving, driven by these factors contributing to a more robust and future-proof pharmaceutical supply chain. The industry's focus on enhancing manufacturing capacity, improving regulatory compliance, and adopting advanced technologies will help ensure North America remains at the forefront of the global API market.
Market Concentration & Characteristics
The North American active pharmaceutical ingredients industry is moderately concentrated, with major players like Eli Lilly, Merck, and Pfizer holding significant market share. The U.S. leads the market, contributing a major revenue share, due to its strong pharmaceutical sector and regulatory environment. Canada is projected to see the highest growth rate, fueled by increasing healthcare needs and government support. Technological advancements and strategic investments are reshaping the competitive landscape.
The North American API market is witnessing a significant degree of innovation, particularly driven by advancements in biotechnology and the growing demand for biologics and biosimilars. In 2024, the FDA approved several new biologic drugs requiring specialized APIs, further enhancing the focus on innovative manufacturing processes. Companies increasingly invest in continuous manufacturing and automated systems to improve efficiency and scalability. The biotech sector, in particular, is expected to experience rapid innovation, with advances in gene therapies and monoclonal antibodies being key drivers.
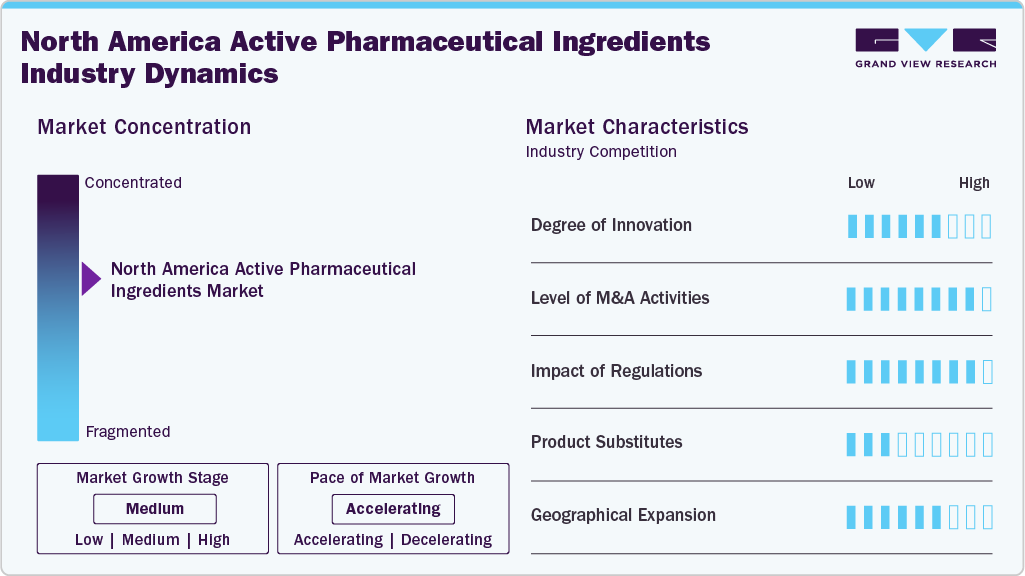
Barriers to entry in the North American API market include high capital investment requirements for manufacturing facilities, stringent regulatory compliance, and intellectual property challenges. The cost of setting up state-of-the-art facilities and maintaining compliance with Good Manufacturing Practices (GMP) standards is considerable. Moreover, established players hold strong patent protections, making it difficult for new entrants to compete in certain segments. These barriers are further exacerbated by the growing consolidation trend among large pharmaceutical companies, reducing opportunities for smaller players to enter the market.
Regulations have a significant impact on the North American API market. The FDA's stringent regulatory framework ensures that APIs meet high-quality and safety standards. In 2024, the FDA amended 21 CFR Part 211, enhancing Current Good Manufacturing Practices (CGMP) for API production, which is expected to lead to higher operational costs but improved product quality. In addition, the FDA's focus on biotechnology and biosimilars pushes API manufacturers to invest in specialized production technologies. These regulations ensure that only high-quality, safe APIs enter the market, but they also increase the cost of compliance and entry barriers.
The availability of product substitutes in the North American API market, particularly for biologics and complex molecules, is relatively low in certain therapeutic segments. However, substitutes are more common in the generic API segment, driven by the high availability of generic versions of off-patent drugs. For instance, when a blockbuster drug’s patent expires, multiple generic manufacturers can offer similar APIs, driving down prices and increasing competition. While alternatives to small-molecule drugs exist, there are limited substitutes for biologics due to the complexity of their manufacturing processes, making the market less competitive in these areas.
Type of Synthesis Insights
Synthetic API segment holds the largest revenue share of 71.1% in 2024, primarily due to its cost-effectiveness and established manufacturing processes. Synthetic APIs, primarily small molecules, account for over 72% of the market due to their widespread use in treating chronic diseases. The segment benefits from efficient manufacturing techniques and scalability. In April 2025, Merck announced a USD 1 billion investment in a new Delaware plant to produce biologic drugs and Keytruda, its leading cancer treatment. This facility is projected to be fully operational by 2028. Furthermore, the widespread adoption of generics, which heavily relies on synthetic APIs, continues to drive growth in this segment.
The biotech API segment is the fastest-growing in 2024, driven by the increasing demand for biologics and biosimilars. These biologic drugs require specialized APIs and are used in treating complex diseases such as cancer and autoimmune disorders. In April 2024, the FDA approved several new monoclonal antibodies, highlighting the rising importance of biotech APIs. The market growth is also supported by advancements in gene therapies, which rely on biotech APIs for production. Furthermore, in February 2024, Amgen announced a USD 1.5 billion expansion of its biologics manufacturing capacity, underscoring the growing demand for biotech-derived APIs.
Type of Manufacturer Insights
Captive APIs hold the largest revenue share of 50.40% in 2024, driven by pharmaceutical companies' preference for self-sourcing APIs for proprietary drugs. Captive manufacturing ensures control over quality, supply chain reliability, and cost efficiency. Large pharmaceutical companies such as Pfizer and Merck continue to invest in captive manufacturing to support their branded drug portfolios. in July 2024, Pfizer opened a $1 billion Active Pharmaceutical Ingredient manufacturing facility in Tuas Biomedical Park, Singapore. This facility is designed to produce small-molecule APIs for Pfizer's oncology, pain, and antibiotic medications, catering to global markets.
The merchant APIs segment is the fastest-growing in 2024, driven by the increasing demand for generics and outsourcing trends. Merchant manufacturers, which supply APIs to multiple clients, benefit from the rise of generic drug production, as companies seek cost-effective solutions. In October 2023, Cambrex completed a $38 million capacity expansion at its small-molecule API manufacturing facility in High Point, North Carolina. This expansion doubled the facility's manufacturing capacity, adding state-of-the-art analytical and chemical development laboratories, two new clinical manufacturing suites, and a small-scale commercial manufacturing operation with three work centers and 2,000-liter reactors. This expansion underscores the sector's rapid growth, driven by the need for outsourced API production and the rising popularity of generics.
Type Insights
The innovative APIs segment holds the largest revenue share of 52.46% and is expected to be the fastest-growing segment from 2024 to 2033. The rising demand for advanced treatments for complex diseases, such as cancer, autoimmune disorders, and genetic conditions, drives this growth. As pharmaceutical companies increasingly focus on biologics, biosimilars, and personalized medicine, the demand for specialized APIs grows. In 2024, the FDA approved over 50 innovative drugs, many relying on cutting-edge APIs, such as Eli Lilly's launch of a novel thyroid cancer treatment in March 2024, further reinforcing the segment's rapid growth.
The generic APIs segment is expected to be the fastest-growing in the North American Active Pharmaceutical Ingredients Market from 2025 to 2033, driven by the rising demand for cost-effective medicines following the patent expirations of several blockbuster drugs. Increasing healthcare expenditures and strong emphasis on affordable treatment options are accelerating the uptake of generics across the United States and Canada. In addition, supportive regulatory frameworks by the U.S. FDA and Health Canada for faster generic approvals, coupled with the growing prevalence of chronic conditions, are boosting production and adoption of generic APIs. Strategic collaborations between pharmaceutical companies and contract manufacturing organizations (CMOs) further fuel this segment's growth.
Application Insights
Cardiology holds the largest revenue share of 21.15% in 2024, driven by the high prevalence of cardiovascular diseases (CVDs) and the increasing demand for treatments. CVDs remain the leading cause of death in North America, which drives substantial demand for APIs used in heart disease treatments. According to the American Heart Association's 2024 Heart Disease and Stroke Statistics Update, approximately 48.6% of U.S. adults had some form of cardiovascular disease (CVD) between 2017 and 2020. This includes conditions such as coronary heart disease, heart failure, stroke, and high blood pressure. Given the U.S. adult population was estimated at over 260 million in 2020, this translates to more than 126 million adults living with CVD. Yes, your statement is accurate. In July 2025, the U.S. Food and Drug Administration (FDA) approved finerenone (Kerendia) for the treatment of adults with heart failure and a left ventricular ejection fraction (LVEF) of 40% or higher, including both mildly reduced (HFmrEF) and preserved (HFpEF) ejection fraction. This further strengthens the cardiology API segment’s dominance.
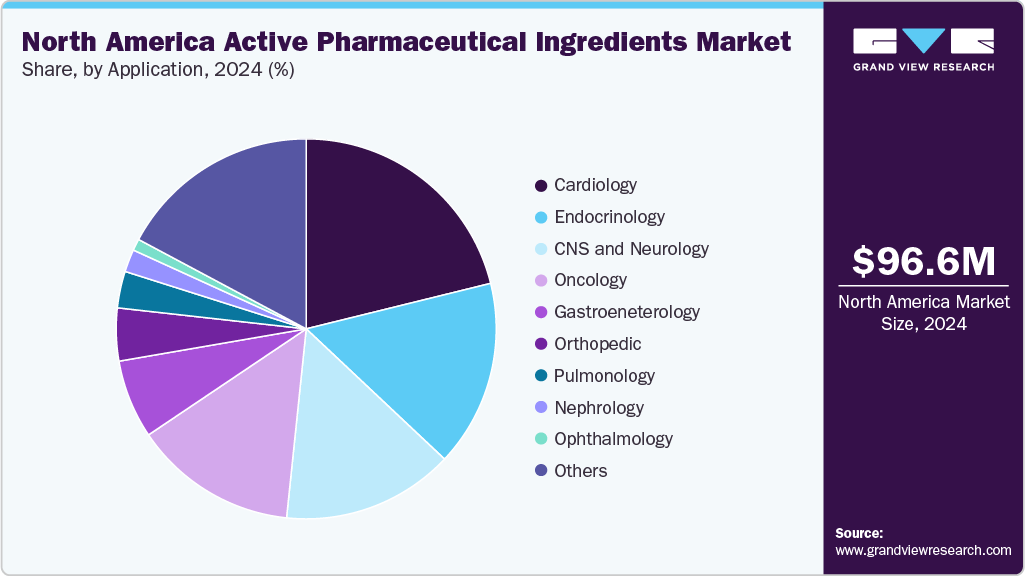
Oncology is expected to be the fastest-growing segment from 2025 to 2033, driven by the increasing cancer incidence and the growing demand for innovative cancer treatments. Cancer remains one of the leading causes of death, with an estimated 2 million new cancer cases in the U.S. in 2024. In June 2025, the U.S. Food and Drug Administration (FDA) approved Datroway™ (datopotamab deruxtecan-dlnk), an antibody-drug conjugate (ADC) targeting the TROP2 protein, for the treatment of adult patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) harboring epidermal growth factor receptor (EGFR) mutations, who have previously received EGFR-directed therapy and platinum-based chemotherapy. This further contributes to the segment's rapid growth. Moreover, advancements in personalized medicine, focusing on tailored treatments for specific cancer types, will continue to fuel the expansion of the oncology API segment.
Country Insights
The U.S. active pharmaceutical ingredients market is witnessing substantial growth, driven by several key factors, including increasing demand for innovative drugs, advancements in biotechnology, and a strong shift towards domestic manufacturing. One of the significant initiatives supporting this shift is the U.S. government's commitment to securing a resilient pharmaceutical supply chain. In August 2025, President Donald Trump signed an executive order establishing the Strategic Active Pharmaceutical Ingredients Reserve (SAPIR) to stockpile critical API components to ensure national health and security. This move reflects growing concerns about the vulnerabilities of relying on foreign sources for essential medicines, especially following the disruptions caused by the COVID-19 pandemic.
In addition to SAPIR, the U.S. pharmaceutical sector has seen substantial investments from major pharmaceutical companies in domestic manufacturing. For example, Eli Lilly announced plans in February 2025 to invest $27 billion in building four new manufacturing plants in the U.S., three of which will be dedicated to API production. This investment aims to reduce reliance on overseas suppliers and address the challenges posed by potential trade tariffs and supply chain disruptions. This trend highlights the pharmaceutical industry’s focus on ensuring a more secure and reliable supply of APIs by bolstering local production capabilities. As pharmaceutical companies continue to innovate and bring new drug treatments to market, the demand for synthetic and innovative APIs is expected to increase, further driving the growth of domestic API manufacturing.
Moreover, advancements in biotechnology are playing a crucial role in expanding the demand for specialized APIs. The rapid growth in biologics, biosimilars, and personalized medicines drives the need for more complex and higher-quality APIs. In March 2024, the U.S. Food and Drug Administration (FDA) approved Elahere (mirvetuximab soravtansine-gynx), a novel immunotherapy for the treatment of folate receptor α (FRα)-positive, platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer in patients who have previously been treated with at least one line of chemotherapy. With the continued focus on personalized medicine, U.S. pharmaceutical companies are expected to increase their investments in R&D and manufacturing infrastructure, further supporting market.
Canada Active Pharmaceutical Ingredients Market Trends
The active pharmaceutical ingredients market in Canada is showing robust growth, driven by the rising demand for generic and biologic drugs and government policies to foster domestic production capabilities. The Canadian pharmaceutical market relies heavily on imports for its API supply, but there has been a noticeable shift towards local manufacturing in recent years. The Canadian government has actively promoted policies to support local production, reduce import reliance, and strengthen the country’s pharmaceutical supply chain.
The government has incentivized companies to invest in domestic API manufacturing that is in line with these goals. A notable example is the expansion of Eurofins' API development laboratory in Ontario in January 2023. This expansion is part of a broader effort to increase local production capacity, improve the efficiency of API manufacturing, and support the growing demand for biologic drugs. The Canadian pharmaceutical industry has seen a rising demand for APIs used in biologics production, as these drugs are becoming increasingly important in treating chronic diseases and cancer.
Moreover, the aging population and increasing prevalence of chronic diseases in Canada are further fueling the demand for pharmaceutical products. This demographic trend is driving the need for more treatments, particularly for conditions such as cardiovascular diseases, diabetes, and cancer, which in turn increases the demand for APIs. As the healthcare system continues to face pressures from an aging population, the demand for more innovative and accessible treatment options is expected to grow, pushing for stronger domestic API production capabilities. This growing need for specialized and high-quality APIs will continue to shape Canada's API market, supporting the government's push for self-sufficiency and supply chain security in the pharmaceutical sector.
Mexico Active Pharmaceutical Ingredients Market Trends
The active pharmaceutical ingredients market in Mexico is experiencing steady growth, largely driven by its strategic position as a hub for pharmaceutical manufacturing and a growing demand for generic and over-the-counter (OTC) drugs. The country has become an attractive location for global pharmaceutical companies due to its proximity to the United States and established trade agreements such as the United States-Mexico-Canada Agreement (USMCA). Mexico's API manufacturing sector benefits from this trade relationship, allowing for easier access to the U.S. and Canadian markets, fostering the expansion of local API production facilities.
In recent years, Mexico has seen a notable increase in investments to enhance domestic API manufacturing capacity. A key development came in October 2023 when the Mexican government announced a strategic initiative to bolster the country’s pharmaceutical manufacturing infrastructure, which includes offering tax incentives and grants to pharmaceutical companies investing in local API production. This initiative aims to reduce Mexico’s dependency on API imports, which have traditionally accounted for a significant portion of the market, particularly for high-demand APIs used in generics. As a result, several major pharmaceutical companies have expanded their operations in Mexico, including multinational firms such as Novartis and Sanofi, which have established or upgraded their production facilities to cater to domestic and international markets.
Moreover, Mexico’s expanding healthcare infrastructure and rising demand for affordable healthcare are contributing to the growth of the API market. With an increasing population, a growing middle class, and a rising incidence of chronic diseases such as diabetes, cardiovascular disease, and cancer, the demand for pharmaceuticals continues to rise. This trend is reflected in the growing demand for APIs, particularly for generics, which offer cost-effective alternatives to branded drugs. The Mexican government’s focus on improving access to healthcare is expected to further stimulate the demand for APIs as part of its broader strategy to strengthen the healthcare system and improve treatment options for its citizens.
In terms of regulatory updates, Mexico's Federal Commission for the Protection against Sanitary Risks (COFEPRIS) plays a pivotal role in overseeing the quality and safety of APIs produced within the country. COFEPRIS has implemented stringent regulations to ensure that locally produced APIs meet international standards, vital for their acceptance in global markets. For instance, in March 2024, Mexico's regulatory authority, the Federal Commission for the Protection Against Health Risks (COFEPRIS), implemented significant updates to its Good Manufacturing Practice (GMP) guidelines These efforts to improve regulatory oversight help position Mexico as a reliable producer of high-quality APIs for both the domestic and international markets, ensuring its competitive standing in the global API landscape.
Key North America Active Pharmaceutical Ingredients Company Insights
Dr. Reddy’s Laboratories Ltd., Sun Pharmaceutical Industries Ltd., Teva Pharmaceutical Industries Ltd., Cipla Inc., AbbVie Inc., Aurobindo Pharma, Sandoz International GmbH (Novartis AG), Viatris Inc., Fresenius Kabi AG, STADA Arzneimittel AG, Lonza, Curia, Pfizer Inc., Bristol-Myers Squibb Company, Merck KGaA, and Catalent, Inc. are some of the key companies operating in the North America API market. A strong pipeline of innovative products is expected to positively influence market expansion over the coming years. In addition, rising outsourcing of API manufacturing due to high production costs, stringent regulatory standards, and increasing demand for cost-efficient solutions are projected to intensify competitive rivalry and shape market dynamics during the forecast period.
Key North America Active Pharmaceutical Ingredients Companies:
- Dr. Reddy’s Laboratories Ltd.
- Sun Pharmaceutical Industries Ltd.
- Teva Pharmaceutical Industries Ltd.
- Cipla Inc.
- AbbVie Inc.
- Aurobindo Pharma
- Sandoz International GmbH (Novartis AG)
- Viatris Inc.
- Fresenius Kabi AG
- STADA Arzneimittel AG
- Lonza
- Curia
- Pfizer Inc.
- Bristol-Myers Squibb Company
- Merck KGaA
- Catalent, Inc.
Recent Developments
-
On August 7, 2025, the U.S. Food and Drug Administration (FDA) introduced the FDA PreCheck program to strengthen the domestic pharmaceutical supply chain. This program aims to facilitate and accelerate the development of U.S.-based drug manufacturing facilities by streamlining regulatory processes and removing unnecessary requirements. It consists of two phases: the first emphasizes increased communication with the FDA during facility planning, design, and construction, while the second supports early engagement on manufacturing and quality control processes.
-
In August 2025, Kodak announced a strategic shift towards pharmaceutical manufacturing, focusing on producing active pharmaceutical ingredients. The company operates an FDA-registered facility producing phosphate-buffered saline and plans to expand into more complex medical products like injectable IV saline. This move aligns with the broader industry trend of reshoring pharmaceutical production to enhance domestic supply chain resilience.
-
On March 20, 2025, Mexico's Federal Commission for the Protection Against Health Risks (COFEPRIS) revised its GMP guidelines to align with international regulatory standards and enhance global recognition. These updates are designed to optimize compliance processes, reduce administrative burdens, and strengthen the efficiency and competitiveness of Mexico's pharmaceutical sector in the global market.
-
In February 2025, Eli Lilly announced plans to invest $27 billion in expanding its U.S. manufacturing capabilities, including constructing four new sites. Three sites will focus on producing active pharmaceutical ingredients, while the fourth will concentrate on future injectable therapies. This investment is part of the company's broader strategy to bolster domestic production and reduce reliance on overseas suppliers.
-
In July 2024, four Mexican pharmaceutical companies announced plans to invest over USD 712 million in drug development and manufacturing. This investment aims to enhance domestic production capabilities and reduce reliance on imported APIs. The initiative reflects the industry's commitment to improving Mexico's pharmaceutical self-sufficiency.
North American Active Pharmaceutical Ingredients Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 101.54 million
Revenue forecast in 2033
USD 147.34 million
Growth rate
CAGR of 4.76% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Type of synthesis, type of manufacturer, type, application, country
Country scope
U.S.; Canada; Mexico
Key companies profiled
Dr. Reddy’s Laboratories Ltd., Sun Pharmaceutical Industries Ltd.; Teva Pharmaceutical Industries Ltd.; Cipla Inc.; AbbVie Inc.; Aurobindo Pharma; Sandoz International GmbH (Novartis AG); Viatris Inc.; Fresenius Kabi AG; STADA Arzneimittel AG, Lonza; Curia; Pfizer Inc.; Bristol-Myers Squibb Company; Merck KGaA; Catalent, Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
North America Active Pharmaceutical Ingredients Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the North America active pharmaceutical ingredients market report based on type of synthesis, type of manufacturer, type, application, and country:
-
Type of Synthesis Outlook (Revenue, USD Million, 2021 - 2033)
-
Biotech
-
Biotech APIs Market, By Type
-
Generic API
-
Innovative API
-
-
Biotech APIs Market, By Product
-
Monoclonal Antibodies
-
Hormones
-
Cytokines
-
Recombinant Proteins
-
Therapeutic Enzymes
-
Vaccines
-
Blood Factors
-
-
-
Synthetic
-
Synthetic APIs Market, By Type
-
Generic API
-
Innovative API
-
-
-
-
Type of Manufacturer Outlook (Revenue, USD Million, 2021 - 2033)
-
Captive APIs
-
Merchant APIs
-
Merchant APIs Market, By Type
-
Generic API
-
Innovative API
-
-
Merchant APIs Market, By Type of Synthesis
-
Biotech
-
Synthetic
-
-
-
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Generic API
-
Innovative API
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Cardiology
-
Oncology
-
CNS and Neurology
-
Orthopedic
-
Endocrinology
-
Pulmonology
-
Gastroenterology
-
Nephrology
-
Ophthalmology
-
Others
-
Frequently Asked Questions About This Report
b. The North America active pharmaceutical ingredients market was estimated at USD 96.64 million in 2024 and is expected to reach USD 101.54 billion in 2025.
b. The North America active pharmaceutical ingredients market is projected to grow at a CAGR of 4.76% from 2025 to 2033 to reach USD 147.34 million by 2033.
b. Based on type of synthesis, synthetic API segment dominated the market with the largest revenue share of 71.1% in 2024, due to its cost-effectiveness and established manufacturing processes.
b. Key players operating in the North America active pharmaceutical ingredients market include Dr. Reddy’s Laboratories Ltd., Sun Pharmaceutical Industries Ltd., Teva Pharmaceutical Industries Ltd., Cipla Inc, AbbVie Inc., Aurobindo Pharma, Sandoz International GmbH (Novartis AG), Viatris Inc., Fresenius Kabi AG, STADA Arzneimittel AG, Lonza, Curia, Pfizer Inc., Bristol-Myers Squibb Company, Merck KGaA, Catalent, Inc.
b. Key factors that are driving market growth include the increasing demand for generic drugs, advancements in biotechnology, a rising aging population, and favorable regulatory support.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










