- Home
- »
- Medical Devices
- »
-
U.S. Antimicrobial Catheters Market, Industry Report, 2033GVR Report cover
![U.S. Antimicrobial Catheters Market Size, Share & Trends Report]()
U.S. Antimicrobial Catheters Market (2026 - 2033) Size, Share & Trends Analysis Report By Product Type (Urological, Vascular Access, Specialty & Surgical), By Technology (Silver-Based Technology, Antibiotic-Impregnated), By Material, By End Use (Hospitals), And Segment Forecasts
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Antimicrobial Catheters Market Summary
The U.S. antimicrobial catheters market size was estimated at USD 506.18 million in 2025 and is projected to reach USD 932.44 million by 2033, growing at a CAGR of 7.94% from 2026 to 2033. The growth of this market is primarily driven by the rising incidence of hospital-acquired infections, the increasing number of catheterization procedures across acute and long-term care settings, and the growing clinical focus on infection prevention and patient safety.
Key Market Trends & Insights
- By product type, the vascular access catheters segment led the market with the largest revenue share in 2025.
- By technology, the silver-based technology segment led the market with the largest revenue share in 2025.
- By material, the polyurethane (PU) segment led the market with the largest revenue share in 2025.
- By end use, the hospitals segment led the market with the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 506.18 Million
- 2033 Projected Market Size: USD 932.44 Million
- CAGR (2026-2033): 7.94%
More than 1 million cardiac catheterization procedures are performed every year in the U.S. In addition, expanding adoption of antimicrobial-coated devices, supportive infection control guidelines, and increasing healthcare investments in advanced medical technologies are further supporting market expansion.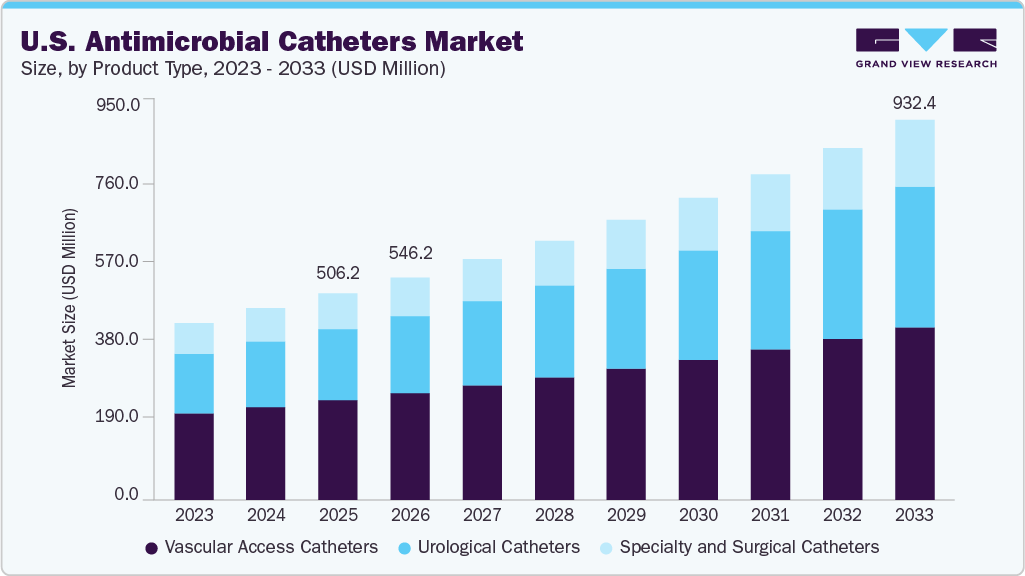
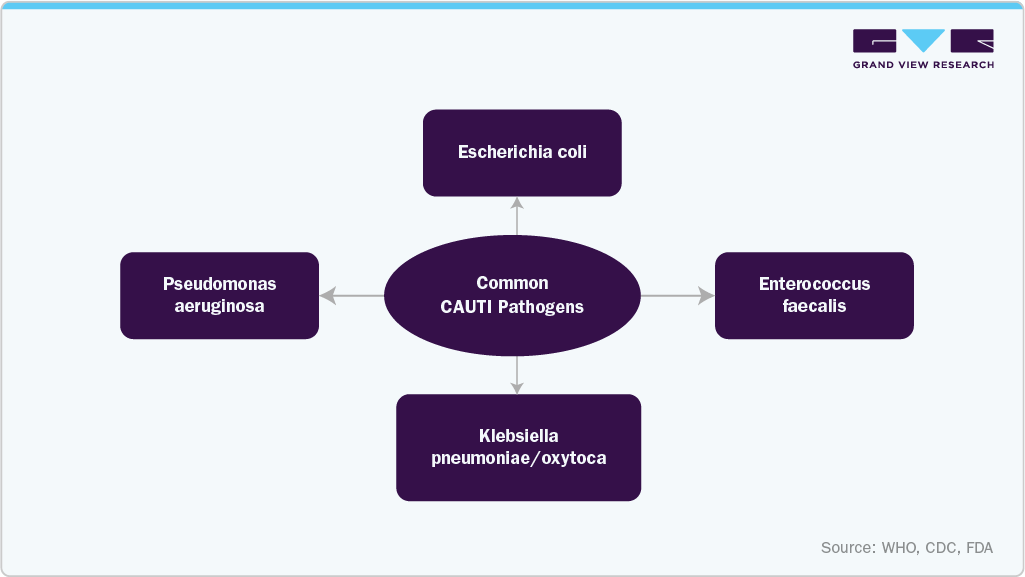
CLABSI (central line-associated bloodstream infections), CAUTI (catheter-associated urinary tract infections), and VAE (ventilator-associated events) are major drivers of the U.S. antimicrobial catheters market. They represent the primary hospital-acquired infection (HAI) burden, increasing clinical and economic pressure on healthcare systems to adopt infection-prevention technologies. High incidence of these device-associated infections leads hospitals to prioritize antimicrobial-coated and drug-eluting catheter technologies to reduce microbial colonization and infection risk. Studies show that antimicrobial-coated catheters can reduce microbial colonization, which directly supports hospital infection reduction programs and procurement shifts toward coated devices. In addition, the broader rise in HAIs has led hospitals to increasingly standardize the use of coated catheters as part of infection control protocols, reinforcing demand for antimicrobial catheter products across ICU, surgical, and long-term care settings.
U.S. Hospital Infection Statistics - CLABSI, CAUTI, and VAE by State and Location, 2024
2024
Central line-associated bloodstream infections (CLABSI)
Catheter-associated urinary tract infections (CAUTI)
Ventilator-associated events (VAE)
Inpatient Locations
Inpatient Locations
Inpatient Locations
State
Total
ICU
Wards
NICU
Total
ICU
Wards
Total
ICU
Wards
Alabama
545
146
375
24
531
146
385
86
69
17
Alaska
59
10
43
6
56
10
46
9
8
1
Arizona
484
100
362
22
468
100
368
43
36
7
Arkansas
347
65
245
37
325
65
260
37
34
3
California
2,895
566
2,069
260
2,667
566
2,101
405
288
117
Colorado
433
80
318
35
408
80
328
73
59
14
Connecticut
Upgrade report license to gain access to the complete analysis.
D.C.
Delaware
Florida
Georgia
Guam
Hawaii
Idaho
Illinois
Indiana
Iowa
Kansas
Kentucky
Louisiana
Maine
Maryland
Massachusetts
Michigan
Minnesota
Mississippi
Missouri
Montana
Nebraska
Nevada
New Hampshire
New Jersey
New Mexico
New York
North Carolina
North Dakota
Ohio
Oklahoma
Oregon
Pennsylvania
Puerto Rico
Rhode Island
South Carolina
South Dakota
Tennessee
Texas
Utah
Vermont
Virgin Islands
Virginia
Washington
West Virginia
Wisconsin
Wyoming
All US
29,837
5,726
22,182
1,929
28,427
5,721
22,706
4,512
3,066
1,446
Source: CDC, 2026
Total hospital admissions in the U.S. drive the antimicrobial catheters market, as higher patient volumes lead to increased use of invasive medical devices such as urinary catheters, central venous catheters, and other indwelling lines that carry infection risk. As admissions rise, especially in critical care, surgical, and long-term care settings, the number of catheterization procedures increases, raising the possibility of hospital-acquired infections. To reduce infection risk, treatment costs, and hospital penalties linked to infection rates, healthcare facilities adopt antimicrobial-coated catheters as part of infection prevention protocols. In addition, higher admission volumes increase procurement demand from hospitals, strengthen bulk purchasing of infection-control devices, and accelerate the shift toward advanced antimicrobial technologies to support patient safety and improve clinical outcomes.
Government infection-control initiatives, such as the HAC Reduction Program and the HAI National Action Plan, are key drivers of the U.S. antimicrobial catheters market, as they create financial, clinical, and operational pressure on hospitals to reduce device-related infections. Under Medicare’s Hospital-Acquired Condition (HAC) Reduction Program, hospitals with higher infection rates face reimbursement penalties that directly incentivize the adoption of infection-prevention technologies, such as antimicrobial-coated catheters.
Evidence shows that hospitals exposed to such penalties adopt coated catheters significantly faster, with studies reporting up to 31% faster adoption and substantial reductions in infections such as CAUTI when antimicrobial coatings are used. National HAI reduction targets support long-term investment in infection control. U.S. safety initiatives have already demonstrated measurable declines in infections; for instance, historical national programs achieved major reductions in catheter-associated UTIs and central line infections, while generating large healthcare cost savings and lowering mortality. These outcomes validate infection-prevention technologies and push hospitals toward advanced catheter solutions that help meet regulatory quality metrics and accreditation requirements.
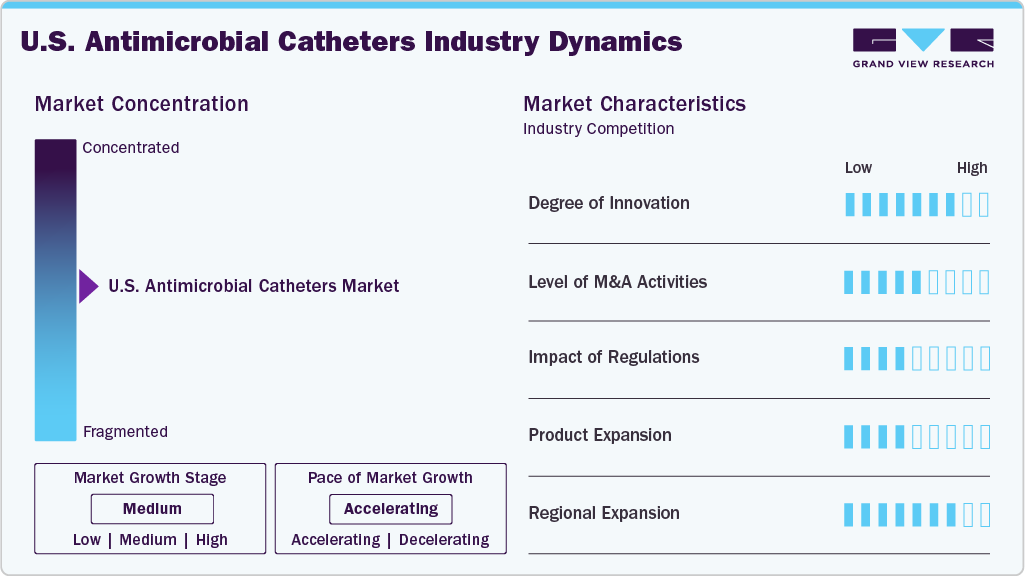
Market Concentration & Characteristics
The rising prevalence of hospital-acquired infections, the increasing focus on infection prevention, and the adoption of advanced antimicrobial technologies in vascular and urinary catheters are driving the U.S. antimicrobial catheters market. Market growth is further supported by technological innovations, including silver-coated and antibiotic-impregnated catheters, as well as regional expansions, strategic partnerships, and collaborations by key market players to enhance distribution and accessibility across the country.
The U.S. antimicrobial catheter market exhibits moderate to high levels of innovation, driven by advancements in coating technologies, materials science, and smart device integration. Companies are moving beyond traditional silver- or antibiotic-coated catheters to develop dual-agent coatings combining multiple antimicrobial agents, as well as extended-efficacy materials that release antimicrobial agents over weeks rather than days. Innovations also include non-antibiotic surfaces, such as hydrophilic and zwitterionic coatings that reduce bacterial adhesion without contributing to antibiotic resistance, and embedded sensors that allow early detection of infections, enabling faster clinical response.
In July 2024, Teleflex Incorporated announced the launch of the Teleflex Center for Antimicrobial Protection (TCAP), a strategic initiative under its Vascular Access business unit. The initiative aims to help reduce complications, such as central line-associated bloodstream infections, which can increase hospital stays and healthcare burden.
Regulations have a significant impact on the U.S. antimicrobial catheter market, shaping product development, market access, and adoption. The FDA classifies these devices as medical devices, requiring stringent 510(k) pre‑market submissions or more extensive data for novel technologies to demonstrate safety, effectiveness, biocompatibility, and antimicrobial performance. Manufacturers must also comply with Quality System Regulations (QSR) and international standards like ISO 13485, which ensure consistent product quality but increase production complexity and costs.
The U.S. antimicrobial catheter market experiences a moderate level of M&A activity, primarily driven by strategic objectives rather than high-volume consolidation. Major medical device companies engaged in targeted acquisitions to expand their antimicrobial catheter portfolios, acquire niche technologies, and enhance distribution capabilities. Many deals focus on startups or specialized firms developing advanced coatings, antimicrobial materials, or innovative catheter designs, allowing larger companies to accelerate product innovation and strengthen their competitive position.
In July 2025, Teleflex Incorporated, a global provider of medical technologies, announced the completion of its acquisition of substantially all of BIOTRONIK SE & Co. KG’s Vascular Intervention business. The acquisition expands Teleflex’s portfolio of interventional access products and strengthens its presence in the Cath lab and peripheral intervention markets.
Regional expansion in the U.S. antimicrobial catheters market is being driven by increasing healthcare infrastructure development, rising awareness about infection prevention, and growing demand for advanced medical devices across emerging economies. Market players are actively expanding their distribution networks, establishing regional offices, and forming partnerships with local healthcare providers to strengthen their market presence.
In January 2026, Medline, U.S. company, announced the opening of a new office in Riyadh, Saudi Arabia, to strengthen its presence and enhance service capabilities across the Middle East. The new facility supports Medline’s expansion strategy in the region and aims to improve customer engagement, supply chain coordination, and localized support for healthcare providers.
Product Type Insights
The vascular access catheters segment dominates the U.S. antimicrobial catheter market due to its high clinical use, its critical role in patient care, and its elevated risk of bloodstream infections. These catheters, including central venous catheters, peripherally inserted central catheters, and port access devices, are widely used in hospitals, intensive care units, and oncology settings to administer medications, fluids, and nutrition. Because vascular access devices are directly connected to the bloodstream, they carry a higher risk of catheter-associated bloodstream infections, driving the preference for antimicrobial-coated catheters to reduce infection rates. The combination of clinical necessity, stringent infection control requirements, and the availability of advanced antimicrobial technologies ensures that vascular access catheters remain the largest and most critical segment in the U.S. market.
The urological catheters segment is expected to grow at the fastest CAGR due to the rising prevalence of urinary incontinence, benign prostatic hyperplasia, and neurogenic bladder disorders, and the increasing number of urological surgeries globally. The growing elderly population, which is more prone to chronic urinary conditions requiring long-term catheterization, is also driving demand. In addition, the growing shift toward home-based care and self-catheterization, combined with rising awareness of catheter-associated urinary tract infections, is accelerating the adoption of antimicrobial urological catheters, particularly in long-term and home care settings.
Technology Insights
Silver-based technology dominates the U.S. antimicrobial catheters market in 2025, due to its proven broad-spectrum antimicrobial efficacy, safety, and widespread clinical adoption. Silver-coated catheters are highly effective at inhibiting bacterial growth and biofilm formation, which significantly reduces the risk of catheter-associated infections (CAUTIs and bloodstream infections) in hospital and long-term care settings. Their long track record of regulatory approval and clinical validation has built strong trust among healthcare providers, making silver-based antimicrobial catheters the preferred choice for both short-term and long-term use. In addition, the ability to combine silver coatings with various catheter materials, such as polyurethane and silicone, enhances versatility and further supports its dominant market position.
Organic or polymer-based antimicrobial technologies are expected to grow at the fastest CAGR, driven by increasing concerns about antimicrobial resistance and the need for non-antibiotic infection-prevention solutions. These technologies use antiseptic agents or antimicrobial polymers that prevent microbial adhesion and biofilm formation on catheter surfaces. Growing research investments, development of next-generation surface modification technologies, and rising regulatory and clinical focus on reducing antibiotic usage are supporting the rapid adoption of polymer-based antimicrobial catheters. In addition, these technologies are gaining traction in long-term catheterization and home care applications due to their safety profile and sustained antimicrobial performance.
Material Insights
The polyurethane (PU) segment dominates the U.S. antimicrobial catheter market in 2025 due to its superior mechanical properties, biocompatibility, and versatility. PU catheters offer greater flexibility and strength compared with other materials, reducing the risk of kinking or breakage during long-term use. They also support advanced antimicrobial coatings, enhancing infection prevention in both hospital and home care settings. In addition, PU catheters are well tolerated by patients and suitable for a wide range of applications, including urinary and vascular catheters. They can accommodate longer indwelling periods, making them the preferred choice for healthcare providers seeking durable, safe, and clinically effective catheter solutions.
The silicone segment is expected to grow at the fastest CAGR in the U.S. antimicrobial catheter market, driven by its excellent biocompatibility, chemical inertness, and patient comfort. Silicone catheters are less likely to cause irritation or allergic reactions, making them ideal for long-term and chronic catheterization, particularly in home care and outpatient settings. Their flexibility and smooth surfaces also reduce the risk of encrustation and biofilm formation, thereby enhancing the effectiveness of antimicrobial coatings. Furthermore, the increasing adoption of customized, patient-friendly catheter solutions and the growing preference for long-term indwelling devices are driving rapid growth in the silicone segment.
End Use Insights
Hospitals dominate the U.S. antimicrobial catheters market in 2025. This dominance is driven by the high volume of catheterized patients in acute care settings, including intensive care units and surgical wards, where the risk of catheter-associated infections is greatest. Hospitals prioritize antimicrobial catheters to comply with stringent infection-control protocols, reduce healthcare-associated infections, and improve patient outcomes. The combination of advanced clinical capabilities, regulatory compliance requirements, and higher adoption of innovative catheter technologies ensures that hospitals remain the largest segment in terms of market share.
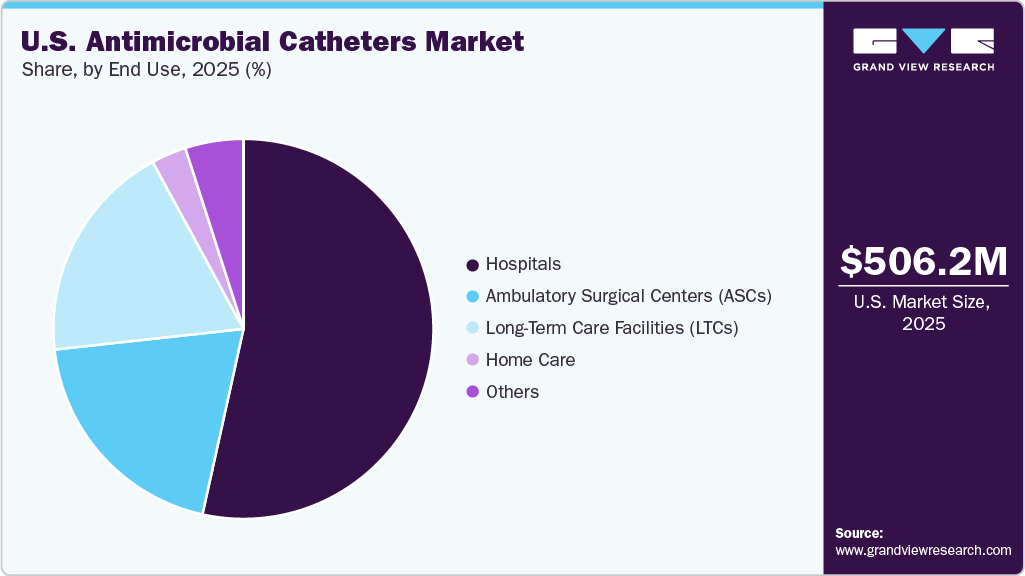
The home care segment of the U.S. antimicrobial catheter market is projected to experience the fastest CAGR in the forecast years. This growth is driven by increasing preference for home-based patient care, the rising prevalence of chronic urinary conditions that require long-term catheter use, and advancements in antimicrobial catheter technologies that enable safer and more convenient use outside hospital settings. In addition, healthcare providers and payers are encouraging home care solutions to reduce hospital stays, minimize healthcare-associated infections, and lower overall treatment costs, further supporting market expansion in this segment. The combination of patient convenience, clinical safety, and cost efficiency positions the home care segment as a key growth driver in the U.S. antimicrobial catheter market.
Key U.S. Antimicrobial Catheters Company Insights
The U.S. antimicrobial catheters market is competitive, with key players holding significant positions. The major companies are undertaking various organic as well as inorganic strategies such as new product development, collaborations, acquisitions, mergers, and regional expansion for serving the unmet needs of their customers.
Key U.S. Antimicrobial Catheters Companies:
- B. Braun SE
- Teleflex Incorporated
- SilvoGuard
- BD
- MEDLINE
- ConvaTec Group PLC
- Silq Technologies
- AvantGuard (Halomine Inc.)
- Medtronic
- Cook
- Integra LifeSciences Corporation
- Vygon
- Camstent
Recent Developments
-
In February 2026, Medline announced it signed a Prime Vendor agreement with McLaren Health Care in September 2025. Headquartered in Grand Blanc, Mich., McLaren is an integrated health system operating 12 hospitals, multiple multispecialty campuses, freestanding emergency departments, clinics, and an employed provider network across Michigan.
-
In November 2025, AvantGuard, Inc., a biotechnology company developing next-generation antimicrobial technologies inspired by the human immune system, announced that it had received three National Institutes of Health (NIH) research grants totaling USD2.65 million.
-
In September 2025, B. Braun SE announced the complete acquisition of True Digital Surgery (TDS), a Goleta, California-based company specializing in digital robotic-assisted 3D surgical microscopy. The acquisition strengthens B. Braun’s position in digital microsurgery and supports continued advancement.
-
In January 2025, BD announced expanded investments to strengthen its U.S. manufacturing network for essential medical devices to support healthcare system demand. Following more than USD 10 million invested in 2024, BD installed new syringe and needle production lines in Connecticut and Nebraska, significantly increasing capacity for safety-engineered injection devices and conventional syringes. The company also plans to invest over USD 30 million in 2025 to expand IV line manufacturing in Utah, building on earlier investments that already boosted IV catheter production.
U.S. Antimicrobial Catheters Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 546.19 million
Revenue forecast in 2033
USD 932.44 million
Growth rate
CAGR of 7.94% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product type, technology, material, end use
Country scope
U.S.
Key companies profiled
B. Braun SE; Teleflex Incorporated; SilvoGuard; BD; Medline; ConvaTec Group PLC; Silq Technologies; AvantGuard (Halomine Inc.); Medtronic; Cook; Integra LifeSciences Corporation; Vygon; Camstent
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Antimicrobial Catheters Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis on the latest industry trends and opportunities in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. antimicrobial catheters market report on the basis of product type, technology, material, and end use:
-
Product Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Urological Catheters
-
Vascular Access Catheters
-
Specialty and Surgical Catheters
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Silver-Based Technology
-
Antibiotic-Impregnated
-
Organic/Polymer-Based (Non-Metal/Non-Antibiotic)
-
Combination Technologies
-
-
Material Outlook (Revenue, USD Million, 2021 - 2033)
-
Silicone
-
Polyurethane (PU)
-
Latex
-
Others
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Ambulatory Surgical Centers (ASCs)
-
Long-Term Care Facilities (LTCs)
-
Home Care
-
Others
-
Frequently Asked Questions About This Report
The global U.S. antimicrobial catheters market size was estimated at USD 506.18 million in 2025.
The global U.S. antimicrobial catheters market is expected to grow at a compound annual growth rate of 7.94% from 2026 to 2033 to reach USD 932.44 million by 2033.
By product type, the vascular access catheters segment accounted for the largest revenue share in 2025.
Key companies operating in the U.S. antimicrobial catheters market include B. Braun SE, Teleflex Incorporated, SilvoGuard, BD, Medline, ConvaTec Group PLC, Silq Technologies, AvantGuard (Halomine Inc.), Medtronic, Cook, Integra LifeSciences Corporation, Vygon, and Camstent, among others.
The U.S. antimicrobial catheters market is primarily driven by the rising incidence of healthcare-associated infections, particularly catheter-associated urinary tract infections (CAUTIs) and bloodstream infections, which increase demand for infection-resistant catheter solutions.
About the Author(s)
Medical Devices Research Team
Healthcare · Medical DevicesThis report was authored by the medical devices Research Team at Grand View Research - comprising two research analysts, one senior research analyst, and one industry expert - with specialized expertise in the medical devices segment of the healthcare industry. All findings are based on proprietary healthcare databases, executive interviews, and regulatory analysis, subject to internal peer review prior to publication.
Last Updated:
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









