- Home
- »
- Medical Devices
- »
-
U.S. OTC Continuous Glucose Monitoring Devices Market, 2033GVR Report cover
![U.S. OTC Continuous Glucose Monitoring Devices Market Size, Share & Trends Report]()
U.S. OTC Continuous Glucose Monitoring Devices Market (2025 - 2033) Size, Share & Trends Analysis Report By Users (Non-Diabetic, Prediabetic, Diabetic), By Application (Lifestyle Wellness, Diabetes Monitoring), By Sales Channel, And Segment Forecasts
- Report ID: GVR-4-68040-717-8
- Number of Report Pages: 125
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
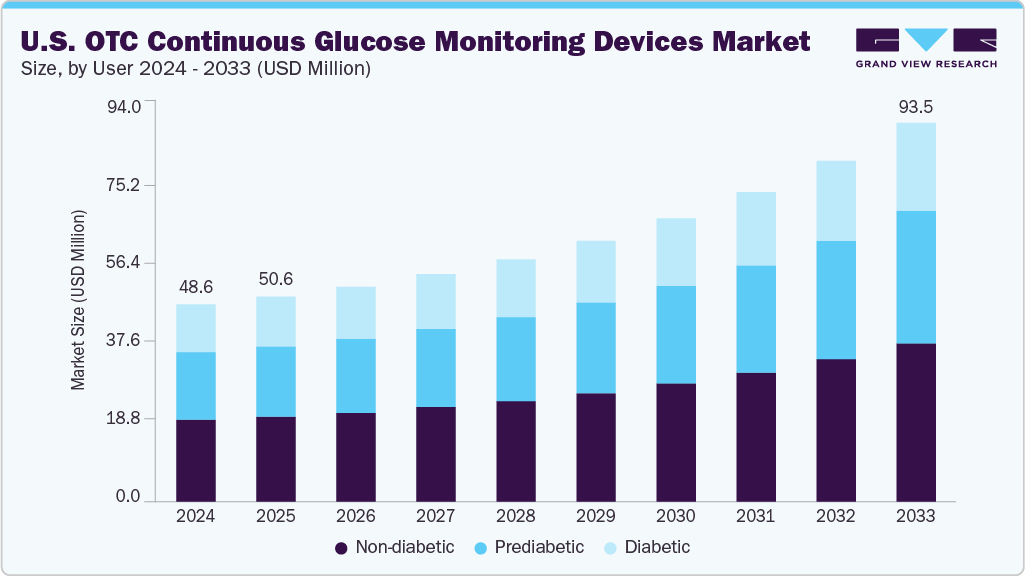
The U.S. OTC continuous glucose monitoring devices market size was estimated at USD 48.61 million in 2024 and is expected to reach USD 93.52 million by 2033, growing at a CAGR of 8.0% from 2025 to 2033. The rising adoption of lifestyle wellness devices, including continuous glucose monitoring (CGM) to ease diabetes management, and the easy access to wearable health tools are fueling demand. For instance, in 2024, more than 44% of American adults adopted wearable health tools for disease monitoring and management.
Additionally, in the coming 5 to 10 years, the adoption rate is expected to reach 50% among elderly Americans. The growing geriatric population and sedentary lifestyles contribute to the disease burden, imposing the development of advanced monitoring solutions such as CGM. Furthermore, expanding healthcare infrastructure and distribution of CGM devices through e-commerce without a prescription would ease accessibility. Technological advancements like sensor accuracy, app integration, and data sharing further support market growth across urban and semi-urban areas.
As of 2025, nearly 38 million American adults are diagnosed with diabetes, marking 11.5% of the total adult population in the U.S. Furthermore, around 97 million adults are prediabetic in the country. Obesity and a sedentary lifestyle are the major contributing factors to the prevalence of diabetes. Continued surge in type 2 diabetes cases and growing awareness of early diagnoses are likely to boost the demand for OTC CGM devices in the U.S.
Newly introduced OTC CGM devices are suggested to be used by type 2 diabetes that are not on insulin. As type 2 diabetes does not get the insurance for prescription CGM, OTC comes in as a help. OTC CGM sells at an affordable rate and without a prescription. Although the OTC CGMs are not covered by insurance, the HAS/FSA funds are generally accepted.
Diabetes is a chronic disease caused by insufficient insulin production or improper insulin usage, leading to high blood glucose levels. Left uncontrolled, it can damage vital organs and cause severe complications, including heart disease, kidney failure, blindness, and nerve damage. Early symptoms include frequent urination, thirst, fatigue, blurred vision, and unexplained weight loss.
Avoiding type 2 diabetes requires healthy lifestyle modification such as consistent physical activity, a low-fat and sugar diet, maintaining a healthy weight, and avoiding tobacco. Experts from the ADA and AACE also highlight the increasing role of continuous glucose monitoring (CGM) devices in proactive diabetes management, especially in non-insulin users and prediabetic patients.
CGMs are transforming diabetes care by offering real-time glucose readings and reducing the need for finger-prick tests. Currently, there are three OTC CGM devices in the U.S. market:
-
Dexcom Stelo - The first FDA-approved OTC CGM device in the U.S. The device got FDA approval in March 2024. The device comes with two sensors, usually worn out in 15 days, thus one pack serves one month of continuous glucose monitoring for adults with type 2 non-insulin users and adult fitness enthusiasts.
-
Lingo by Abbott - FDA approved in June 2024, is a wellness-focused OTC CGM device built for pre diabetic or non-diabetic users. The device is not suggested for diabetes management. However, it can help in understanding the best diet and lifestyle practices.
-
Libre Rio by Abbott - FDA approved in June 2024, is used for non-insulin type 2 diabetics. The device is built on FreeStyle Libre platforms for OTC settings and simplified use.
Although the market has only two players, the unsaturated and untapped market space gives beneficial revenue opportunities for new entrants.
Market Concentration & Characteristics
The U.S. OTC Continuous Glucose Monitoring (CGM) devices industry is concentrated, with currently two players dominating market share through advanced technology and established distribution networks. The industry is characterized by rapid innovation, increasing demand for real-time glucose monitoring, and growing awareness of early diagnosis. New players and startups are projected to enter the field owing to the rising digital health adoption. The market has seen significant vertical expansion via e-commerce channels. High regulatory standard and pricing sensitivity also define the landscape, making innovation and affordability key to sustained growth and market penetration.
The OTC continuous glucose monitoring (CGM) industry in the U.S. is yet in a nascent stage, giving wider room for innovation and expansion. The market players Dexcom and Abbott are targeting a broader application range, falling in preventive measures. Stelo by Dexcom and Lingo by Abbott promise blood glucose range monitoring for non-insulin users or prediabetics. These devices, after successful FDA approval, were made available on Amazon for easy marketing and avoiding healthcare gatekeepers, thus broadening the product’s image to a lifestyle wellness tool.

The OTC continuous glucose monitoring (CGM) industry has witnessed moderate but steadily increasing merger and acquisition (M&A) activity in recent years, driven by the growing demand for convenient, real-time glucose tracking solutions. Strategic acquisitions are primarily aimed at expanding product portfolios, integrating advanced sensor technologies, and gaining regulatory expertise to accelerate market entry. Larger medical device companies are acquiring innovative startups to access proprietary CGM technologies and strengthen distribution networks. Additionally, partnerships with digital health firms are common, enabling integration with mobile platforms and health apps. However, regulatory complexities and reimbursement challenges continue to influence the pace of M&A deals.
Regulations significantly impact the U.S. OTC continuous glucose monitoring (CGM) devices industry by ensuring safety, quality, and compliance. Under the FDA regulations, OTC CGM devices are classified as Class II medical devices under 21 CFR 862.1355, requiring FDA 510 (k) premarket notification approval for manufacturing and import. These regulations align U.S. standards with global practices, improving product reliability and user trust. However, they also pose challenges for smaller manufacturers due to complex documentation and extended approval timelines, potentially slowing market entry while encouraging innovation and adherence to international norms.
The key substitutes include prescription CGM devices that are widely adopted due to the favorable insurance scenario. Traditional self‑monitoring of blood glucose (SMBG) through finger-prick glucometers is another substitute for OTC CGM owing to its lower cost and easier accessibility. Flash glucose monitoring systems providing intermittent glucose readings without real-time alert also serve as alternatives. Adding to this, OTC at-home A1c test kits act as an alternative for OTC CGM. These factors also contribute to slowing the adoption of OTC CGM devices in the region.
OTC continuous glucose monitoring (CGM) devices market in the U.S. is expanding beyond major metropolitan areas into Tier-2 and Tier-3 cities, driven by growing diabetes prevalence, rising awareness about metabolic health, easy availability, and rising disposable incomes. Urban regions remain the primary adopters due to better infrastructure and digital literacy. However, growth in the outskirts of the nation and semi-urban areas is increasing due to telemedicine, mobile health services, and e-commerce. Companies are adopting oligo-channel policy to reach a larger patient pool. Such approaches would reflect positive growth in the long run.
User Insights
The non-diabetic users dominated the market and accounted for the largest revenue share of 41.46% in 2024. Due to rising awareness about preventive care for diabetes and metabolic health, the adoption of OTC CGM is highly observed in non-diabetic or fitness enthusiasts. For instance, by 2024, over 120 million Americans were active wellness tracker users, of which 66% accounted adult-aged Americans. Their wider adoption stems from ease of use, affordability, and accessibility across urban and emerging markets. Users benefit from continuous data, alerts, and insights into glucose trends without needing complex system integrations.
The prediabetic user segment is projected to experience the fastest growth in the OTC CGM devices market in the U.S. over the forecast period. The constant rise in the number of prediabetic populations in the U.S. is enhancing the adoption of diabetes monitoring devices, including OTC CGM. Their advanced features, such as real-time data collection and remote monitoring capabilities, appeal to patients seeking smarter and more fitting care. As technology becomes more affordable and users become more health-conscious, adoption is rising among tech-savvy consumers and healthcare providers. Regulatory approvals and increasing physician awareness will further drive market penetration and growth in this segment.
Application Insights
The lifestyle wellness segment dominated the U.S. OTC CGM devices market with a revenue share of 62% in 2024. OTC CGM like Dexcom Stelo is suggested to be used by non-insulin users and as a wellness tracker instead of a diabetes management device. The device fails to read blood glucose levels when out of range. An individual living with diabetes goes through out-of-range blood sugar hits often. In such instances, the failed alert on high or low blood glucose might cause inconvenience to the users, especially for diabetic patients who often face hyperglycemia. In addition, early onset and the need for continuous care are driving demand for real-time monitoring solutions, including OTC CGM for lifestyle wellness applications.
The diabetes monitoring segment is expected to register the fastest growth in the U.S. OTC continuous glucose monitoring (CGM) devices market over the forecast period, driven by the rising prevalence of diabetes and prediabetes, increasing health awareness, and demand for convenient, needle-free glucose tracking. Advancements in sensor accuracy, wearable comfort, and smartphone integration are making CGM devices more appealing for daily monitoring without clinical supervision. Supportive regulatory approvals for OTC use, along with growing retail availability, are further accelerating adoption. Additionally, the trend toward preventive healthcare and self-management is boosting demand, positioning diabetes monitoring as the key growth driver in this segment.
Sales Channel Insights
The company website segment dominated the U.S. OTC CGM devices market, accounting for the largest revenue share in 2024, due to its established distribution networks across urban and rural areas. Dexcom and Abbott launched their OTC CGM devices initially on the company website. The companies have wider networking across the nation, thus easy to capture the target population. Moreover, no other sales channel is available for the year, making it predominant for 2024. Company websites offer trusted, point-of-care locations where customers can buy OTC CGM devices without prescriptions or lengthy waits.
The retailer’s segment is poised to achieve the fastest growth in the U.S. OTC CGM devices market due to various contributing factors. Majorly due to its convenient buying allows users, especially in remote and semi‑urban areas, to access a wide range of OTC CGM products without relying on company sites. Users could easily buy the OTC CGM along with other goods and avoid dealing with multiple packages. Moreover, inexpensive online pricing, frequent promotions, and bundled deals make OTC CGM devices more affordable. Add to this, e-commerce retailers such as Amazon offer features like ease of comparing specifications, reading peer reviews, and accessing customer support via digital platforms, which build user confidence, driving adoption across diverse demographic segments.
Key U.S. OTC Continuous Glucose Monitoring Devices Company Insights
Market players include Abbott and Dexcom, who have positioned themselves significantly in the U.S. OTC CGM market through innovation and competitive pricing. By focusing on the pricing side for users, user-friendliness by skipping the requirement for a prescription, and expanded access, they have significantly driven OTC CGM adoption.
Key U.S. OTC Continuous Glucose Monitoring Devices Companies:
- Abbott Laboratories
- Dexcom Inc.
Recent Developments
-
In February 2025, Abbott announced the availability of its Lingo OTC CGM device on Amazon. With the exclusive distribution deal, the company is targeting to expand the clientele in the nation and capture the untapped market.
-
After Abbott’s Lingo Dexcom in May 2025, Dexcom made its Stelo OTC CGM available via Amazon. The supply dealership with Amazon is expected to broaden Stelo’s (Dexcom) penetration into Type 2 diabetes, prediabetes, and lifestyle wellness.
U.S. OTC Continuous Glucose Monitoring Devices Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 50.56 million
Revenue forecast in 2033
USD 93.52 million
Growth rate
CAGR of 8.0% from 2025 to 2033
Actual data
2021 -2024
Forecast period
2025- 2033
Quantitative units
Revenue in USD million and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
User, application, sales channel
Regional scope
U.S.
Key companies profiled
Abbott Laboratories; Dexcom Inc.
Customization scope
Free report customization (equivalent upto 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. OTC Continuous Glucose Monitoring Devices Market Report Segmentation
This report forecasts revenue growth at country levels and analyzes the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. OTC continuous glucose monitoring devices market report based on user, application, and sales channel:
-
User Outlook (Revenue, USD Million, 2021 - 2033)
-
Non-diabetic
-
Prediabetic
-
Diabetic
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Lifestyle Wellness
-
Diabetes Monitoring
-
-
Sales Channel Outlook (Revenue, USD Million, 2021 - 2033)
-
Company Website
-
Retailers
-
Frequently Asked Questions About This Report
b. The U.S. OTC continuous glucose monitoring devices market size was valued at USD 48.61 million in 2024 and is expected to reach a value of USD 50.56 million in 2025.
b. The U.S. OTC continuous glucose monitoring devices market is expected to grow at a compound annual growth rate of 8.0% from 2025 to 2033 to reach USD 93.52 million by 2033.
b. The non-diabetic users segment accounted for the largest revenue share in 2024, due to the rising awareness about preventive care for diabetes and metabolic health.
b. Some key players operating in the U.S. OTC continuous glucose monitoring (CGM) devices market include Abbott Laboratories and Dexcom Inc.
b. Key factors driving the growth of the U.S. OTC continuous glucose monitoring (CGM) devices market include the rising adoption of lifestyle wellness devices, including CGM, to ease diabetes management, and the easy access to wearable health tools, which are fueling demand.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










