- Home
- »
- Medical Devices
- »
-
U.S. Investigational New Drug CDMO Market Report, 2033GVR Report cover
![U.S. Investigational New Drug CDMO Market Size, Share & Trends Report]()
U.S. Investigational New Drug CDMO Market (2025 - 2033) Size, Share & Trends Analysis Report By Service (Contract Development, Contract Manufacturing), By End Use (Pharmaceutical Companies, Biotech Companies), And Segment Forecasts
- Report ID: GVR-4-68040-736-1
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Investigational New Drug CDMO Market Summary
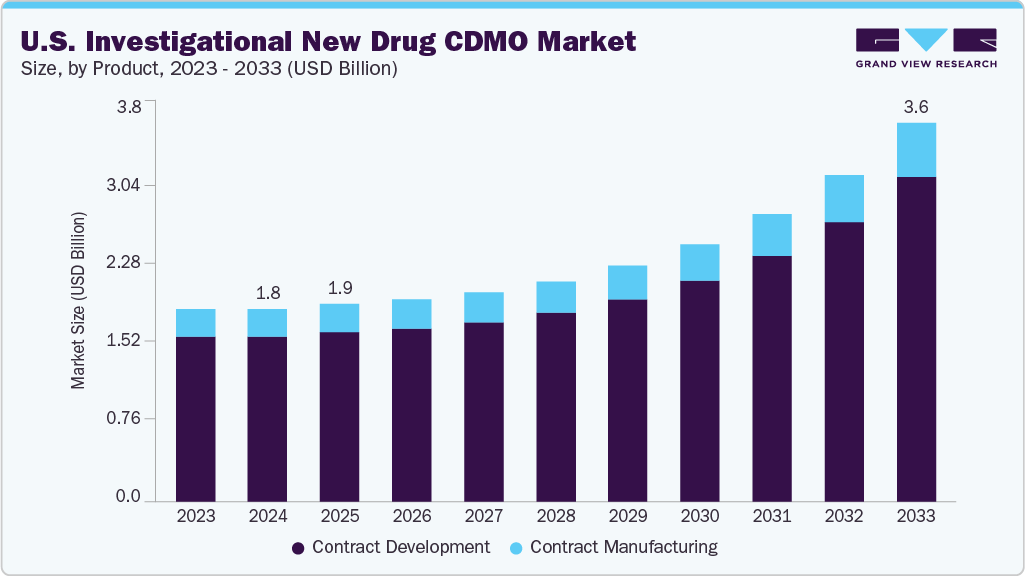
The U.S. investigational new drug CDMO market size was estimated at USD 1.82 billion in 2024 and is projected to reach USD 3.58 billion by 2033, growing at a CAGR of 9.38% from 2025 to 2033. The Investigational New Drug (IND) CDMO is experiencing significant growth in the U.S., driven by an extensive and complex early-stage pipeline, particularly in areas such as oncology, rare diseases, and advanced modalities.
Key Market Trends & Insights
- The investigational new drug CDMO in the U.S. is expected to grow significantly over the forecast period.
- Based on service, the contract development segment held the largest market share in 2024.
- Based on end use, the pharmaceutical companies segment held the largest market share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 1.82 Billion
- 2033 Projected Market Size: USD 3.58 Billion
- CAGR (2025-2033): 9.38%
Besides, emerging biopharma companies are focusing on cost efficiency and swift IND readiness as they depend on external partners for Chemistry, Manufacturing, and Controls (CMC), as well as nonclinical and clinical materials. Aspects like supply-chain resilience, a preference for domestic capacity, and faster regulatory pathways are further encouraging outsourcing. The implementation of platform processes, structured tech transfers, and phase-appropriate quality measures has helped mitigate program risks, thereby making comprehensive IND enablement more favorable outsourcing market.
In addition, growing technological advancements, particularly in digital capabilities and modality-specific approaches, have contributed to market growth. The adoption of AI-assisted Design of Experiments (DoE), digital twin technology, and automated batch-recording is helping to shorten timelines and improve execution accuracy. The use of single-use and continuous processing methods is being extended into Phase I manufacturing where feasible, and high-throughput analytics and advanced bioassays are being employed to reduce CMC package risks. Expertise in various domains, such as mRNA/Lipid Nanoparticles (LNP), viral vectors, plasmids, and highly potent active pharmaceutical ingredients (HPAPI), has been enhanced through techniques, including flow chemistry, solid-state control, and Process Analytical Technology (PAT). Besides, data integrity is being firmly integrated through electronic Quality Management Systems (eQMS) and Laboratory Information Management Systems (LIMS).
Moreover, growing investments in modular Good Manufacturing Practice (GMP), aseptic fill-finish capabilities, vector, and plasmid production capacity, microfluidic LNP platforms, and expandable Quality Control (QC) laboratories support the market growth. IND-enabling toxicology and bioanalytical capabilities are being scaled up to ensure single-contract accountability. Strategic mergers and acquisitions are being undertaken to incorporate essential capabilities and geographic redundancy, while new builds and retrofits are being prioritized for speed. Funding for workforce development and technician training is aimed at alleviating bottlenecks in biologics and sterile operations. The adoption of commercial constructs that include milestone-based pricing and reserved capacity options is intended to align incentives closely.
Furthermore, the regulatory landscape is being influenced by FDA expectations regarding phase-appropriate CMC as outlined in ICH Q8-Q12, strict data integrity measures (coded as 21 CFR Part 11), and well-defined comparability strategies. Pre-IND engagements are being utilized to ensure alignment on control strategies, analytical validation methods, and stability requirements. Expedited designations such as Fast Track, Breakthrough Therapy, and Regenerative Medicine Advanced Therapy (RMAT) status are being employed, necessitating robust processes and thorough documentation early in development. Standards for sterile products regarding aseptic processing and contamination control practices are being established from the beginning, while the incorporation of digital audit trails and Computer Software Assurance (CSA)-based validation has become a standard practice. Thus, the industry is witnessing a divide between full-service IND enablers and hyper-specialized providers.
Operational frameworks are being refined to leverage standardized platforms, simultaneous workstreams, and integrated project management, which are key to reducing cycle times. Preferred-provider agreements and long-term master service contracts are gaining traction to ensure priority access to limited resources such as vectors and sterile filling. Risk-sharing mechanisms are being employed through success-fee models and capacity reservations. Hence, the market is likely to see further consolidation and selective specialization, with growth opportunities favoring CDMOs that can demonstrate digital sophistication, depth in various modalities, and resilient domestic supply chains, all while consistently achieving first-pass IND approvals.
Opportunity Analysis
The U.S. market offers significant growth potential, especially due to an increase in complex pharmaceutical pipelines in areas like oncology, rare diseases, and advanced therapies, including cell and gene therapies, mRNA, and highly potent active pharmaceutical ingredients (APIs). Besides, rising pressure on emerging biopharmaceutical companies to accelerate their IND clearance processes has led to a greater demand for integrated development and manufacturing partners who can provide tailored, phase-appropriate solutions.
Key opportunities have become pronounced in areas such as advanced analytical platforms, digital-enabled Chemistry, Manufacturing, and Controls (CMC) development, and scalable Good Manufacturing Practice (GMP) manufacturing, particularly through single-use and modular technologies. The growing dependence on U.S.-based facilities, motivated by supply-chain security and alignment with regulatory standards, further enhances these growth prospects. CDMOs that focus on specialized capabilities-sterile fill-finish, viral vectors, plasmids, and combination product development-are in a strong position to succeed. Moreover, the trend towards strategic partnerships and flexible capacity models allows CDMOs to distinguish themselves through speed, reliability, and regulatory expertise, paving the way for substantial market expansion in the coming years.
Impact of U.S. Tariffs on the U.S. Investigational New Drug CDMO Market
The U.S. industry has faced significant challenges due to tariff impositions that have disrupted global supply chains and driven up input costs for early-stage development. Key raw materials, intermediates, and specialized equipment sourced primarily from China and Europe have incurred higher duties, leading to escalated project expenses and procurement delays. Biopharmaceutical sponsors, already operating under tight IND timelines, have encountered additional uncertainty regarding Chemistry, Manufacturing, and Controls (CMC) execution and cost forecasting. For CDMOs, these tariffs have necessitated renegotiating vendor contracts, altering sourcing strategies, and, in some cases, have led to delays in achieving readiness for analytical and GMP manufacturing. Smaller CDMOs, with their limited bargaining power and financial resources, have been particularly affected by these disruptions.
Although due to these adverse conditions, the U.S. IND CDMO market has shown resilience by bolstering domestic capabilities, accelerating nearshoring efforts, and diversifying its supplier base. The adoption of modular facility builds and single-use technologies has reduced dependency on global supply chains, while the implementation of digital procurement systems has enhanced overall resilience. Regulatory flexibility, reflected in phase-appropriate CMC guidance, has allowed CDMOs to adjust development timelines more effectively. Furthermore, the sector has benefited from strategic partnerships, mergers, and investments in advanced manufacturing, which have positioned it for recovery. Thus, tariffs have temporarily pressured costs and timelines, they have also driven significant structural changes towards improved supply security, enhanced innovation, and long-term competitiveness within the IND CDMO landscape.

In the U.S. market, technological advancements are significantly transforming early-stage development and manufacturing processes. The incorporation of artificial intelligence (AI) and machine learning is becoming increasingly common, as these technologies help optimize experimental design, predict stability outcomes, and streamline Chemistry, Manufacturing, and Controls (CMC) workflows. This shift is leading to reductions in both cost and cycle time. In addition, advanced analytical methods and bioassay technologies such as next-generation sequencing, high-throughput bioassays, and high-resolution mass spectrometry are being utilized to enhance IND data packages and ensure robust regulatory submissions. These tools not only improve data quality but also help meet the stringent requirements set by regulatory agencies. On the manufacturing side, the adoption of single-use and modular platforms is facilitating flexible, contamination-free Good Manufacturing Practice (GMP) production, which supports rapid scale-up and enhances cost efficiency. Additionally, continuous and flow manufacturing methods are offering tighter process control, increased reproducibility, and shorter production cycles, particularly for small molecules and active pharmaceutical ingredients (APIs).
Moreover, to support these advancements, CDMOs are integrating digital quality and data management systems, such as electronic Quality Management Systems (eQMS), Laboratory Information Management Systems (LIMS), and cloud-based platforms. These systems ensure compliance, provide real-time visibility, and streamline documentation processes. Thus, these innovations are allowing CDMOs to deliver IND services that are faster, more reliable, and compliant with regulatory standards, thereby establishing them as essential partners in the biopharmaceutical development process.

Pricing models are designed to meet the diverse needs of clients and the complexities of different projects. Besides, one commonly employed approach is milestone-based pricing, which links payments to specific development achievements, such as the release of GMP batches or the submission of IND applications. This model effectively reduces upfront costs for sponsors while ensuring accountability from the CDMOs.
Fixed-fee models are also prevalent, particularly for well-defined services such as analytical development. This approach offers clients predictable costs, although it can limit flexibility in cases where the project scope expands or changes. Emerging along with these traditional models is value-based pricing, which aligns the compensation received by CDMOs with the outcomes of their work, such as accelerated IND filings or successful regulatory approvals. This creates a shared risk-and-reward dynamic between CDMOs and their sponsors, fueling a more collaborative relationship. In addition, subscription or retainer models are gaining traction, providing clients with ongoing access to analytical, CMC, or regulatory support. This ensures that capacity is always available for clients while allowing CDMOs to gain predictable revenue streams. Hence, these pricing models signify a strategic shift from a purely transactional approach to a partnership-oriented framework. This evolution enables greater flexibility, efficiency, and alignment with sponsor priorities in the competitive and fast-paced landscape of IND outsourcing.
Market Concentration & Characteristics
The chart below shows that the market growth stage is moderate, and growth is accelerating. The industry is characterized by the degree of innovation, level of M&A activities, regulatory impact, service expansion, and regional expansion.
CDMOs are experiencing significant innovation by adopting AI-driven process design, advanced analytics, single-use technologies, and continuous manufacturing. The development of modality-specific platforms for cell and gene therapies, mRNA, and HPAPI underscores their commitment to differentiated, phase-appropriate solutions that accelerate IND enablement.

M&A activities in the CDMO sector are robust, fueled by the demand for niche capabilities such as sterile fill-finish, viral vector production, and advanced bioanalytics. These strategic consolidations are allowing CDMOs to provide comprehensive end-to-end IND services while also improving their geographic reach, scalability, and regulatory-compliant infrastructure.
The FDA's phase-appropriate CMC guidance, along with the ICH Q8-Q12 frameworks and a strong focus on data integrity, has significantly influenced operations in the CDMO sector. Stricter requirements regarding sterile manufacturing, comparability, and analytical robustness compel CDMOs to invest in compliance systems, ensuring that IND packages meet regulatory standards efficiently and transparently.
Service portfolios in the CDMO sector have broadened to encompass more than just core CMC services, now including integrated bioanalytical testing, IND-enabling toxicology, formulation, and regulatory documentation support. The demand for end-to-end solutions is rising, prompting CDMOs to provide bundled packages that streamline development timelines and minimize coordination challenges for sponsors.
Regional expansion efforts are concentrated on enhancing U.S. capacity while selectively extending operations into Canada and Europe. CDMOs are implementing nearshoring strategies, modular facility constructions, and forming partnerships to create resilient IND supply chains, reduce risks associated with global tariffs, and meet localized sponsor needs.
Service Insights
On the basis of service, the contract development segment held the largest market share of 85.52% in 2024. The segment is gaining momentum due to increasing demand for contract development services, as biopharmaceutical companies look for phase-appropriate solutions for early-stage assets. There has been a strong emphasis on comprehensive offerings, including pre-formulation, analytical method development, and process optimization, to meet accelerated timelines. Platform-based approaches for small molecules, biologics, and advanced modalities are being widely adopted to minimize risks and costs. In addition, the integration of AI-driven modeling and high-throughput analytics is anticipated to strengthen data packages. Thus, as outsourcing continues to rise, contract development partnerships are expected to drive the need for advanced IND submissions and ensure regulatory alignment.
The contract manufacturing segment is expected to grow significantly during the forecast period. The segment growth is driven by rising demand for clinical trial materials and phase-appropriate GMP production. There is a strong emphasis on flexible small-batch manufacturing, aseptic fill-finish, and sterile operations to support early clinical phases. The deployment of single-use technologies and modular facilities ensures both scalability and speed in production. Besides, high-potency API handling, mRNA/LNP systems, and viral vector production are increasingly outsourced due to their specialized infrastructure requirements. As early-stage pipelines continue to expand, contract manufacturing has become essential for achieving rapid IND enablement while minimizing capital expenditure for sponsors and aligning with regulatory expectations. Such factors are expected to drive the market over the estimated time period.
End Use Insights
In terms of end use, the pharmaceutical companies segment accounted for the largest revenue share in 2024. Pharmaceutical companies in the U.S. are increasingly turning to CDMOs to advance their Investigational New Drug (IND) programs amid expanding pipelines and resource limitations. CDMOs are engaged in CMC development, analytical testing, and GMP production to help accelerate timelines and mitigate operational risks. Early-stage collaborations are being utilized to ensure that phase-appropriate quality standards and robust data packages are met. Strategic partnerships are structured to offer flexibility in scaling efforts across various therapeutic areas, particularly in oncology and rare diseases. By outsourcing IND-enabling activities, pharmaceutical companies can optimize their R&D productivity while allowing internal resources to concentrate on core discoveries and the execution of clinical strategies. Such aforementioned factors are expected to drive the market.
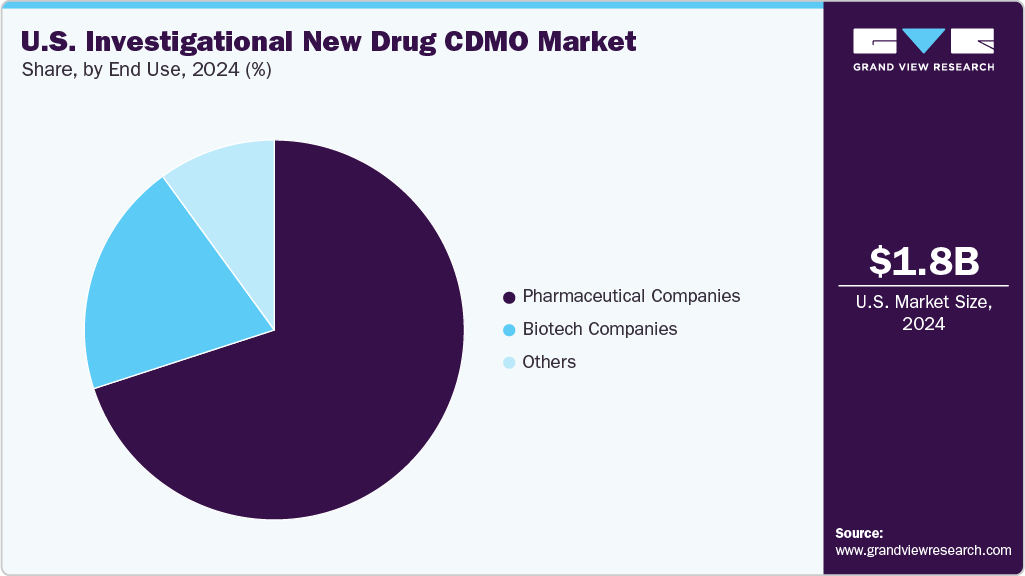
The biotech companies segment is expected to grow at the second-fastest CAGR during the forecast period. The segment is driven by rising early-stage drug innovations and expanding product pipelines. CDMOs play a vital role in providing development, analytical, and manufacturing support, which helps biotech companies efficiently navigate IND submissions while managing costs and risks. As biologics, cell and gene therapies, and regulatory complexities grow, biotech firms increasingly rely on specialized CDMOs. This partnership not only enhances clinical pipelines but also promotes innovation by incorporating advanced technologies such as AI-driven trial design, modular manufacturing, and data management systems, solidifying the U.S. as a leading biopharma hub.
Key U.S. Investigational New Drug CDMO Company Insights
The key players operating across the market are adopting strategic initiatives such as service launches, mergers & acquisitions, partnerships & agreements, and expansions to gain a competitive edge in the market. For instance, in June 2025, Zydus Lifesciences announced its plans to enter the global biologics contract development and manufacturing (CDMO) market through the acquisition of Agenus’ advanced biologics manufacturing facility in the U.S. This strategic move strengthens Zydus’ position in the rapidly expanding biologics CDMO space and establishes a significant footprint in one of the world’s leading biotech hubs.
Key U.S. Investigational New Drug CDMO Companies:
- Catalent, Inc.
- Lonza
- Recipharm AB
- Siegfried Holding AG
- Patheon Inc.
- Covance
- IQVIA Holdings Inc.
- Cambrex Corporation
- Charles River Laboratories International, Inc.
- Syneos Health
- Syngene International
- Jubilant Pharmova
- Piramal Pharma Solutions
- Dr. Reddy’s CPS
- Dishman Carbogen Amcis Ltd.
- Biocon Biologics
- Laurus Labs
Recent Developments
-
In March 2024, Lonza inked an agreement for the acquisition of the manufacturing facility in the U.S. from Roche for USD 1.2 billion. This is expected to significantly increase Lonza's capacity to produce biologic drugs & expand its presence in the U.S. Lonza plans to invest an additional USD 500 million to upgrade the facility and meet the demand for next-generation biologic therapies.
-
In March 2023, Societal CDMO, Inc. announced that it received approval from the U.S. FDA to serve as the manufacturer of a commercial tablet product. The company plans to initiate commercial-scale production at its Gainesville, Georgia facility later in 2023.
U.S. Investigational New Drug CDMO Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 1.87 billion
Revenue forecast in 2033
USD 3.58 billion
Growth rate
CAGR of 9.38% from 2025 to 2033
Actual Data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Service, end use
Country scope
U.S.
Key companies profiled
Catalent, Inc.; Lonza, Recipharm AB; Siegfried Holding AG; Patheon Inc.; Covance; IQVIA Holdings Inc.; Cambrex Corporation; Charles River Laboratories International, Inc.; Syneos Health; Syngene International; Jubilant Pharmova; Piramal Pharma Solutions; Dr. Reddy’s CPS; Dishman Carbogen Amcis Ltd.; Biocon Biologics; Laurus Labs
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Investigational New Drug CDMO Market Report Segmentation
This report forecasts revenue growth at and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. investigational new drug CDMO market report based on service and end use.
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Contract Development
-
Small Molecule
-
Bioanalysis and DMPK Studies
-
Toxicology Testing
-
Pathology and Safety Pharmacology Studies
-
Drug Substance Development
-
Synthetic Route Development
-
Process Development
-
Form Selection Crystallization Process Development
-
Scale-up of Drug Substance
-
-
Formulation Development
-
Pre Formulation
-
Preclinical Formulation Selection
-
First In Man Formulation/ Process Development
-
-
Analytical & Quality Services
-
Analytical Method Development / Validation
-
Release Testing of Drug Substance and Drug Product
-
Formal Stability of Drug Substance and Drug Product
-
-
Process Optimization
-
Work Up Purification Steps
-
Telescoping & Process Refining
-
Initial Optimization
-
-
-
Large Molecule
-
Cell Line Development
-
Process Development
-
Upstream
-
Microbial
-
Mammalian
-
Others
-
-
Downstream
-
MABs
-
Recombinant Proteins
-
Others
-
-
-
-
-
Contract Manufacturing
-
Small Molecule
-
Oral Solids
-
Semi-Solids
-
Liquids & Injectables
-
Others
-
-
Large Molecule
-
MABs
-
Recombinant Proteins
-
Others
-
-
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical Companies
-
Biotech Companies
-
Others
-
Frequently Asked Questions About This Report
b. The U.S. investigational new drug CDMO market size was estimated at USD 1.82 billion in 2024 and is expected to reach USD 1.87 billion in 2025.
b. The U.S. investigational new drug CDMO market is expected to grow at a compound annual growth rate (CAGR) of 9.38% from 2025 to 2033 to reach USD 3.58 billion by 2033.
b. The contract development segment dominated the U.S. investigational New Drug CDMO market in 2024 with a market share of 85.52%. The segment is driven by increasing demand for contract development services, as biopharmaceutical companies look for phase-appropriate solutions for early-stage assets. Besides, strong emphasis on comprehensive offerings, including pre-formulation, analytical method development, and process optimization, to meet accelerated timelines is contributing to segment growth.
b. Some of the key market players include Catalent, Inc., Lonza, Recipharm AB, Siegfried Holding AG, Patheon Inc., Covance, IQVIA Holdings Inc., Cambrex Corporation, Charles River Laboratories International, Inc., Syneos Health, Syngene International, Jubilant Pharmova, Piramal Pharma Solutions, Dr. Reddy’s CPS, Dishman Carbogen Amcis Ltd., Biocon Biologics, and Laurus Labs, among others.
b. The market for investigational new drug CDMO is driven by an extensive and complex early-stage pipeline, particularly in areas such as oncology, rare diseases, and advanced modalities. Besides, emerging biopharma companies are focusing on cost efficiency, and swift IND readiness has led the companies to increasingly depend on external partners for Chemistry, Manufacturing, and Controls (CMC) and nonclinical and clinical materials, which further contributes to the market growth.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










