- Home
- »
- Clinical Diagnostics
- »
-
Syphilis Testing Market Size & Share, Industry Report, 2033GVR Report cover
![Syphilis Testing Market Size, Share & Trends Report]()
Syphilis Testing Market (2026 - 2033) Size, Share & Trends Analysis Report By Technology (Molecular Diagnostics, Immunoassay), By Location Of Testing (Laboratory Testing, Point Of Care Testing), By Region (North America, Europe, APAC, Latin America, MEA), And Segment Forecasts
- Report ID: GVR-4-68040-747-4
- Number of Report Pages: 236
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Syphilis Testing Market Summary
The global syphilis testing market size was estimated at USD 1.57 billion in 2025 and is projected to reach USD 2.32 billion by 2033, growing at a CAGR of 5.23% from 2026 to 2033. Key market drivers for dual HIV/syphilis rapid diagnostics include the rising global burden of congenital syphilis and HIV co-infections, which continue to cause significant adverse pregnancy outcomes.
Key Market Trends & Insights
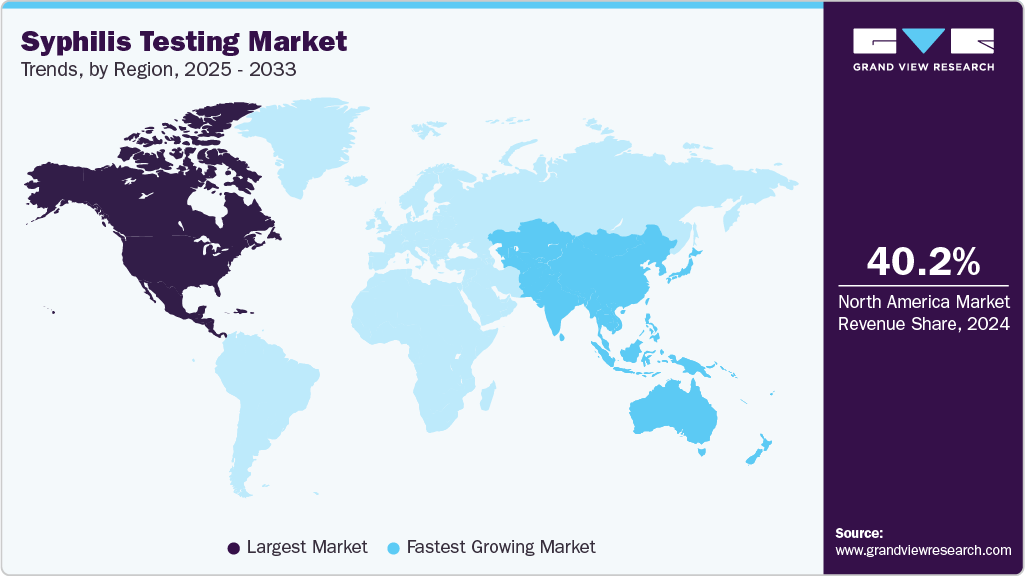
- North America held the dominant position in the global market in 2025, capturing 36.32% of the revenue share.
- The U.S. syphilis testing industry is driven by a sharp epidemiologic rebound-reported cases rose ~80% from 2018-2022-plus major policy and regulatory responses.
- By technology, the immunoassay segment held the largest market share of 71.20% in 2025.
- By location of testing, the laboratory testing segment held the largest market share of 79.56% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 1.57 Billion
- 2033 Projected Market Size: USD 2.32 Billion
- CAGR (2026-2033): 5.23%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Growing emphasis on elimination of mother-to-child transmission (EMTCT) programs by the WHO and national health bodies is creating strong demand for cost-effective, point-of-care solutions. Regulatory approvals, such as Health Canada’s clearance of MedMira’s Multiplo Rapid TP/HIV Test, and partnerships, such as CHAI-MedAccess-SD Biosensor, are accelerating adoption by ensuring affordability and accessibility. Additionally, the push for decentralized testing in rural and underserved regions supports uptake, while technological improvements in accuracy and speed enhance market confidence.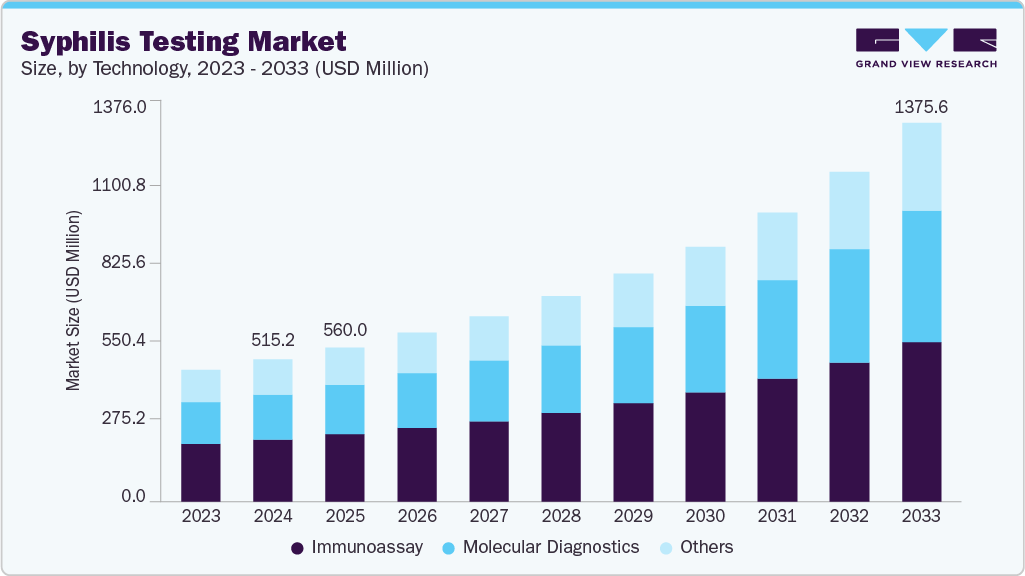
Syphilis, once considered a disease of the past, has reemerged as a significant public health and market concern in North America and globally. While the widespread use of penicillin in the mid-20th century had nearly eliminated syphilis in high-income countries, recent years have seen a sharp resurgence. In the U.S., reported cases increased by more than 80% between 2018 and 2022, rising from approximately 115,000 to over 207,000-the highest level since the 1950s. Of particular concern is congenital syphilis, which grew by 183% during the same period, reflecting gaps in maternal screening and early intervention. In Canada, rising incidence in the Prairie provinces and northern territories-particularly among Indigenous populations further highlights disparities in access to timely diagnostics and treatment. These epidemiological shifts are creating heightened demand for innovative diagnostic solutions, decentralized testing models, and scalable public health interventions, positioning the infectious disease diagnostics market for accelerated growth.
Regulatory Shifts & Diagnostic Innovation in the U.S.
One of the most significant regulatory milestones came in August 2024, when the U.S. Food and Drug Administration (FDA) authorized the First To Know Syphilis Test from NOWDiagnostics, Inc.-the first over-the-counter, at-home syphilis antibody test available in the U.S. The device uses a finger-stick blood sample and provides results in about 15 minutes, delivering a level of accessibility comparable to at-home pregnancy or COVID-19 tests. Although the test cannot distinguish between past and current infection, since it only detects antibodies, the FDA’s decision to approve it through the De Novo pathway was groundbreaking. This regulatory classification not only recognizes the urgent public health need but also lowers barriers for future developers, effectively opening the market to additional consumer-friendly syphilis diagnostics.
The FDA’s action aligns with broader federal strategies. In 2024, the Department of Health and Human Services (HHS) launched the National Syphilis and Congenital Syphilis Syndemic Federal Task Force to coordinate response strategies across federal agencies. Together, these measures represent a new era of urgency, in which regulatory flexibility and innovation are central to tackling an epidemic once thought under control.
NIH Investment in Next-Generation Testing
Complementing regulatory progress, the U.S. National Institutes of Health (NIH) has scaled up investment in syphilis research. In 2024, NIH awarded $2.4 million in grants to institutions, including the University of Washington and Johns Hopkins University, to develop diagnostics that move beyond antibody detection. Projects include mass spectrometry platforms capable of directly identifying syphilis proteins, aptamer-based DNA assays for rapid point-of-care use, and infant-specific diagnostics to address the congenital crisis. In 2025, NIH further expanded this portfolio, awarding $2.7 million to Penn State to support the development of an electrochemical biosensor that can detect both treponemal and non-treponemal antibodies in a single step within 10 minutes. This innovation could collapse the cumbersome two-test algorithm into a one-step process, transforming clinical workflows and enabling real-time, confirmatory diagnosis in both resource-rich and resource-limited settings.
Clinical & Community Strategies
Parallel to research and regulatory activity, implementation models are evolving. A 2024 study at the University of Chicago demonstrated that routine opt-in syphilis screening in emergency departments more than doubled case detection, particularly among asymptomatic patients and pregnant individuals. Such findings underscore how expanding testing criteria in high-throughput clinical environments can serve as critical intervention points.
Meanwhile, community-driven responses are making headway. In the Navajo Nation, mobile outreach teams have pioneered on-site testing and same-day treatment at food pantries, jails, and motels. This approach addresses a key weakness of traditional care pathways, loss to follow-up, and has shown that culturally grounded, flexible models can effectively reach vulnerable populations.
Canadian Advances: MedMira and Indigenous-Led Programs
In Canada, public health officials face many of the same diagnostic and treatment challenges as their U.S. counterparts, but with distinct regional dynamics. Between 2018 and 2024, syphilis incidence surged in the Prairie provinces, particularly Manitoba, Saskatchewan, and Alberta, where Indigenous populations are disproportionately affected. In March 2025, the Canadian Institutes of Health Research (CIHR), in partnership with Indigenous leaders, launched the Ayaangwaamiziwin Centre, an Indigenous-led initiative backed by over $7 million in funding to deliver testing, treatment, and culturally safe care in Prairie and northern communities. This program recognizes that Indigenous leadership and community control are central to effective outbreak response.
On the regulatory side, Health Canada approved MedMira’s Multiplo TP/HIV Rapid Test on 26 December 2024, marking another turning point. The finger-prick device simultaneously detects HIV-1/2 and syphilis antibodies with results in minutes and requires no specialized storage, making it well-suited for clinics, hospitals, and remote communities. A 2020-2022 clinical study in Alberta, co-led by Dr. Sean Rourke (REACH Nexus, St. Michael’s Hospital, Toronto) and Dr. Ameeta Singh (University of Alberta), involving more than 1,500 participants, demonstrated 100% accuracy for HIV and over 98% accuracy for syphilis. Already deployed in Europe and Colombia, the test is being rolled out in Canada in collaboration with REACH Nexus. MedMira’s CEO, Hermes Chan, highlighted its ability to bridge diagnostic gaps at a fraction of the cost of conventional methods. Following this license, Health Canada is expected to authorize the Reveal TP (Syphilis) test in 2025, further strengthening the country’s diagnostic capacity.
Global Context: Dual-Testing and Maternal Health
The global burden of congenital syphilis remains devastating. The infection causes an estimated 355,000 adverse pregnancy outcomes each year, including 210,000 stillbirths and neonatal deaths. In Africa, syphilis accounts for approximately 11% of all stillbirths. The World Health Organization projects that scaling dual HIV/syphilis testing in the 20 highest-burden countries could save 74,000 lives and prevent 53,000 severe illness cases annually. These efforts also align with global strategies to eliminate mother-to-child transmission (EMTCT) of HIV, syphilis, and hepatitis B.
Challenges & the Road Ahead
Despite these advances, critical challenges remain. Diagnostic technologies are still dominated by antibody-based assays, which cannot definitively confirm active infection or determine cure. Treatment depends almost exclusively on Bicillin L-A (benzathine penicillin G), a formulation with limited suppliers and frequent global shortages. No vaccine exists, leaving prevention dependent on timely detection, partner notification, and consistent drug supply. Moreover, public health funding has not kept pace with the epidemic. In the U.S., CDC funding for STD prevention dropped from $169 million in 2003 to about $80 million by 2023, even as cases more than doubled. Canadian public health programs face similar budgetary and access constraints, particularly in rural and northern regions.
Yet the convergence of regulatory approvals, NIH-funded research, Indigenous-led programs, and global partnerships offers cautious optimism. The FDA’s De Novo pathway for at-home testing sets a precedent for consumer-friendly innovations. NIH investments are driving technologies that could provide real-time, confirmatory results and infant-specific Location of Testings. Health Canada’s MedMira approval and CIHR’s Indigenous partnerships show how national strategies can integrate technology with community-led care. Globally, partnerships like CHAI and SD Biosensor demonstrate that equitable access and affordability are achievable at scale.
Market Concentration & Characteristics
Innovation is reshaping dual rapid testing, with companies like MedMira launching the Multiplo Rapid TP/HIV Test, capable of detecting both HIV-1/2 and syphilis antibodies from a single finger-prick with 100% HIV accuracy and 98% syphilis accuracy. Already deployed in Europe and Colombia and recently approved by Health Canada (Dec 2024), such devices offer immediate results without refrigeration. This innovation supports decentralized care, bridging gaps in underserved communities and advancing EMTCT goals.
M&A and collaborations are driving accessibility and affordability. For instance, in November 2021, MedAccess, CHAI, and SD Biosensor formed a partnership to distribute the STANDARD Q HIV/Syphilis Combo Test at under US$1 across 100+ LMICs. While outright acquisitions are moderate, such global collaborations mirror consolidation effects, enabling scale, affordability, and distribution synergies. Larger diagnostics firms are expected to target similar partnerships or acquisitions to strengthen infectious disease testing portfolios.
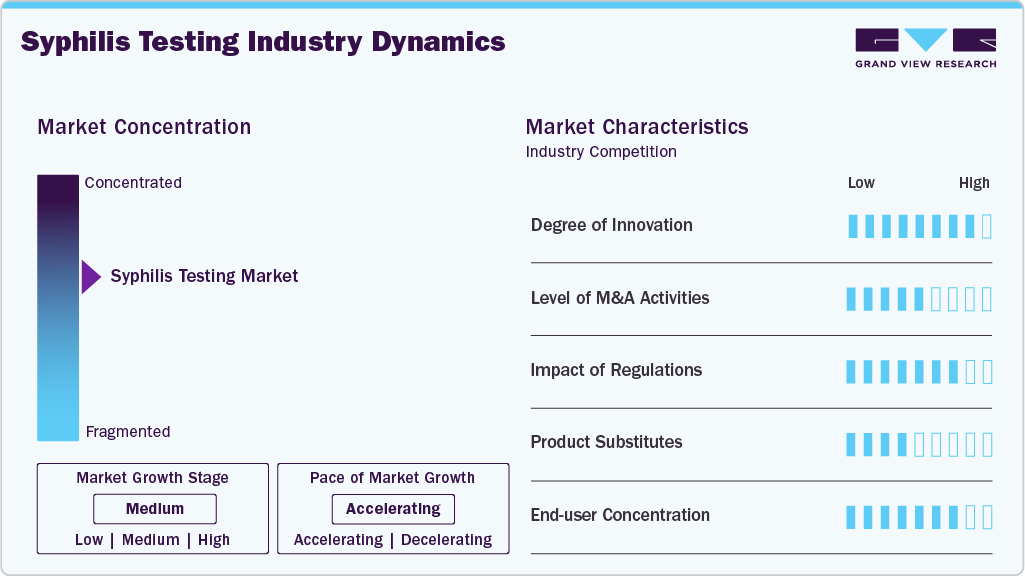
Regulation remains a catalyst, shaping adoption and market entry. Health Canada’s approval (Dec 2024) of MedMira’s dual test followed a 1,500+ subject clinical study in Alberta (2020 - 2022) co-led by top institutions, ensuring credibility. Similarly, the WHO prequalification of SD Biosensor’s STANDARD Q combo test allowed procurement by donor-funded programs in LMICs. Such regulatory endorsements reduce market hesitancy, ensure compliance with global EMTCT strategies, and create strong differentiation for approved products.
Substitution risk exists but remains manageable. Traditional ELISA and PCR assays deliver gold-standard accuracy but are limited by infrastructure needs and slower turnaround. Rapid combo tests, like MedMira’s Multiplo and SD Biosensor’s STANDARD Q, provide point-of-care results in under 20 minutes at a fraction of the cost. However, emerging HIV self-tests and multiplex molecular platforms could act as substitutes. To maintain an edge, rapid test developers emphasize affordability (e.g., 32% price cut via CHAI-MedAccess) and ease of use.
End-users are highly concentrated in public health agencies, NGOs, and maternal health programs. For example, the CHAI-MedAccess-SD Biosensor partnership directly targets antenatal care in 100+ LMICs, aligning with WHO EMTCT goals. In high-income regions, adoption depends on government approvals, as seen with Health Canada’s 2024 clearance enabling rollout in Canadian hospitals and clinics. Indigenous populations in Canada’s Prairie provinces, experiencing disproportionately higher infection rates, highlight the need for targeted community health programs.
Technology Insights
The immunoassay segment currently holds the largest share of 71.20% in 2025. Its dominance stems from widespread clinical adoption, established use in hospital laboratories, cost-effectiveness, and compatibility with high-throughput platforms. Immunoassays, including ELISA and chemiluminescent assays, are considered the gold standard for antibody detection, particularly in confirmatory testing. Their strong regulatory acceptance and extensive integration into public health programs further consolidate market leadership.
Molecular diagnostics, on the other hand, represents the fastest-growing technology segment, driven by rising demand for early, highly sensitive detection of infections. PCR and next-generation sequencing (NGS)-based tools are gaining traction, particularly for monitoring viral load and resistance mutations, and in detecting low-titer infections that may be missed by serology. Growth is accelerated by decentralization trends, falling costs of molecular platforms, and integration into point-of-care and multiplex assays. These attributes make molecular diagnostics critical in addressing gaps left by traditional methods, particularly in mother-to-child transmission screening and high-burden geographies.
Location of Testing Insights
Laboratory testing continues to represent the largest share of 79.56% the syphilis diagnostics market in 2025, owing to its entrenched role as the clinical gold standard. Centralized laboratories and hospital-based diagnostic centers rely heavily on automated immunoassays, ELISAs, and confirmatory treponemal/non-treponemal workflows that ensure high accuracy and consistency. These platforms support large-scale testing volumes, making them particularly valuable in national screening initiatives and prenatal care programs where reliability is paramount.
In the United States, laboratory giants such as Quest Diagnostics and LabCorp have built extensive syphilis testing infrastructure, often bundled with comprehensive STD panels that streamline testing workflows for clinicians. In Canada, providers such as LifeLabs and provincial public health labs dominate, integrating syphilis testing into routine sexual health and antenatal programs. Laboratories also benefit from established reimbursement pathways and integration with electronic medical records (EMRs), reinforcing their central role in healthcare systems.
Despite the growth of decentralized alternatives, laboratories continue to maintain their dominance because of their unmatched ability to handle confirmatory testing, complex case management, and integration with advanced molecular diagnostics. Particularly for high-risk populations and congenital syphilis investigations, laboratory workflows remain essential for providing definitive results.
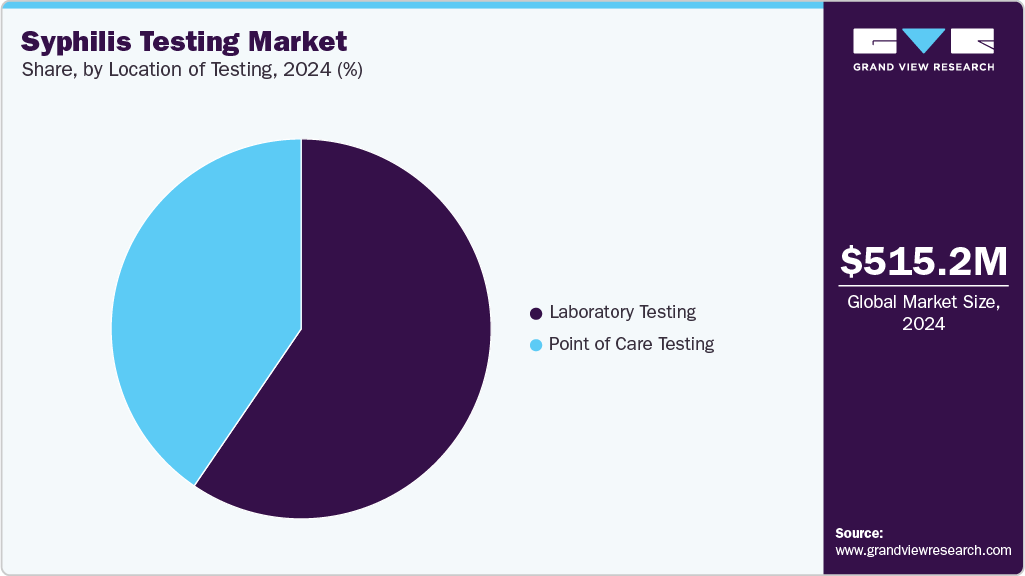
Point-of-care (POC) testing is experiencing the fastest growth, reflecting shifting public health priorities toward accessibility, speed, and patient-centered care. The surge in congenital syphilis, which demands immediate detection and treatment, has highlighted the limitations of lab-centric models that rely on multiple visits and long turnaround times. In response, both regulators and companies are accelerating the adoption of rapid and at-home testing solutions. A landmark example is Health Canada’s approval (December 2024) of MedMira’s Multiplo Rapid TP/HIV Test, a dual-purpose finger-prick device that delivers near-instant results for both HIV and syphilis without special storage requirements. Already in use across Europe and Colombia, the device is now positioned to close diagnostic gaps in both urban hospitals and remote Canadian communities. Similarly, in August 2024, the U.S. FDA authorized NOWDiagnostics’ First To Know Syphilis Test through the De Novo pathway, marking the first over-the-counter syphilis diagnostic available for at-home use. This regulatory milestone not only enables consumer-driven testing but also establishes a precedent for other developers to bring innovative syphilis POC solutions to market.
POC solutions are particularly critical in Indigenous and underserved communities across North America, where access to centralized laboratories is limited and travel times are prohibitive. Mobile outreach programs in regions like the Navajo Nation in the U.S. and Prairie provinces in Canada are leveraging these rapid devices to deliver same-day testing and treatment, reducing loss to follow-up. Globally, WHO-endorsed dual HIV/syphilis combo tests, such as the STANDARD Q (SD Biosensor) supplied under the CHAI-MedAccess partnership, are further demonstrating the role of POC platforms in advancing elimination of mother-to-child transmission (EMTCT) targets. As syphilis rates rise, the market is shifting toward hybrid models, where centralized labs maintain their dominance for confirmatory and large-scale screening, while POC testing grows rapidly as a frontline solution to improve accessibility, equity, and speed of diagnosis.
Regional Insights
The syphilis testing industry in North America held the dominant position in the global market in 2025, capturing 36.32% of the revenue share. The U.S. drives this leadership through high testing volumes at Quest Diagnostics, Labcorp, and public health laboratories, where syphilis is included in comprehensive STD panels. Recent FDA clearances of innovative diagnostics, such as NOWDiagnostics’ at-home syphilis test, are expanding access and creating a consumer-directed market. In Canada, LifeLabs and provincial health authorities emphasize antenatal screening and community outreach programs, particularly in the Prairie provinces. Growing congenital syphilis rates and strong federal funding continue to fuel demand across both laboratory and point-of-care channels.
U.S. Syphilis Testing Market Trends
The U.S. syphilis testing industry is driven by a sharp epidemiologic rebound-reported cases rose ~80% from 2018-2022-plus major policy and regulatory responses. The FDA’s August 2024 De Novo authorization for NOWDiagnostics’ at-home antibody test created an OTC consumer testing channel, while NIH investments ($2.4M in 2024; $2.7M in 2025 grants) are accelerating next-gen molecular and electrochemical POC diagnostics. Implementation models (e.g., University of Chicago ED opt-in screening; Navajo Nation mobile outreach) emphasize decentralized testing and same-day treatment to reduce loss to follow-up. Persistent challenges-Bicillin L-A shortages, declining CDC STD prevention funding, and congenital syphilis spikes-sustain demand for rapid, confirmatory, and field-deployable tests.
Europe Syphilis Testing Market Trends
The syphilis testing industry in Europe is shaped by growing incidence and strong national health systems prioritizing targeted screening. The UK and Germany report steady case increases, prompting expansion of free sexual health services and antenatal programs. Laboratories dominate with automated immunoassays, while CE-marked rapid tests are used in community clinics. Digital integration into public health networks improves surveillance and contact tracing. Procurement remains cost-sensitive, requiring suppliers to deliver value-driven pitches. Future growth is likely through dual HIV/syphilis rapid tests in antenatal care, and enhanced use of digital reporting platforms. Strong regulation and robust reimbursement systems create barriers but ensure market stability.
The UK syphilis testing industry shows modest upticks in syphilis diagnoses (early-stage cases: 9,535 in 2024 vs 9,375 in 2023; overall cases 13,030 vs 12,456). Public programs emphasize free, confidential testing and targeted messaging to high-risk groups (MSM, young adults). Policy momentum-NHS rollout of gonorrhoea vaccination programs-illustrates prevention orientation, while surveillance highlights rising antibiotic resistance in gonorrhoea. Market opportunities center on integrated sexual-health POC devices and digital linkage-to-care solutions. Buyers favor validated rapid dual tests for antenatal screening and community clinics; regulatory acceptance and NHS procurement cycles will determine commercial scale-up.
The syphilis testing industry in Germany mirrors broader EU increases in bacterial STIs; testing and genomic surveillance (WGS) are expanding to track resistant strains. Clinical laboratories remain primary purchasers, using automated immunoassays for screening and molecular techniques for complex cases. Growing public-health focus on MSM and young adult engagement, plus improved surveillance, is creating demand for rapid POC diagnostics in sexual-health clinics and outreach programs. Vendors that combine high sensitivity with integration into municipal public-health reporting and partner-notification workflows will find traction as Germany scales targeted screening and antimicrobial-resistance monitoring.
Asia Pacific Syphilis Testing Market Trends
The syphilis testing industry in the Asia Pacific is projected to show significant growth over the forecast period with a CAGR of 6.62%. Asia-Pacific presents diverse dynamics, with China and Japan leading in laboratory-based immunoassays and antenatal screening, while Southeast Asia emphasizes cost-effective rapid testing. Rising incidence, urbanization, and maternal health programs drive demand. China’s provincial procurement favors high-volume labs and WHO-prequalified dual tests, while Japan expands targeted syphilis clinics but requires rigorous validation for new technologies. In lower-income countries, donor-driven programs supply affordable rapid kits for maternal health. Market opportunities lie in combining rapid testing with digital follow-up, ensuring timely treatment. Strategic partnerships and localized regulatory alignment are critical for success in this fragmented yet high-growth regional market.
The China syphilis testing industry is characterized by expanding laboratory capacity and rapid scale-up of point-of-care screening in urban and provincial health systems. National antenatal screening initiatives and rising STI surveillance drive demand for affordable dual HIV/syphilis rapid assays, lab immunoassays, and molecular confirmatory tests. Manufacturers that offer WHO-aligned, low-cost combo tests and scalable platforms can access procurement through provincial health networks. Challenges include bridging urban-rural access gaps and ensuring confirmatory lab follow-through; opportunities exist for POC integration with telemedicine and maternal health programs.
The syphilis testing industry in Japan is conservative, favoring laboratory immunoassays for confirmatory diagnosis, but authorities are piloting POC expansions to improve early detection in young adults. According to NIH studies, syphilis cases in Japan remain at historically high levels, exceeding 13,000 annually for four consecutive years (2022-2025). After hitting a record high of 14,906 cases in 2023, the number remained elevated with 14,663 cases in 2024. Vendors should prioritize regulatory compliance, clinician education, and demonstration projects tied to prenatal care; culturally sensitive outreach and anonymity-preserving distribution channels will support uptake among younger and stigmatized populations.
Latin America Syphilis Testing Market Trends
The syphilis testing industry in Latin America, led by Brazil, prioritizes syphilis testing within maternal and child health programs due to high congenital syphilis rates. Public procurement dominates, with Ministries of Health sourcing WHO-prequalified dual HIV/syphilis tests for antenatal care. Laboratories remain important for confirmatory testing, but POC diagnostics in community clinics are increasingly adopted to enable same-visit treatment initiation. Brazil’s federal health system purchases at scale, driving strong demand for affordable, reliable supply chains. The region’s price sensitivity favors cost-effective solutions, and partnerships with local distributors and NGOs are essential. Demonstrating improved maternal outcomes is key to sustained growth and procurement renewals.
The Brazil syphilis testing industry has observed the country prioritize public health, emphasize antenatal screening, and reduce congenital syphilis, creating sizable demand for dual HIV/syphilis rapid tests and low-cost POC kits. Regional initiatives and NGO partnerships (mirrored by CHAI/MedAccess/SD Biosensor models) have created price-sensitive procurement channels across LMICs. Large public purchasers (Ministry of Health, regional health secretariats) prioritize WHO-prequalified products that enable on-site testing in primary care and maternity clinics. Vendors that can combine cost competitiveness, supply reliability, and integration with prenatal programs will be well-positioned to scale across Brazil and neighboring markets.
Middle East and Africa Syphilis Testing Market Trends
The syphilis testing industry in the Middle East and Africa expects promising opportunities in the region. For instance, in Africa, syphilis remains a major driver of adverse birth outcomes-~11% of stillbirths are attributable to syphilis-so maternal screening is a key market. The WHO and donor-funded procurement drive uptake of low-cost, WHO-prequalified combo tests (e.g., STANDARD Q). Infrastructure constraints favor POC and rapid combo diagnostics that do not require cold chain or complex lab infrastructure. In the Middle East, markets are heterogeneous: wealthier GCC countries invest in lab and molecular capacity, while low-resource countries rely on NGO and donor programs. Scalability depends on procurement frameworks, donor support, and local capacity-building.
The syphilis testing industry in Saudi Arabia has witnessed the health system emphasize centralized lab capacity and antenatal screening within public hospitals; demand for high-quality immunoassays and molecular confirmatory testing remains strong. At the same time, Gulf Cooperation Council (GCC) initiatives and private hospitals are open to rapid POC solutions for screening travelers, migrant worker clinics, and perinatal care. Market entry requires regulatory clearance, adherence to national procurement, and partnerships with major hospital groups. Opportunities exist for accredited dual HIV/syphilis rapid tests that meet Saudi regulatory standards and can be deployed in maternal health and occupational-health screening programs.
Key Syphilis Testing Company Insights
Market leaders are engaged in extensive R&D to develop technologically advanced, cost-efficient testing products. Besides, several strategies, such as mergers & acquisitions, are being incorporated by various organizations to expand their market presence and are anticipated to create significant growth opportunities over the forecast period.
Key Syphilis Testing Companies:
The following key companies have been profiled for this study on the syphilis testing market.
- Abbott Laboratories
- F. Hoffmann-La Roche Ltd.
- Bio-Rad Laboratories, Inc.
- Siemens Healthineers AG
- Danaher Corporation (Beckman Coulter)
- Thermo Fisher Scientific Inc.
- bioMérieux SA
- OraSure Technologies, Inc.
- Chembio Diagnostics, Inc.
- SD Biosensor, Inc.
Recent Developments
-
Date: August 2024
The U.S. FDA authorized the First To Know Syphilis Test by NOWDiagnostics, Inc., the first over-the-counter, at-home syphilis antibody test. Using a finger-stick blood sample, it delivers results in 15 minutes, similar to pregnancy or COVID-19 home tests. Although it cannot differentiate between past and active infections, its approval via the De Novo pathway reflects regulatory recognition of urgent public health needs. This milestone lowers barriers for consumer-focused diagnostics and sets a precedent for rapid, accessible syphilis testing. Enabling at-home testing expands early detection, reduces transmission, and empowers individuals to seek timely care.
-
NIH-Funded Next-Generation Diagnostics Research
Date: 2024 - 2025
The U.S. National Institutes of Health (NIH) invested heavily in innovative syphilis diagnostics, awarding $2.4 million in 2024 and $2.7 million in 2025. Projects include mass spectrometry platforms for direct detection of syphilis proteins, aptamer-based DNA assays for rapid point-of-care use, and infant-specific diagnostics addressing congenital syphilis. Penn State researchers developed an electrochemical biosensor capable of detecting treponemal and non-treponemal antibodies in 10 minutes, potentially collapsing the traditional two-step testing algorithm into a single step. These investments aim to improve real-time, confirmatory diagnosis, enhance clinical workflows, and address gaps in both resource-rich and resource-limited healthcare settings globally.
-
National Syphilis and Congenital Syphilis Syndemic Task Force
Date: 2024
The U.S. Department of Health and Human Services (HHS) launched the National Syphilis and Congenital Syphilis Syndemic Federal Task Force to coordinate cross-agency strategies for outbreak response. This initiative aligns regulatory flexibility, diagnostic innovation, and public health programming to address surging syphilis rates, including a concerning rise in congenital infections. The task force facilitates policy alignment, data sharing, and resource allocation across federal, state, and local levels. By integrating testing, treatment, and prevention strategies, it strengthens national readiness, targets high-risk populations, and underlines the federal commitment to reversing decades of neglect in syphilis control and maternal-infant health outcomes.
-
Indigenous-Led Health Program - Ayaangwaamiziwin Centre
Date: March 2025
In Canada, the Canadian Institutes of Health Research (CIHR), in partnership with Indigenous leaders, established the Ayaangwaamiziwin Centre, a $7 million Indigenous-led initiative to combat syphilis in Prairie and northern communities. The program delivers culturally safe testing, treatment, and community-based care, addressing disproportionately high infection rates among Indigenous populations. By embedding community leadership and culturally appropriate approaches, it reduces barriers to access, improves follow-up adherence, and enhances trust in healthcare interventions. The initiative integrates modern diagnostics with mobile outreach strategies and local engagement, serving as a model for equitable public health interventions targeting vulnerable populations in both rural and urban settings.
Syphilis Testing Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 1.63 billion
Revenue forecast in 2033
USD 2.32 billion
Growth rate
CAGR of 5.23% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, Number of tests in million, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Technology, location of testing, and region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Sweden; Denmark; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; Saudi Arabia; South Africa; UAE; Kuwait
Key companies profiled
Abbott Laboratories; F. Hoffmann-La Roche Ltd.; Bio-Rad Laboratories, Inc.; Siemens Healthineers AG; Danaher Corporation (Beckman Coulter); Thermo Fisher Scientific Inc.; bioMérieux SA; OraSure Technologies, Inc.; Chembio Diagnostics, Inc.; SD Biosensor, Inc.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Syphilis Testing Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global syphilis testing market report based on technology, location of testing, and region:
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Molecular Diagnostics
-
Immunoassay
-
Others
-
-
Location of Testing Outlook (Revenue, USD Million, 2021 - 2033)
-
Laboratory Testing
-
Commercial/Private labs
-
Public Health Labs
-
-
Point of Care Testing
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. Some key players operating in the syphilis testing market include Abbott Laboratories, F. Hoffmann-La Roche Ltd., Bio-Rad Laboratories, Inc., Siemens Healthineers AG, Danaher Corporation (Beckman Coulter), Thermo Fisher Scientific Inc., bioMérieux SA, OraSure Technologies, Inc., Chembio Diagnostics, Inc., SD Biosensor, Inc.
b. Key market drivers for dual HIV/syphilis rapid diagnostics include the rising global burden of congenital syphilis and HIV co-infections, which continues to cause significant adverse pregnancy outcomes. Growing emphasis on elimination of mother-to-child transmission (EMTCT) programs by WHO and national health bodies is creating strong demand for cost-effective, point-of-care solutions. Regulatory approvals, such as Health Canada’s clearance of MedMira’s Multiplo Rapid TP/HIV Test, and partnerships like CHAI–MedAccess–SD Biosensor, are accelerating adoption by ensuring affordability and accessibility
b. The global syphilis testing market size was estimated at USD 1.57 billion in 2025 and is expected to reach USD 1.63 billion in 2026.
b. The global syphilis testing market is expected to grow at a compound annual growth rate of 5.23% from 2026 to 2033 to reach USD 2.32 billion by 2033.
b. The immunoassay segment currently holds the largest share of 71.20% in 2025. Its dominance stems from widespread clinical adoption, established use in hospital laboratories, cost-effectiveness, and compatibility with high-throughput platforms. Immunoassays, including ELISA and chemiluminescent assays, are considered the gold standard for antibody detection, particularly in confirmatory testing. Their strong regulatory acceptance and extensive integration into public health programs further consolidate market leadership.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










