- Home
- »
- Pharmaceuticals
- »
-
Vaccine Contract Manufacturing Market Size Report, 2030GVR Report cover
![Vaccine Contract Manufacturing Market Size, Share & Trends Report]()
Vaccine Contract Manufacturing Market (2024 - 2030) Size, Share & Trends Analysis Report By Vaccine Type (Attenuated, Inactivated), By Workflow (Downstream, Upstream), By Application (Human, Veterinary), By Region, And Segment Forecasts
- Report ID: GVR-2-68038-087-3
- Number of Report Pages: 133
- Format: PDF
- Historical Range: 2018 - 2022
- Forecast Period: 2024 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Vaccine Contract Manufacturing Market Summary
The global vaccine contract manufacturing market size was estimated at USD 2.14 billion in 2023 and is expected to reach USD 3.60 billion by 2030, growing at a CAGR of 7.8% from 2024 to 2030. Increased demand for vaccines trend of contact manufacturing are key growth contributors to this market.
Key Market Trends & Insights
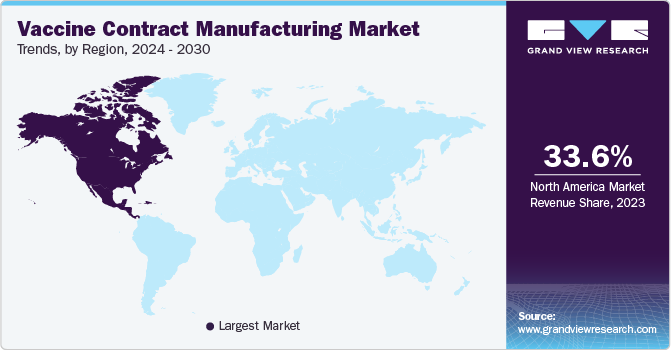
- North America accounted for the largest share in vaccine contract manufacturing market.
- The vaccine contract manufacturing market in the U.S. held a largest share in North America.
- Based on vaccine type, the attenuated vaccines segment led the market in 2023 with the largest revenue share of 29.9%.
- Based on workflow, the downstream segment dominated the market with the largest revenue share of 50.9% in 2023.
- Based on application, the human-use segment led the market with the largest revenue share of 92.24% in 2023.
Market Size & Forecast
- 2023 Market Size: USD 2.14 Billion
- 2030 Projected Market Size: USD 3.60 Billion
- CAGR (2024-2030): 7.8%
- North America: Largest market in 2023
Apart from the safety and effectiveness of inoculations, the number of products developed per year is also an important factor to consider while planning the manufacturing process. This is mainly due to an increase in the number of countries demanding the introduction of vaccines. The pharmaceutical industry needs to address this rising global demand to cater to the growing population of varied age groups.
Increasing vaccination coverage and ongoing innovations are expected to serve as high-impact rendering drivers for the market. The number of vaccines in the clinical developmental phase is constantly increasing due to an effectiveness of vaccines in combating several illnesses. An increase in demand for vaccines bolsters the requirement for contract services to help reduce the timeline for launch of products. Well-established and emerging players are focusing on CMOs/CROs as a strategic tool to accelerate their R&D for vaccines. Hence, increased adoption of these services is anticipated to result in development of a robust pipeline of pharmaceuticals in the near future.
Growing concern over global shortage and inequitable access to vaccines are some of a high impacts rendering drivers expected to augment the scope of vaccine contract manufacturing. This is particularly applicable to emerging economies with limited resources. With technological advancements, the number of companies outsourcing to CMOs in the market is increasing. As a result, CMOs are expanding their manufacturing and R&D capabilities to meet the growing consumer demand.
For instance, in October 2023, Lonza and Vaxcyte expanded their collaboration to enhance global commercial production of Pneumococcal Conjugate Vaccines. The new agreement aimed to establish worldwide commercial manufacturing capacity for Vaxcyte's PCV candidates-VAX-31 and VAX-24 -catering to adult & pediatric populations. This expanded collaboration aligned with Vaxcyte's existing strategy to initiate the commercial launch of VAX-24 for adults, leveraging Lonza's facilities in Visp, Switzerland.
The adoption of biologics has also increased in the recent past. Various companies have increased investment in biologics to meet the growing demand. For instance, in September 2023, the Australian biotech company GPN Vaccines extended its collaboration with the domestic Contract Development and Manufacturing Organization (CDMO) BioCina.
Market Concentration & Characteristics
The market growth stage is medium, and pace of the market growth is accelerating. The market is characterized by a high degree of innovation. Vaccine manufacturing is inherently complex due to the need for precise biological processes and quality control measures. This complexity drives innovation in manufacturing techniques, technologies, and processes to improve efficiency, scalability, and safety.
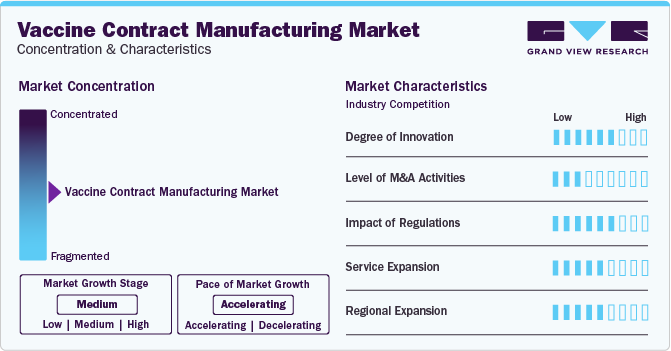
The market is also characterized by a key player's medium level of merger and acquisition (M&A) activity. Companies are pursuing M&A to diversify their service portfolio and expand their offering beyond vaccine manufacturing. This includes capabilities in research and development, formulation, fill-finish services, or other aspects of the pharmaceutical value chain.
The market is characterized by a high level of regulations impact. Vaccines are administered to healthy individuals to prevent diseases, so regulatory agencies impose strict safety and efficacy standards. Contract manufacturers must adhere to these regulations to ensure that vaccines meet quality and performance requirements.
Companies are expanding their service offerings in the market due to increasing demand for vaccines globally, driven by population growth, expanding vaccination programs, and emergence of new infectious diseases. Contract manufacturing provides a flexible and scalable solution for meeting this rising demand.
Companies are expanding their regional presence in the global market. The demand for vaccines continues to rise globally due to population growth, increasing awareness of preventive healthcare, and the need to combat infectious diseases. Expanding regionally allows companies to tap into new markets and capture a larger share of the growing market.
Vaccine Type Insights
Based on type, the attenuated vaccines segment led the market in 2023 with the largest revenue share of 29.9%. These vaccines are relatively easy to create for certain viruses; hence, with growing cases of viral illness, this segment is also expected to witness a significant CAGR during the forecast period. Furthermore, with maturity in manufacturing technology, the reversion of attenuated vaccines to a virulent, disease-producing state is exceptionally low or nonexistent. This factor also supports the large share of this segment.
However, DNA vaccines segment is expected to witness the fastest CAGR of 9.5% over the forecast period. DNA vaccines are expected to offer lucrative growth opportunities in the future. Several types are being tested on humans. Continuous experiments & investigations to study the efficacy and safety of these vaccines are expected to drive the market growth. DNA vaccines are anticipated to revolutionize immunization.
Workflow Insights
Based on workflow, the downstream segment dominated the market with the largest revenue share of 50.9% in 2023. The segment is expected to maintain its position over a forecast period. Vaccine production requires skilled personnel for upstream as well as downstream processing. Contract manufacturing organizations (CMOs) are providing producers with expertise and innovative technologies to accelerate an entire production chain.As downstream processing includes product recovery and purification steps, it requires immense attention and capital. Hence, a high requirement for enhanced biotechnological tools, capital, and skilled personnel for recovery of the vaccine as the final product is a major factor that has boosted the segment's growth over the past years.
The upstream segment is expected to witness the fastest CAGR of 8.0% over the forecast period.Upstream workflow includes the development of scalable production & high-titer processes to express different target molecules through cell culture systems, microbial fermentation, transgenic sources, and insect cells. Upstream workflow includes process transfer-in & development, supply & toxicology study supply batches, recovery of soluble or solids materials, cell disruption, and process monitoring. Ongoing development in recombinant technologies and process control strategies is expected to drive growth in this segment.
Application Insights
Based on application, the human-use segment led the market with the largest revenue share of 92.24% in 2023. According to a CDC report, vaccinations given to young children and infants in the past 20 years are expected to prevent 322 million illnesses and save 732,000 lives in the U.S. in a coming year. Various programs have been conducted by government bodies and other healthcare agencies to meet a demand for vaccines. WHO collaborates with nations worldwide to promote the significance of vaccines and immunization and guarantees that governments receive an essential guidance & technical assistance to establish high-quality immunization programs.
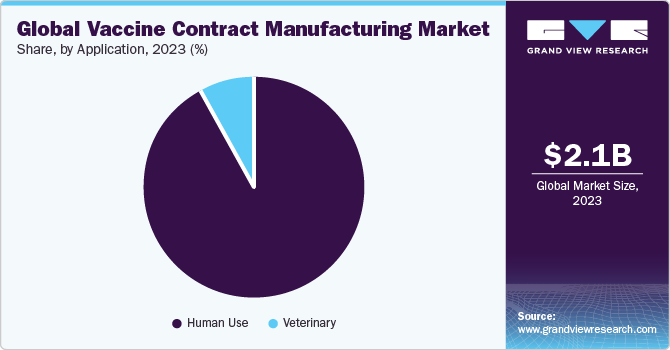
However, it has been observed that vaccination plays an important role in improving veterinary health and preventing animal diseases. This is anticipated to drive the use of CMOs for animal inoculation production significantly. Vaccination reduces the need for antibiotics to treat animals. Moreover, with the growth in the human population, demand for animal-based food continues to grow. Hence, the importance of vaccination in the meat-production industry is also expected to increase. These factors are likely to stoke a growth of the segment.
Regional Insights
The vaccine contract manufacturing market in North America accounted for the largest share in this market mainly due to a widespread manufacturing of vaccines in the region, coupled with the growing presence of a substantial number of biopharmaceutical facilities. State and other laws about developing vaccines and other biologics in North America, particularly in the U.S. and Canada, are expected to significantly impact the progress of contract manufacturing organizations (CMOs) and contract research organizations (CROs) in the field.
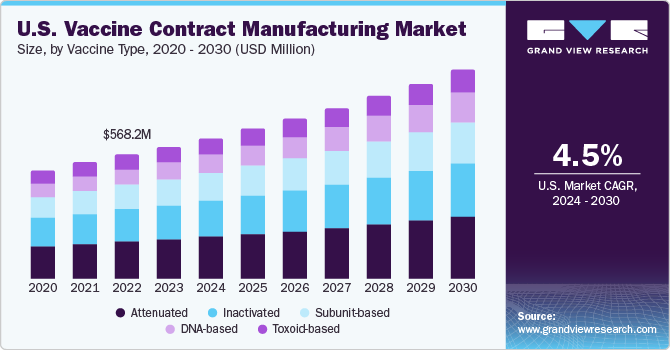
U.S. Vaccine Contract Manufacturing Market Trends
The vaccine contract manufacturing market in the U.S. held a largest share in North America. The U.S. is recognized as a largest biopharmaceutical market concerning biopharmaceutical manufacturing & sales, in addition to a local presence of biomanufacturing facilities. According to the Pharmaceutical Research and Manufacturers Association (PhRMA), most pharmaceutical R&D is being conducted in this country. These factors have strongly affected the market growth over the forecast period.
Asia Pacific Vaccine Contract Manufacturing Market Trends
The vaccine contract manufacturing market in Asia Pacific is expected to grow at a lucrative CAGR over a forecast period. This can be attributed to favorable regulatory changes, improving infrastructure, and a presence of a large number of potential study subjects.
Growing R&D costs have compelled many U.S. biopharma companies to consider outsourcing vaccine development to Asia Pacific countries, where a large number of players offer low-cost production facilities & labor.
The China vaccine contract manufacturing market held the largest share in Asia Pacific. China is a cost-effective market for vaccine contract manufacturing. Moreover, Chinese CMOs adhere to cGMP standards and have improved regulatory oversight, which ensures superior quality. Thus, increasing demand for superior quality and ongoing advancements in the pharmaceutical sector are expected to continue driving the market growth.
The vaccine contract manufacturing market in India is expected to grow at the significant CAGR over the forecast period. This can be attributed to low costs, availability of industry experts, and presence of WHO-cGMP-compliant facilities. Furthermore, some of the key players in India's vaccine contract manufacturing services are Charles River Laboratories International, Inc. and Catalent, Inc. Local players include Dr. Reddy's Laboratories, Cadila Healthcare Limited, and Cipla Ltd. Several CMOs & CROs operating in India have expanded their facilities to meet the growing demand for vaccine CMOs.
Europe Vaccine Contract Manufacturing Market Trends
The vaccine contract manufacturing market in Europe was the second-largest contributor with the revenue share of 31.0% in 2023. Furthermore, government bodies and funding agencies have played a pivotal role in funding vaccine research. Moreover, presence of the European Investment Bank (EIB) financing facilities which are engaged in supporting R&D of vaccines for combating infectious diseases through various initiatives is expected to drive the country market.
The Germany vaccine contract manufacturing market held a largest share in Europe. Increase in pharmaceutical development by local CROs/CMOs is a major factor responsible for market growth. Moreover, demand for CMOs to improve drug development, product maintenance, and related services is expected to drive market growth in the coming years. In addition, government initiatives for research activities have accelerated the demand for vaccine contract manufacturing services.
Latin America Contract Manufacturing Market Trends
Latin America region is expected to offer lucrative growth opportunities for the vaccine contract manufacturing market. Lower costs of conducting a clinical trial is also expected to drive the growth of vaccine CMOs in this region. Presence of regulatory bodies such as Agência Nacional de Vigilância Sanitária, Administración Nacional de Medicamentos, Alimentos Tecnología Médica, Instituto Nacional de Vigilancia de Medicamentos e Alimentos, which grant GMP certifications to manufacturing facilities are expected to play a crucial role in propelling the market growth over the forecast period.
Among the Latin American countries majority of clinical trials are conducted in Brazil owing to low cost, easy patient recruitment, and diverse population. As large number of trials are conducted in this region, it is expected to propel market growth. The Brazilian government has implemented several initiatives to support the development and production of vaccines, including tax incentives and funding for research and development. This has encouraged both local and international companies to invest in the Brazilian market.
The vaccine contract manufacturing market in Mexico is expected to grow at a significant CAGR over the forecast period. Mexico is attracting international pharma companies and contract manufacturers. In addition, the country provides a favorable market environment to attract R&D investment and expansion, stimulating the demand for CMOs. Most pharmaceutical companies in the country are gradually expanding and modernizing their production criteria with the support of CMO to increase local & international market supply of oral solid dosage products.
Key Vaccine Contract Manufacturing Company Insights
Key companies are undertaking various market strategies, such as merger & acquisition, collaboration, regional expansion, service portfolio expansion, and competitive pricing, to sustain in competitive environment and acquire a higher market share. For instance,For instance, in October 2023, Vaxcyte and Lonza announced expanding their collaboration for a global commercial manufacturing of broad-spectrum Pneumococcal Conjugate Vaccines (PCVs).
Key Vaccine Contract Manufacturing Companies:
The following are the leading companies in the vaccine contract manufacturing market. These companies collectively hold the largest market share and dictate industry trends.
- Lonza
- FUJIFILM Diosynth Biotechnologies
- Ajinomoto Co., Inc.
- Merck KGaA
- Catalent, Inc
- IDT Biologika GmbH
- Curia Global, Inc.
- ICON plc.
- Cytovance Biologics (Hepalink)
- Syngene International Limited
Recent Developments
-
In June 2023, FUJIFILM Corporation announced an opening of its commercial office in Tokyo. The facility aimed to provide enhanced sales and customer support services for contract development and manufacturing services for biologics and advanced therapies to Asia-based pharmaceutical and biotechnology companies
-
In December 2023, Ajinomoto Co., Inc. has recently acquired All Forge Biologics Holdings, LLC shares. The facility was aimed to accelerate production capability for manufacturing and development of biologics. This expansion is expected to foster growth and innovation in Japan
Vaccine Contract Manufacturing Market Report Scope
Report Attribute
Details
Market size value in 2024
USD 2.29 billion
Revenue forecast in 2030
USD 3.60 billion
Growth rate
CAGR of 7.8% from 2024 to 2030
Base year for estimation
2023
Historical data
2018 - 2022
Forecast period
2024 - 2030
Report updated
March 2024
Quantitative units
Revenue in USD million/billion and CAGR from 2024 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Vaccine type, workflow, application, type, product type, phase, scale of operation, service type, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; India; Japan; Australia; Thailand; South Korea; Brazil; Mexico; Argentina; Saudi Arabia; UAE; South Africa, Kuwait
Key companies profiled
Lonza; FUJIFILM Diosynth Biotechnologies; Ajinomoto Co., Inc.; Merck KGaA; Catalent, Inc.; IDT Biologika GmbH; Curia Global, Inc.; ICON plc.; Cytovance Biologics (Hepalink); Syngene International Limited.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Vaccine Contract Manufacturing Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global vaccine contract manufacturing market report based on vaccine type, workflow, application, type, product type, phase, scale of operation, service type, end-use and region:
-
Vaccine Type Outlook (Revenue, USD Million, 2018 - 2030)
-
Attenuated
-
Inactivated
-
Subunit-based
-
Toxoid-based
-
DNA-based
-
-
Workflow Outlook (Revenue, USD Million, 2018 - 2030)
-
Downstream
-
Fill & Finish Operations
-
Analytical & QC studies
-
Packaging
-
-
Upstream
-
Mammalian Expression Systems
-
Bacterial Expression Systems
-
Yeast Expression Systems
-
Baculovirus/Insect Expression Systems
-
Others
-
-
-
Type Outlook (Revenue, USD Million, 2018 - 2030)
-
Viral
-
Bacterial
-
Others
-
-
Product Type Outlook (Revenue, USD Million, 2018 - 2030)
-
Single Vaccines
-
Combination Vaccines
-
-
Phase Outlook (Revenue, USD Million, 2018 - 2030)
-
Preclinical
-
Clinical
-
Commercial
-
-
Application Outlook (Revenue, USD Million, 2018 - 2030)
-
Human Use
-
Veterinary
-
-
Scale of Operation Outlook (Revenue, USD Million, 2018 - 2030)
-
Small Scale
-
Medium Scale
-
Large Scale
-
-
Service Type Outlook (Revenue, USD Million, 2018 - 2030)
-
Fill-Finish
-
Bulk Products
-
-
End-use Outlook (Revenue, USD Million, 2018 - 2030)
-
Biopharmaceutical Companies
-
Research Academies & Government Institutes
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
U.K.
-
Germany
-
France
-
Italy
-
Spain
-
Sweden
-
Norway
-
Denmark
-
-
Asia Pacific
-
Japan
-
China
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global vaccine contract manufacturing market size was estimated at USD 2.14 billion in 2023 and is expected to reach USD 2.29 billion in 2024.
b. The global vaccine contract manufacturing market is expected to grow at a compound annual growth rate of 7.8% from 2024 to 2030 to reach USD 3.60 billion by 2030.
b. North America dominated the vaccine contract manufacturing market with a share of 33.9% in 2023. This is attributable to the widespread manufacturing of vaccines in the region, coupled with the growing presence of a substantial number of biopharmaceutical facilities.
b. Some key players operating in the vaccine contract manufacturing market include Lonza; FUJIFILM Diosynth Biotechnologies Inc.; Ajinomoto Co., Inc.; Merck KGaA; Cytovance Biologics; Catalent, Inc.; IDT Biologika GmbH; ICON plc.; IDT Biologika GmbH; Curia Global, Inc.; and Syngene International Limited
b. Key factors that are driving the market growth include capacity expansion by contract manufacturers for vaccine manufacturing, a global increase in vaccination coverage, and vaccination innovation, and a robust vaccine pipeline.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










