- Home
- »
- Healthcare
- »
-
Point-of-Care Influenza Diagnostics Market Size Report, 2033GVR Report cover
![Point-of-Care Influenza Diagnostics Market Size, Share & Trends Report]()
Point-of-Care Influenza Diagnostics Market (2025 - 2033) Size, Share & Trends Analysis Report By Test Type (Rapid Antigen Tests, Molecular Point-of-Care Tests), By Influenza Strain (Influenza A, Influenza B), By Technology, By End-use (Hospitals, Clinics, Homecare Settings), By Region, And Segment Forecasts
- Report ID: GVR-4-68040-785-8
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Research
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Point-of-Care Influenza Diagnostics Market Summary
The global point-of-care influenza diagnostics market size was estimated at USD 415.56 million in 2024 and is projected to reach USD 709.52 million by 2033, growing at a CAGR of 6.14% from 2025 to 2033. Increasing global prevalence and severity of seasonal & zoonotic influenza outbreaks, growth in adoption of rapid POC tests, technological advancements in the influenza POC tests, and telehealth & home-based testing integration are key drivers of market growth.
Key Market Trends & Insights
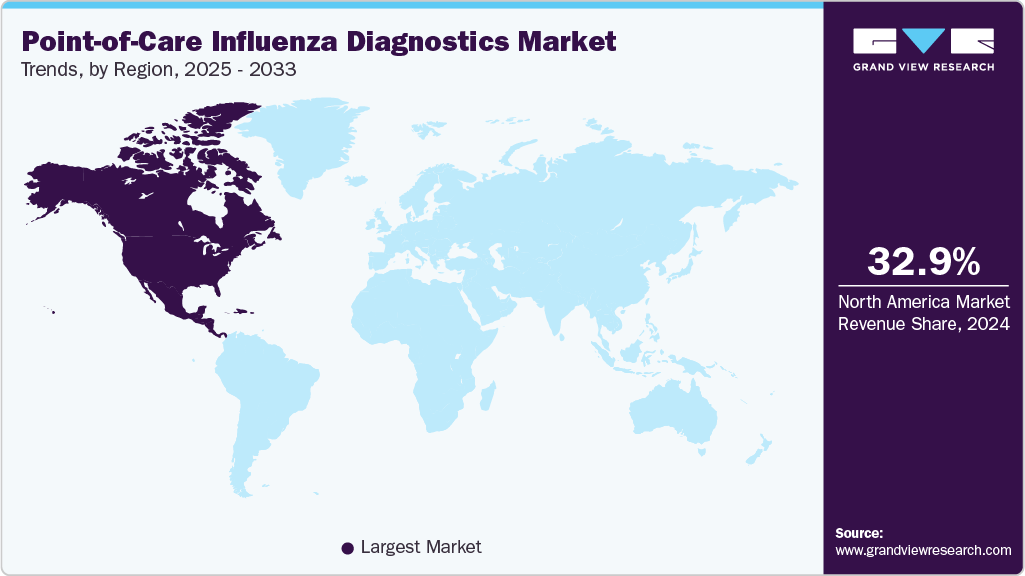
- North America point-of-care influenza diagnostics market dominated the global market and accounted for the largest revenue share of 32.90% in 2024.
- The U.S. led the North American market, holding the largest revenue share in 2024.
- Based on test type, the rapid antigen tests (RIDTs) segment dominated the global market, and molecular point-of-care tests accounted for the largest revenue share of 46.49% in 2024.
- Based on influenza strain, the influenza A tests segment held the largest revenue share of 48.25% in 2024.
- Based on technology, the immunoassay-based technologies segment held the largest revenue share of 42.74% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 415.56 Million
- 2033 Projected Market Size: USD 709.52 Million
- CAGR (2025-2033): 6.14%
- North America: Largest Market in 2024
For instance, in October 2023, the International Federation of Red Cross and Red Crescent Societies reported around a billion seasonal flu cases annually, including 3-5 million cases of severe illness, causing 290,000 to 650,000 respiratory deaths annually. Furthermore, in June 2024, F. Hoffmann-La Roche (Switzerland) received approval from the U.S. Food and Drug Administration (FDA) (Emergency Use Authorization (EUA)) for its cobas Influenza A/B & RSV nucleic acid test, an Influenza A multiplex real-time polymerase chain reaction (RT-PCR) assay on the cobas liat system. Producing results in just 20 minutes on a compact analyzer suitable for most healthcare settings, the test uses either a single nasopharyngeal or anterior nasal swab sample to confirm or rule out infection with influenza A and B viruses.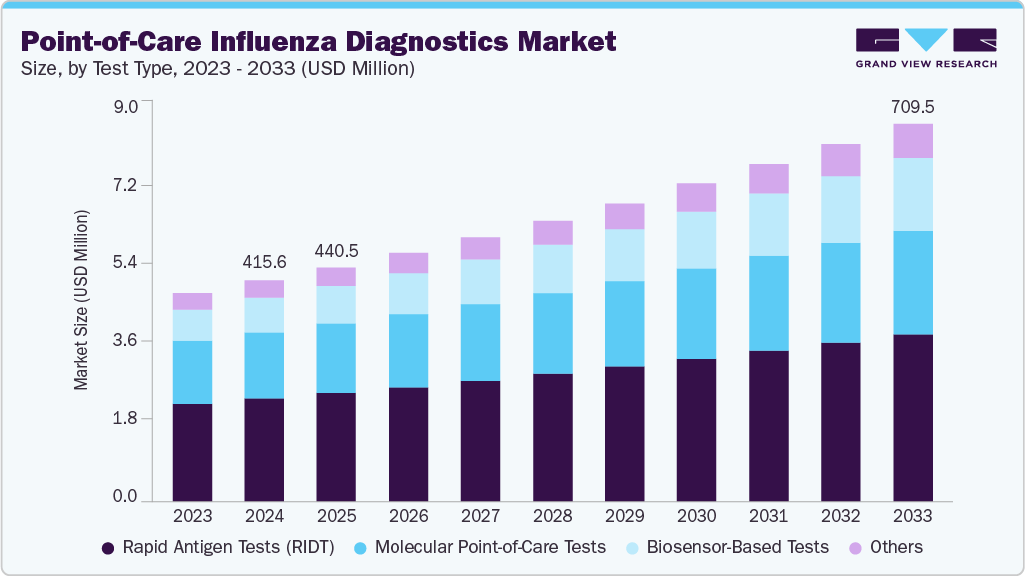
The rapid adoption of point-of-care (POC) tests is a significant factor fostering market growth. These rapid tests provide prompt results, often within minutes, allowing for timely diagnosis and treatment decisions for outpatient and urgent care settings. The convenience and ease of use of rapid influenza diagnostic tests (RIDTs) reduce reliance on centralized laboratories, reduce turnaround time, and improve patient outcomes. Furthermore, these tests are becoming more affordable and accessible, allowing their use in various healthcare settings, such as clinics, pharmacies, and emergency rooms. This shift towards decentralized testing is reinforced by ongoing technological advancements that improve the sensitivity and specificity of rapid tests. Therefore, healthcare providers are more likely to use POC testing as part of routine influenza management, driving market growth, particularly in North America and Europe, with growing adoption in Asia-Pacific markets.
Moreover, one revolutionary factor propelling market expansion is the combination of telehealth and home-based influenza testing. FDA-approved home flu test kits, which are frequently used with telehealth platforms that allow virtual consultations and real-time results transmission to healthcare professionals, have made it possible for people to perform quick diagnostics in the convenience of their own homes. For instance, in October 2024, the Healgen Rapid Check Flu A&B Antigen Test was authorized for sale by the US Food and Drug Administration. The test, available without a prescription and intended for people with respiratory symptoms, uses a nasal swab sample to provide COVID-19 and flu results at home in about 15 minutes. The test can identify proteins from influenza A and B (the viruses that cause the flu).
This integration supports public health initiatives by promoting early detection and isolation, which slows the spread of influenza, and improves patient convenience and testing accessibility. The COVID-19 pandemic made home-based diagnostics easier, raising awareness and acceptance of remote healthcare solutions.

In addition, AI-powered tools integrated with telehealth platforms improve result interpretation and clinical decision support, giving users more confidence in home testing solutions. The convergence of telehealth and home diagnostics is broadening the diagnostic landscape beyond traditional healthcare settings, enabling more widespread influenza surveillance and management, and ultimately driving global market growth.
Market Concentration & Characteristics
The market is highly innovative, as evidenced by the quick advancements in molecular point-of-care (POC) testing, the integration of digital health tools, and the development of rapid antigen tests with improved sensitivity and specificity. For instance, in September 2025, QuidelOrtho Corporation expanded its QUICKVUE portfolio with the QUICKVUE Influenza + SARS Test, a CLIA-waived, 510(k)-cleared rapid, easy-to-use immunoassay designed for professional use in physician office laboratories, urgent care centers, pharmacies, emergency departments, and decentralized hospital labs. Moreover, the key features and benefits include triple detection capability, fast results, professional use, efficient workflow, and affordable access. At the point of care, technologies like CRISPR-based diagnostics and multiplex platforms are expanding the possibilities for influenza detection.
This industry sees moderate mergers and acquisitions, with diagnostic firms purchasing or collaborating with smaller innovators to broaden their test portfolios, increase their molecular capabilities, or penetrate new geographical markets. Strategic activity is consistent even though M&A isn't as aggressive as other biotech sectors. Regulatory frameworks significantly impact the market, particularly for tests used in clinical or over-the-counter settings. FDA approvals, CE marking, and Emergency Use Authorizations (EUAs) significantly impact market entry and adoption.
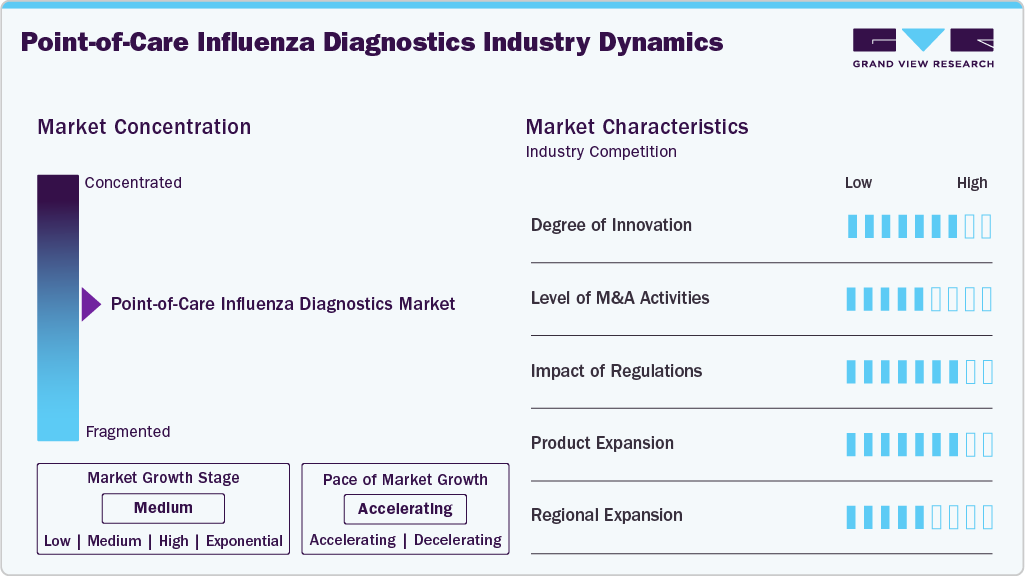
Regulatory scrutiny of test accuracy and clinical utility has escalated since the pandemic. Companies are introducing new POC tests that provide better multiplexing e.g. flu + COVID-19 + RSV, quicker turnaround times, and enhanced usability in decentralized settings like pharmacies and clinics, indicating a strong momentum in product expansion. The region's expansion is moderate. Adoption is leading in North America and parts of Europe, but improved access to healthcare and increased disease awareness are driving growth in Asia-Pacific and Latin America. However, cost sensitivity and infrastructure deficiencies restrict penetration in some lower-income areas.
Test Type Insights
Rapid antigen tests (RIDTs) accounted for the largest market share of 46.49% in 2024, mainly due to their ability to deliver quick results in less than 15 minutes, which is crucial for timely diagnosis and treatment during outbreaks. Their simplicity, ease of use, and affordability in various settings, including hospitals, clinics, and at home, make them highly accessible. Moreover, technological advancements have improved their accuracy and sensitivity, with the introduction of digital and multiplex RIDTs capable of detecting multiple respiratory pathogens simultaneously. For instance, in June 2023, Kaneka launched an influenza virus simultaneous antigen rapid test kit. This diagnostic kit employs immunochromatography*1 to detect COVID-19 and influenza virus antigens in nasopharyngeal and nasal cavity swabs with high accuracy in approximately ten minutes, without requiring specialized testing equipment.
In addition to its ability to handle mutated strains, the new kit is equipped with a "screw-type filter" that prevents liquid leakage while keeping operator safety in mind. Moreover, governmental initiatives and public health programs to enhance early influenza diagnosis and surveillance have boosted demand for RIDTs. The growing availability of rapid test kits and increased awareness of the importance of early detection, particularly in the context of recent pandemics, all contribute to their dominant market position.
Molecular point-of-care tests held the second-largest share in 2024. This is because molecular tests have higher sensitivity and specificity than traditional rapid antigen tests, allowing for more accurate and timely detection of influenza viruses. The growing emphasis on timely diagnosis to control seasonal outbreaks has increased the demand for advanced molecular POCTs. Furthermore, technological advancements have resulted in portable, user-friendly molecular diagnostic devices that can produce lab-quality results in under a minute, making them suitable for use in clinics, emergency departments, and even at home.
Influenza Strain Insights
The influenza A segment accounted for the largest market share of 48.25% in 2024. This is because compared to other strains, influenza A viruses are more common, transmissible, and prone to mutation. Influenza A accounts for most seasonal flu cases and was the primary cause of previous pandemics such as H1N1 and H3N2. Due to its ability to infect multiple species and its propensity for antigenic changes, influenza A poses a serious threat to public health, underscoring the importance of timely and accurate diagnostic testing.
Moreover, influenza A & B combined tests are growing fastest with the rising demand for multiplex tests covering multiple strains. For instance, in October 2025, QuidelOrtho Corporation (U.S.) introduced the QUICKVUE Influenza + SARS Test, a 510(k)-cleared, CLIA-waived rapid immunoassay intended for use in urgent care centers, physician office labs, emergency rooms, pharmacies, and decentralized hospital labs, thereby broadening its point-of-care diagnostic offerings. At the same time, the new test can detect and differentiate influenza A and B antigens from a single patient sample in as little as 10 to 15 minutes. By assisting medical professionals in rapidly distinguishing between the flu and COVID-19, two respiratory diseases that share symptoms, the test promotes quicker treatment choices, better patient outcomes, and less community spread.
Technology Insights
Based on technology, immunoassay-based technologies accounted for the largest market share of 42.74% in 2024. Their cost-effectiveness, quick turnaround time, ease of use, and broad availability are the main reasons for this. In point-of-care settings, immunoassays-like lateral flow assays-are frequently employed to identify influenza antigens with little setup time and expertise. They are perfect for high-throughput screening during the busiest flu seasons because they can produce minute results. Their dominant market position is further supported by the trust that healthcare providers have developed due to their long-standing use in clinical settings.
Furthermore, the immunoassay-based technologies segment is the fastest-growing segment, with a CAGR of 5.64%, due to their rapid turnaround time, ease of use, low cost, and wide availability in point-of-care settings. These tests are especially valuable for quick influenza detection during seasonal outbreaks, allowing timely clinical decisions. The ongoing improvements in test sensitivity and specificity, and growing adoption in both developed and developing regions, contribute to this segment’s accelerated growth.
Moreover, molecular diagnostics is the second largest due to high sensitivity and specificity, the ability to detect low viral loads, and quick turnaround times with lab-quality results. For instance, in December 2021,[7] Eurofins Technologies (Luxembourg) launched a New Multiplex RT-PCR Respiratory Panel for Flu A and Flu B. To improve patient care, clinicians will need access to accurate and reliable tests because the initial symptoms produced by these pathogens are indistinguishable. These new CE-marked multiplex kits can identify and distinguish three distinct viral infections in a single PCR run. The Flu A and B (C1) RT-PCR assay facilitates differentiation in a single reaction between Influenza A, B, and SARS-CoV-2. These technologies are becoming increasingly popular for accurately detecting influenza strains, particularly in critical and outbreak situations where early detection is critical. Advancements in portable molecular platforms, integration with digital health tools, and increased awareness among healthcare providers about the limitations of traditional rapid tests are all driving molecular diagnostics adoption in point-of-care settings.
End-use Insights
Hospitals dominated the market in 2024, accounting for a 38.35% share, due to the greater availability of advanced diagnostic infrastructure, high patient inflow, and the ability to perform rapid and molecular point-of-care tests efficiently. Diagnostic tests are performed because hospitals are frequently thefirst place patients with severe flu symptoms are seen. Moreover, the extensive use of point-of-care influenza diagnostics is further supported by the availability of qualified medical personnel and the requirement for prompt clinical decision-making in hospital settings.
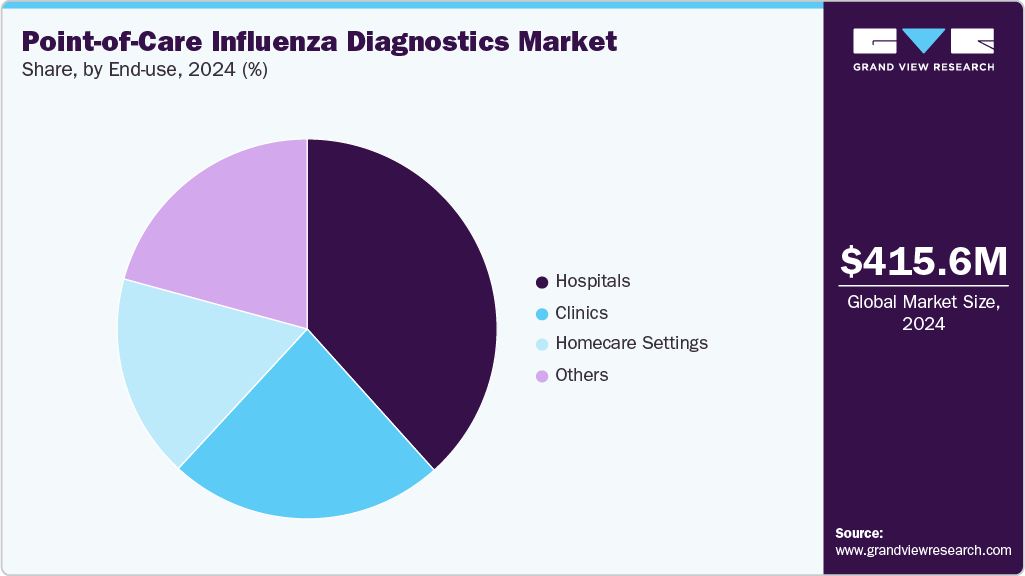
Furthermore, the home care settings segment is driving the market expansion due to a list of factors, including rising demand for convenient and accessible testing solutions, increased availability of over-the-counter (OTC) rapid influenza test kits, growing awareness among consumers about early detection and disease management, and advancements in user-friendly diagnostic technologies suitable for self-use. For instance, in February 2023, the FDA approved the first over-the-counter at-home test to detect both influenza. The Lucira Flu Home Test is a one-time at-home test kit that returns results from self-collected nasal swab samples in about 30 minutes.
Regional Insights
North America point-of-care influenza diagnostics market dominated the global market and accounted for the largest revenue share of 32.90% in 2024. The North America point-of-care influenza diagnostics market is expected to grow steadily due to the high prevalence of seasonal influenza, increased healthcare spending, the dominance of key market players, and widespread adoption of advanced diagnostic technologies. Furthermore, supportive government initiatives for infectious disease surveillance, increased awareness of early disease detection, and the expansion of decentralized healthcare services, such as urgent care centers and home testing, are all driving market growth in the region.
U.S. Point-of-Care Influenza Diagnostics Market Trends
The point-of-care influenza diagnostics market in the U.S. is expected to grow significantly due to a rising seasonal flu, supported by rapid advances in molecular and rapid antigen testing technologies and robust public health initiatives. According to the Centers for Disease Control and Prevention, during the 2024-25 influenza season, clinical laboratories that reported to the CDC tested 3,978,954 respiratory specimens for influenza viruses using clinical diagnostic tests. Of these, 489,579 (12.3%) tested positive, with 434,985 (88.8%) for influenza A and 54,594 (11.2%) for influenza B. The growing demand for early, accurate, and accessible influenza detection in hospitals, clinics, and homes drives advancements in rapid flu diagnostic tests (RIDTs) and RT-PCR assays.
Recent FDA approvals for multiplexed, AI-enabled, and at-home test kits have accelerated and simplified influenza diagnosis, with results often available in minutes. Government programs that improve testing capacity, pandemic preparedness, and outbreak surveillance help to drive market growth by encouraging widespread use of reliable diagnostic tools. Furthermore, the post-COVID healthcare infrastructure and increased public awareness of respiratory infections have bolstered the value of nationwide rapid, point-of-care flu testing, resulting in significant market growth over the forecast period.
Europe Point-of-Care Influenza Diagnostics Market Trends
The point-of-care influenza diagnostics market in Europe is experiencing notable growth driven by the increasing prevalence of influenza and the rising adoption of advanced diagnostic technologies. Key trends include a growing preference for point-of-care testing in hospitals and at home, which is aided by technological advancements such as molecular diagnostics and lateral flow assays (LFA). Germany leads the market due to its large aging population and high prevalence of infectious diseases. Furthermore, the growing healthcare infrastructure and healthcare professionals' preference for point-of-care tests over traditional lab tests propel market growth. Furthermore, the blood sample segment is expected to grow the fastest due to the variety of tests available. Overall, the European market is moving toward faster, more accessible, and user-friendly flu diagnostic tests suitable for a variety of healthcare settings.
The UK point-of-care influenza diagnostics market is steadily increasing due to several factors, including rising flu prevalence and the increasing adoption of rapid and accurate diagnostic technologies. For instance, as per the UK Health Security Agency, overall activity was higher than in the 2023 to 2024 season. Compared to the 2022 to 2023 season, the peak was similar in intensity and timing, but the 2024 to 2025 season had a slower overall decline due to the late influenza B activity.
Moreover, the market growth is fueled by the convenience and speed of point-of-care testing, which allows for early diagnosis and timely treatment, reducing disease spread and healthcare burden. Furthermore, government initiatives and public health programs to improve influenza surveillance and vaccination propel market growth. AI integration and multiplex molecular assays are examples of innovations that will enhance diagnostic accuracy and efficiency. Rising awareness among healthcare providers and patients about the importance of early flu detection and the increasing availability of FDA and EMA-approved rapid influenza diagnostic tests contribute to market growth in the UK.
The point-of-care influenza diagnostics market in Germany is expanding rapidly, owing to factors such as rising influenza cases, increased adoption of rapid diagnostic technologies, and favorable government policies. Key drivers include the country's strong diagnostic manufacturing capabilities, a large aging population more susceptible to influenza, and increased awareness of infectious diseases. Furthermore, technological advancements such as AI integration for faster and more accurate result interpretation and an increasing preference for point-of-care testing in outpatient clinics and home care settings are driving market growth. The growing accessibility of healthcare facilities and ongoing investments in pandemic preparedness support the market growth.
Asia Pacific Point-of-Care Influenza Diagnostics Market Trends
The point-of-care influenza diagnostics market in the Asia Pacific is expanding rapidly, driven primarily by the growing demand for rapid and accurate diagnostic results in resource-constrained and remote settings. The market benefits from technological advancements like rapid molecular diagnostics and lateral flow assays, which improve the speed, accuracy, and accessibility of influenza testing. Government initiatives to improve healthcare infrastructure and increase healthcare spending in the region have accelerated market growth. Moreover, countries such as China, India, Japan, and South Korea play important roles in manufacturing and innovation, owing to strong industrial bases and rising demand for decentralized testing. Furthermore, rising awareness of influenza and infectious diseases and the integration of AI and Industry 4.0 technologies in diagnostics contribute to the promising growth trajectory of the Asia Pacific point-of-care influenza diagnostics.
Japan point-of-care influenza diagnostics market is growing steadily, driven by multiple factors including a rising incidence of influenza outbreaks, increasing demand for rapid and accurate testing, and a growing elderly population that is more vulnerable to severe flu complications. Public health initiatives and government-sponsored vaccination campaigns raise awareness and encourage early testing, accelerating market growth. Rapid antigen diagnostic tests (RIDTs), molecular assays like RT-PCR, and multiplex testing have all contributed to faster, more accurate, and convenient influenza diagnosis. For instance, in September 2025, Japan expanded flu testing capabilities. The influenza diagnostics market focuses on tools and technologies for rapidly detecting influenza viruses (A, B, and subtypes). The Influenza Diagnostics Market focuses on tools and technologies for quickly detecting influenza viruses (A, B, and their subtypes). These diagnostics are critical for early disease management, outbreak control, and guiding antiviral treatment plans.
The point-of-care influenza diagnostics market in China is rapidly expanding as respiratory infections rise, and rapid diagnostic technologies are more widely used. The market was valued in hundreds of millions of dollars in 2024 and is expected to grow rapidly over the next decade, owing to increased public and healthcare provider awareness of influenza testing. Technological advancements, such as AI integration for faster and error-free sample processing and increased government investment in healthcare infrastructure, drive market growth.
Latin America Point-of-Care Influenza Diagnostics Market Trends
The point-of-care influenza diagnostics market in Latin America is experiencing gradual but robust growth, driven by increasing healthcare awareness and rising prevalence of influenza diseases. Moreover, key factors boosting the increased adoption of rapid and cost-effective diagnostic technologies in hospitals, assisted living, and homecare facilities, coupled with government initiatives aimed at disease control and improved healthcare infrastructure. Also, the growing geriatric population, which is more vulnerable to infections, increases demand for easily accessible diagnostic testing. Brazil currently leads the regional market in revenue share, thanks to healthcare reforms and increased healthcare spending, followed by Mexico, which has significant growth potential.
Middle East and Africa Point-of-Care Influenza Diagnostics Market Trends
The point-of-care influenza diagnostics market in the MEA is steadily expanding. The rising prevalence of influenza, as well as the growing demand for rapid, accessible, and dependable diagnostic solutions in remote and underserved areas, are major drivers. Saudi Arabia and the UAE dominate the market due to expanded healthcare infrastructure and government initiatives to improve diagnostic access. Moreover, the user-friendly testing devices, adoption of portable devices, and incorporation of digital health technologies such as artificial intelligence (AI) contribute to consistent market growth. The region's emphasis on improving disease surveillance and early detection strategies continues to support this upward trend.
Key Point-of-Care Influenza Diagnostics Company Insights
The market features several key players driving innovation and adoption. Leading companies include Abbott, Becton Dickinson (BD), Danaher/Cepheid, Quidel Corporation, Qiagen, F. Hoffmann-La Roche Ltd, Bio-Rad, BioMerieux (Biofire/Idaho Technologies), Diasorin (Focus Diagnostics), Hologic, Thermo/Remel, and Fujirebio. These firms are heavily investing and rapidly evolving with continuous product innovation, geographic expansion, and strategic collaboration.
Key Point-of-Care Influenza Diagnostics Companies:
The following are the leading companies in the point-of-care influenza diagnostics market. These companies collectively hold the largest market share and dictate industry trends.
- Abbott
- Becton Dickinson (BD)
- Danaher/Cepheid
- Quidel Corporation
- Qiagen
- F. Hoffmann-La Roche Ltd
- Bio-Rad
- BioMerieux (Biofire/Idaho Technologies)
- Diasorin (Focus Diagnostics)
- Hologic
- Thermo/Remel
- Fujirebio
- Visby Medical
- Twist
Recent Developments
-
In January 2023, Visby Medical received FDA emergency authorization for a respiratory health test in CLIA-waived settings. Moreover, the Visby Medical Respiratory Health Test is a fast, polymerase chain reaction (PCR) device that detects and differentiates between upper respiratory infections caused by influenza (flu) A & B.
-
In December 2022, Diasorin filed a 510(k) premarket notification and CLIA waiver application with the US Food and Drug Administration (FDA) for its LIAISON NES, a next-generation molecular POC testing platform. Furthermore, the submission covers the LIAISON NES FLU A/B, RSV, and COVID-19 panel, which is designed to detect and differentiate the most common and clinically significant respiratory pathogens, including influenza A and influenza B.
Point-of-Care Influenza Diagnostics Market Report Scope
Report Attribute
Details
Market size in 2025
USD 440.47 million
Revenue forecast in 2033
USD 709.52 million
Growth rate
CAGR of 6.14% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Test type, influenza strain, technology, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Norway; Sweden; Denmark; Japan; China; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Abbott; Becton Dickinson (BD); Danaher/Cepheid; Quidel Corporation; Qiagen; F. Hoffmann-La Roche Ltd; Bio-Rad; BioMerieux (Biofire/Idaho Technologies); Diasorin (Focus Diagnostics); Hologic; Thermo/Remel; Fujirebio; Visby Medical; Twist
Customization scope
Free report customization (equivalent up to 8 analysts’ working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Point-of-Care Influenza Diagnostics Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global point-of-care influenza diagnostics market based on test type, influenza strain, technology, end-use, and region:
-
Test Type Outlook (USD Million, 2021 - 2033)
-
Rapid Antigen Tests (RIDT)
-
Molecular Point-of-Care Tests
-
Biosensor-Based Tests
-
Others
-
-
Influenza Strain Outlook (USD Million, 2021 - 2033)
-
Influenza A
-
Influenza B
-
Influenza A & B Combined Tests
-
-
Technology Outlook (USD Million, 2021 - 2033)
-
Immunoassay-based
-
Molecular Diagnostics
-
Nanotechnology based Systems
-
Others
-
-
End-use Outlook (USD Million, 2021 - 2033)
-
Hospitals
-
Clinics
-
Homecare Settings
-
Others
-
-
Regional Outlook (USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global point-of-care influenza diagnostics market size was estimated at USD 415.56 million in 2024 and is expected to reach USD 440.47 million in 2025.
b. The global point-of-care influenza diagnostics market is expected to grow at a compound annual growth rate of 11.54% from 2025 to 2033 to reach USD 709.52 million by 2033.
b. North America dominated the point-of-care influenza diagnostics market with a share of 32.90% in 2024. This is attributable grow steadily due to the high prevalence of seasonal influenza, increased healthcare spending, the dominance of key market players, and widespread adoption of advanced diagnostic technologies.
b. Some key players operating in the point-of-care influenza diagnostics market include Abbott, Becton Dickinson (BD), Danaher/Cepheid, Quidel Corporation Qiagen, F. Hoffmann-La Roche Ltd, Bio-Rad, BioMerieux (Biofire/Idaho Technologies), Diasorin (Focus Diagnostics), Hologic, Thermo/Remel, Fujirebio
b. Key factors that are driving the market growth include factor such as the increasing global prevalence and severity of seasonal & zoonotic influenza outbreaks, growth in adoption of rapid POC tests, technological advancements in the influenza POC tests, and telehealth & home-based testing integration are key drivers of the market's growth.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










