- Home
- »
- Pharmaceuticals
- »
-
Phenylketonuria Treatment Market, Industry Report, 2030GVR Report cover
![Phenylketonuria Treatment Market Size, Share & Trends Report]()
Phenylketonuria Treatment Market (2025 - 2030) Size, Share & Trends Analysis Report By Drug (Kuvan, Palynziq), By Mode of Administration (Oral Administration, Parenteral Administration), By Region, And Segment Forecasts
- Report ID: GVR-3-68038-310-2
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2018 - 2023
- Forecast Period: 2025 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Phenylketonuria Treatment Market Summary
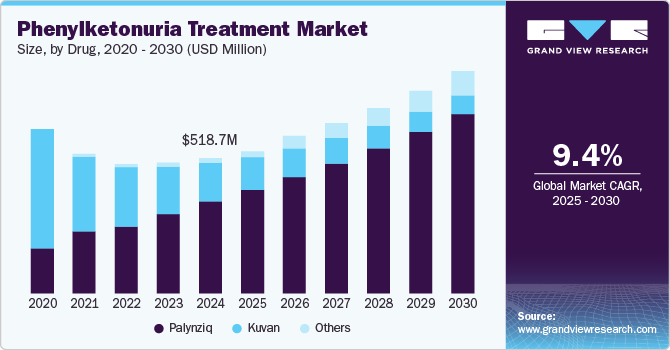
The global phenylketonuria treatment market size was estimated at USD 518.7 million in 2024 and is projected to reach USD 851.0 million by 2030, growing at a CAGR of 9.4% from 2025 to 2030. The market is driven by the increasing prevalence of phenylketonuria and rising healthcare expenditures that boost treatment accessibility.
Key Market Trends & Insights
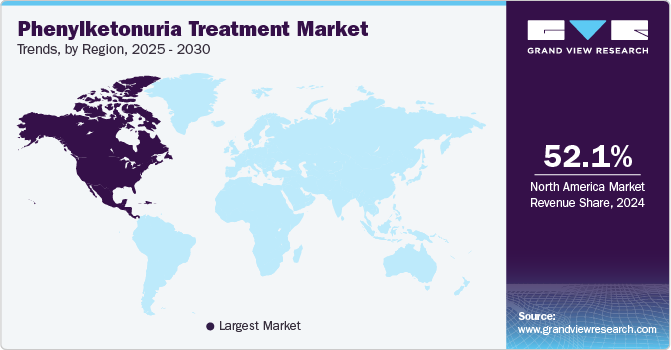
- The North America phenylketonuria treatment market held the largest share of 52.1% in 2024.
- The Asia Pacific phenylketonuria treatment market is anticipated to grow at a CAGR of 10.8% during the forecast period.
- Based on drug, the palynziq accounted for the largest share of 67.9% in 2024 and is also expected to grow at a significant CAGR over the forecast period.
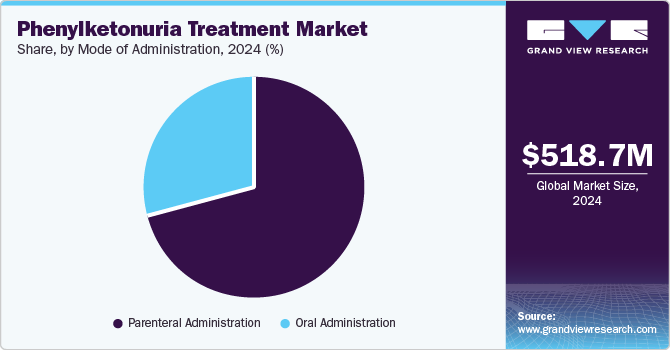
- Based on mode of administration, the parenteral administration segment held the largest market share in 2024 and is also expected to grow at the fastest CAGR over the forecast period.
Market Size & Forecast
- 2024 Market Size: USD 518.7 Million
- 2030 Projected Market Size: USD 851.0 Million
- CAGR (2025-2030): 9.4%
- North America: Largest market in 2024
It is further supported by pipeline drugs and growing collaborations among key players to enhance research and development efforts.
Rising healthcare expenditures propel the phenylketonuria treatment market by enabling increased access to advanced therapies and improved patient care. Higher investments in healthcare facilitate research, development, and the availability of specialized treatments. For instance, in 2022, U.S. health spending grew by 4.1% to USD 4.5 trillion, matching pre-pandemic growth rates. Pandemic-related government spending fell in 2021, but medical goods and services usage rebounded. Health expenditure was 17.3% of GDP, aligning closely with pre-pandemic levels after spikes in 2020 and 2021.
Pipeline drugs and growing collaborations among key players drive the phenylketonuria treatment market by accelerating research and development efforts. These partnerships foster innovation and expedite the introduction of new and effective treatment options. For instance, in March 2024, Eton Pharmaceuticals acquired the U.S. rights to PKU GOLIKE from Relief Therapeutics to treat phenylketonuria (PKU), enhancing its metabolic product line. The acquisition includes taste-masked, extended-release formulas, and rights to future formulations for related conditions. Eton expects this move to boost earnings, with peak annual sales projected to exceed USD 10 million.
Drug Insights
Palynziq accounted for the largest share of 67.9% in 2024 and is also expected to grow at a significant CAGR over the forecast period. Advancements in targeted therapies such as Palynziq that effectively control blood phenylalanine levels are driving the segment's growth. For instance, in May 2023, PTC Therapeutics, Inc. announced a significant development in rare disease treatments, specifically for Phenylketonuria (PKU), a genetic disorder affecting phenylalanine metabolism. The company presented clinical trial results for its investigational drug, PTC923, showing positive outcomes for patients with PKU. These results were promising advancements in PKU treatment, offering a potential oral therapy that could reduce the need for restrictive dietary management, which was the standard of care for PKU patients.
Palynziq can effectively reduce blood phenylalanine levels and maintain long-term control in patients with PKU who have not responded adequately to other treatments. According to the European Medicines Agency (EMA), Palynziq is a phenylketonuria (PKU) treatment that helps break down phenylalanine, a harmful amino acid, using a PEGylated bacterial enzyme called pegvaliase. It has more effect than a placebo in controlling blood phenylalanine levels in patients whose condition was not well-managed with other treatments. Long-term use of Palynziq effectively keeps phenylalanine levels under control for most patients over 18 months.
Mode of Administration Insights
The parenteral administration segment held the largest market share in 2024 and is also expected to grow at the fastest CAGR over the forecast period, attributed to the need for rapid and complete drug absorption to achieve precise therapeutic levels, bypassing gastrointestinal variability and enhancing treatment efficacy. According to the National Center for Biotechnology Information (NCBI), parenteral drugs, especially when given intravenously, have 100% bioavailability, meaning the full dose reaches systemic circulation. This route allows faster absorption and onset of action compared to oral medications, as the drug bypasses the gastrointestinal tract. As a result, parenteral drugs often have a stronger effect due to different metabolic processes.
The need for rapid, reliable therapeutic effects and limited effective oral treatments drives the segment’s growth. Emerging gene therapies that require parenteral delivery offer long-term solutions. Positive regulatory approvals and reimbursement support enhance market growth. These factors collectively fuel the demand for parenteral routes in managing PKU.
Regional Insights
North America phenylketonuria treatment market held the largest share of 52.1% in 2024 owing to the rising diagnoses through enhanced newborn screening programs, advances in treatment options such as enzyme replacement and gene therapies, and government initiatives supporting research and early detection. In addition, growing awareness, improvements in genetic testing, and a shift toward personalized medicine contribute to market growth.
The well-established healthcare infrastructure and funding options facilitate patient treatment access, boosting market expansion. For instance, in June 2023, Synlogic launched a Phase III trial, Synpheny-3, to assess the safety and efficacy of SYNB1934 for treating phenylketonuria (PKU). The study was expected to be conducted at 30 sites across several countries, including the U.S., Germany, and Israel. It involves a multi-part design to evaluate the drug's effects on plasma phenylalanine levels in PKU patients.
U.S. Phenylketonuria Treatment Market Trends
The U.S. phenylketonuria treatment market held a dominant position in 2024 due to the development of innovative therapies that enable better management of phenylalanine levels and support diet liberalization for PKU patients. For instance, in October 2024, PTC Therapeutics, Inc. announced that the FDA accepted the New Drug Application (NDA) for sepiapterin to treat phenylketonuria (PKU) in adult and pediatric patients. The NDA is supported by positive Phase 3 trial results, showing significant reductions in phenylalanine (Phe) levels. The sapropterin may allow diet liberalization for PKU patients by enabling higher protein intake while maintaining Phe control.
Europe Phenylketonuria Treatment Market Trends
The Europe phenylketonuria treatment market was identified as a lucrative region in 2024. European governments provide funding and implement policies to support rare disease treatments such as PKU, enhancing access to therapies and driving innovation. Technological advancements, such as enzyme replacement and gene therapies, improving treatment options and patient outcomes. The growing interest in rare disease treatments, particularly in the orphan drug market, encourages pharmaceutical companies to develop more effective therapies for PKU. For instance, in October 2023, POA Pharma announced its expansion into Austria with the launch of its PKU Easy Microtabs Plus, a treatment for Phenylketonuria (PKU). Following its previous product launches in Germany, the company broadens its European presence in the inborn metabolic disease (IMD) sector. The expansion aims further to enhance the availability of treatments for PKU in Europe.
The UK phenylketonuria treatment market is expected to grow rapidly in the coming years due to the authorities' initiatives to increase patients' access to advanced treatments and ensure equitable care across regions. For instance, in January 2024, the Alliance Party in Northern Ireland welcomed the Department of Health's decision to provide access to Kuvan (sapropterin) for the treatment of PKU patients over 21.
The phenylketonuria treatment market in Germany held a substantial market share in 2024 due to the increasing availability of innovative and convenient treatment options that address unmet patient needs and enhance disease management. For instance, in September 2021, Relief Therapeutics launched a treatment for Phenylketonuria (PKU), the PKU GoLike Krunch, in Germany and Italy. This product was designed to help manage the PKU metabolic disorder by offering a convenient nutritional supplement to address unmet needs in the PKU patient community.
Asia Pacific Phenylketonuria Treatment Market Trends
The Asia Pacific phenylketonuria treatment market is anticipated to grow at a CAGR of 10.8% during the forecast period, attributed to the advancements in diagnostic technologies, enabling early detection and treatment, and proactive government initiatives with healthcare policies that enhance newborn screening and accessibility.
In addition, growing healthcare investments in key countries such as Japan, China, India, and South Korea support better diagnostics and patient care options, boosting market growth. For instance, the decline in Out-of-Pocket expenditure from 64.2% to 39.4% of total health expenditure (2013-2022) indicated a reduced financial burden on individuals. Government health expenditure's share in GDP rose from 1.13% to 1.84%, showing increased public health investment. The rise in government health expenditure's share from 29.0% to 48.0% of total health expenditure highlights enhanced public funding in healthcare.
The Japan phenylketonuria treatment market is expected to grow rapidly in the coming years due to driven by advancements in medical research, including innovative therapies such as enzyme replacement and gene therapy. Government support and funding for rare disease research enhances treatment accessibility. Expanded neonatal screening and awareness programs lead to early diagnosis and better PKU management—collaborations between pharmaceutical companies and research institutions further boost treatment development and commercialization.
The phenylketonuria treatment market in Australia held a substantial market share in 2024 owing to the nationwide newborn screening program and the emphasis on lifelong adherence to a strict, low-protein diet to ensure normal development in diagnosed individuals. For instance, people in Australia are tested for phenylketonuria (PKU) through newborn screening to detect the disorder early, which allows for immediate intervention, such as adopting a strict low-protein diet, which prevents brain damage and developmental issues associated with untreated PKU. This early diagnosis and lifelong dietary management help individuals develop normally, supporting overall health and avoiding severe outcomes such as intellectual disability. This approach benefits Australia by reducing the burden of medical complications and enhancing the quality of life for those diagnosed.
Key Phenylketonuria Treatment Company Insights
Some of the key companies in the phenylketonuria treatment market include BioMarin, Synlogic, Travere Therapeutics, Inc., and others. Organizations are focusing on increasing the customer base to gain a competitive edge in the industry. Therefore, key players are taking several strategic initiatives, such as mergers and acquisitions and partnerships with other major companies.
-
BioMarin is a biotechnology company specializing in developing and commercializing innovative treatments for rare genetic diseases such as Duchenne muscular dystrophy (DMD), achondroplasia, phenylketonuria (PKU), and hemophilia A. The company is focused on breakthrough therapies for serious conditions and has a portfolio that includes clinical and preclinical products.
-
Synlogic is a clinical-stage company developing living medicines for rare and genetic metabolic diseases, including SYNB1618 for phenylketonuria, using its synthetic biology and microbiome platform. It collaborates with pharmaceutical partners to create innovative treatments for various conditions, including liver diseases and cancer.
Key Phenylketonuria Treatment Companies:
The following are the leading companies in the phenylketonuria treatment market. These companies collectively hold the largest market share and dictate industry trends.
- BioMarin
- Synlogic
- Travere Therapeutics, Inc.
- DAIICHI SANKYO COMPANY, LIMITED.
- Codexis, Inc.
- SOM BIOTECH
- Homology Medicines, Inc. (Q32 Bio Inc.)
Recent Developments
-
In August 2024, Otsuka Holdings Co., Ltd. agreed to acquire Jnana Therapeutics Inc., a biotechnology company known for its investigational phenylketonuria (PKU) drug, in a deal valued at up to USD 800 million. This strategic move aims to enhance Otsuka's portfolio of rare metabolic diseases. The acquisition is expected to accelerate the development of Jnana's innovative small-molecule therapeutics targeting solute carrier (SLC) metabolite transporters.
-
In March 2024, Q32 Bio Inc. completed its merger with Homology Medicines, forming a combined company focused on immune system-targeted biologic therapeutics. The merger includes USD 42 million in private placement and around USD 130 million in cash to fund operations through mid-2026. The company's clinical focus involves Phase 2 trials for treatments targeting atopic dermatitis, alopecia areata, and complement system regulation.
Phenylketonuria Treatment Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 543.8 million
Revenue forecast in 2030
USD 851.0 million
Growth rate
CAGR of 9.4% from 2025 to 2030
Base year for estimation
2024
Historical data
2018 - 2023
Forecast period
2025 - 2030
Report updated
November 2024
Quantitative units
Revenue in USD million and CAGR from 2025 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Drug, mode of administration, region
Regional scope
North America, Europe, Asia Pacific, Latin America, MEA
Country scope
U.S., Canada, Mexico, UK, Germany, France, Italy, Spain, Denmark, Sweden, Norway, Japan, China, India, Australia, South Korea, Thailand, Brazil, Argentina, South Africa, Saudi Arabia, UAE, Kuwait
Key companies profiled
BioMarin; Synlogic; Travere Therapeutics, Inc.; DAIICHI SANKYO COMPANY, LIMITED.; Codexis, Inc.; SOM BIOTECH; Homology Medicines, Inc. (Q32 Bio Inc.)
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Phenylketonuria Treatment Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global phenylketonuria treatment market report based on drug, mode of administration, and region.
-
Drug Outlook (Revenue, USD Million, 2018 - 2030)
-
Kuvan
-
Palynziq
-
Others
-
-
Mode of Administration Outlook (Revenue, USD Million, 2018 - 2030)
-
Oral Administration
-
Parenteral Administration
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










