- Home
- »
- Clinical Diagnostics
- »
-
Advanced Cancer Diagnostics Market, Industry Report, 2033GVR Report cover
![Advanced Cancer Diagnostics Market Size, Share & Trends Report]()
Advanced Cancer Diagnostics Market (2025 - 2033) Size, Share & Trends Analysis Report By Test (Tumor Marker Tests, Fine Needle Aspiration Cytology (FNAC) & Biopsy, Imaging-Based Diagnostics, Bone Marrow Aspiration & Biopsy), By End-use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-751-1
- Number of Report Pages: 130
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Advanced Cancer Diagnostics Market Summary
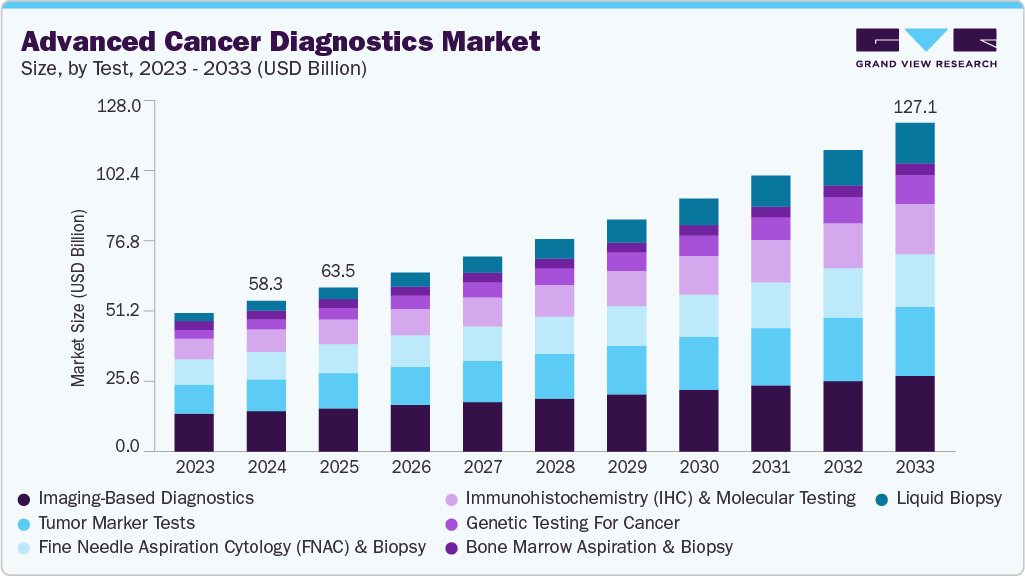
The global advanced cancer diagnostics market size was estimated at USD 58.30 billion in 2024 and is expected to reach USD 127.07 billion by 2033, expanding at a CAGR of 9.05% from 2025 to 2033. The market is driven by several strong factors. Rising global cancer incidence continues to expand demand for early and precise detection, while patient survival benefits from timely diagnosis are pushing adoption of genomic, molecular, and imaging-based tools.
Key Market Trends & Insights
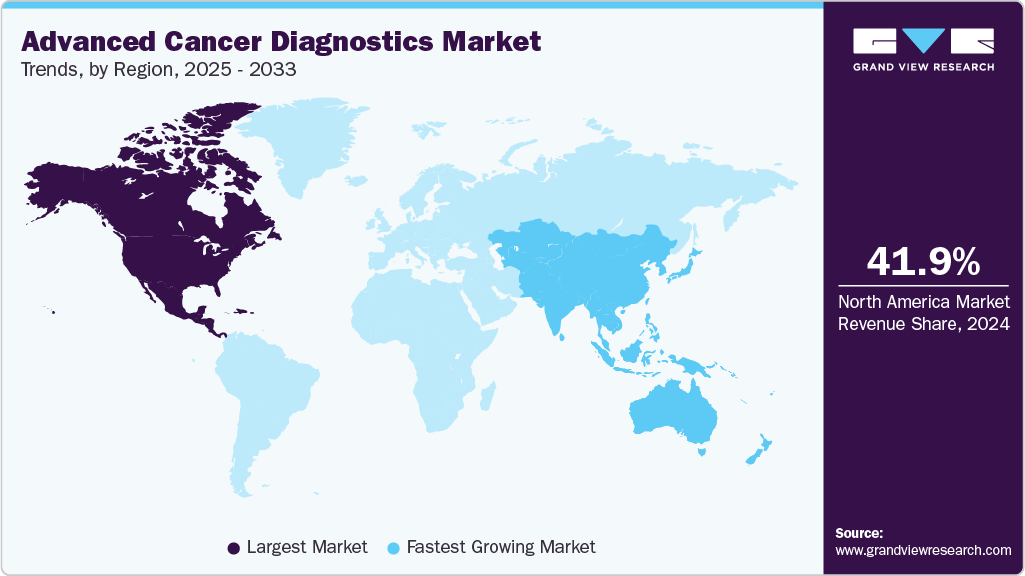
- North America dominated the Advanced cancer diagnostics market, accounting for 41.88% of global revenue in 2024.
- The U.S. remains the most mature and innovation-driven market for advanced cancer diagnostics
- By test, the imaging-based diagnostics segment held the highest market share of 26.77% in 2024.
- Based on end use, the hospitals & cancer specialty centers segment held the highest market share of 45.01% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 58.30 Billion
- 2033 Projected Market Size: USD 127.07 Billion
- CAGR (2025-2033): 9.05%
- North America: Largest market in 2024
- Asia Pacific: Fastest growing market
Breakthroughs such as liquid biopsy, AI-enabled pathology, and biomarker-driven companion diagnostics are accelerating precision oncology, enabling more personalized and effective treatment. Cost-efficiency also acts as a driver, with case studies showing significant healthcare savings when diagnostics reduce late-stage treatments and adverse drug reactions. Alongside this, expanding reimbursement frameworks and large-scale initiatives like ECII are improving access and adoption.The market dynamics for advanced cancer diagnostics are being shaped by rapid technological progress, evolving policy frameworks, and shifting healthcare priorities. Historically, cancer diagnosis and treatment were guided primarily by tumor location and stage, but the advent of advanced diagnostics has changed this paradigm. These new tools provide granular insights into the molecular, genetic, and biological underpinnings of cancer, making precision medicine a tangible reality. By enabling earlier and more accurate diagnosis, advanced diagnostics reduce the chances of missed or incorrect diagnoses and open the door to more personalized treatment selection. This precision minimizes patient exposure to ineffective therapies and their associated side effects, while also supporting real-time monitoring of treatment effectiveness.
The potential benefits extend beyond patient outcomes to health system efficiency. Health systems that integrate advanced diagnostics could see significant cost savings by reducing the need for ineffective treatments and minimizing hospitalizations from adverse drug reactions. One case study from Ireland illustrates this impact: testing colorectal cancer patients for specific genetic variations generated savings of more than USD 55 million per patient, primarily by avoiding severe side effects that would have required hospital care. Similarly, early detection yields strong cost advantages. In breast cancer, early-stage diagnosis has been shown to reduce medical costs by nearly 50%, equivalent to hundreds of millions of USD per patient cohort when compared with late-stage disease.
Technology adoption can also save resources within the health system itself. A U.S. case study demonstrated that the use of digital pathology at an academic medical center delivered savings of USD 1.3 million over five years, while simultaneously improving accuracy and cutting diagnostic review times by more than 30%. Artificial intelligence integration thus emerges as both a clinical and economic catalyst, reshaping workflow efficiency and lowering error rates.
At the same time, barriers to adoption remain significant. Advanced diagnostics are still largely restricted to research facilities or specialist hospitals, meaning equitable access across wider health systems is limited. Regulatory and reimbursement challenges are particularly acute. Traditional frameworks are not well-suited to adaptive technologies like AI-based diagnostics, which continue to evolve after approval. The EU’s TEF-Health initiative, launched in 2023 with funding of nearly USD 66 million, serves as a case study of regulatory innovation, providing a sandbox for real-world testing of new approaches under supervision. Yet, delays tied to the rollout of the In Vitro Diagnostic Medical Devices Regulation (IVDR) highlight the risks of bottlenecks. Analysts estimate that up to 27,400 cancer patients may face delays in clinical trial access, translating into indirect economic and treatment costs of several hundred million USD.
Alternative funding models also reveal the scale of potential benefits. Australia’s reimbursement scheme for hepatitis C, introduced in 2016 with funding of USD 792 million, stands as a landmark case study. The program secured projected productivity gains worth USD 4.1 billion and a net economic benefit of USD 3.8 billion by 2030. Applying such models to oncology diagnostics could unlock sustainable access and uptake, particularly in areas like liquid biopsy and genetic testing.
Infrastructure remains another challenge. In many European health systems, biopsy samples still travel across multiple facilities before reaching final analysis, delaying care. Whole-genome sequencing at diagnosis could streamline this process, but requires large-scale investments. Data-sharing barriers also remain critical. The European Cancer Imaging Initiative (ECII), launched in 2023, provides a case study of collaborative infrastructure, establishing a continent-wide platform for cancer image exchange worth more than USD 55 million. Such efforts aim to break down silos, broaden datasets, and accelerate diagnostic innovation.
On the technology front, Liquid biopsy, as demonstrated in a study from England, detected relapse three to five months earlier than scans and saved USD 72 million for every 100 brain tumor patients tested. Similar advances are being seen with total-body PET imaging, which improves sensitivity 40-fold while lowering scan time and radiation dose, and with multiparametric MRI, which has already spared more than 25% of suspected prostate cancer patients from invasive biopsies, amounting to system-level savings in the hundreds of millions of USD annually.
Artificial intelligence continues to unify these advances. A case study in breast cancer demonstrated AI’s ability to detect high HER2 expression and improve patient stratification, ensuring better treatment matching and delivering system-level efficiencies worth tens of millions of USD per year. Computational pathology further accelerates analysis by up to 50% compared to manual reviews, while maintaining or improving accuracy.
The advanced cancer diagnostics are moving from promise to practice, with case studies across Ireland, the U.S., Australia, and England proving their clinical and economic impact. Yet, gaps in regulation, funding, infrastructure, and equity remain. These dynamics suggest a future defined by both opportunities and challenges. If policymakers and industry leaders act in tandem adapting regulatory frameworks, piloting innovative funding models, investing in digital and diagnostic infrastructure, and ensuring equitable implementation advanced diagnostics will not only transform cancer care but also reshape healthcare economics across the next decade.
Market Concentration & Characteristics
The degree of innovation in advanced cancer diagnostics is exemplified by Guardant Health’s 2025 launch of Guardant360 Tissue. This test combines DNA, RNA, genome-wide methylation data, and AI-powered PD-L1 analysis to deliver a multiomic view of cancer. Built on the Guardant Infinity platform, it reflects how innovation is expanding the role of diagnostics beyond single-biomarker testing. By offering deeper insights into tumor biology, the platform pushes oncology diagnostics toward integrated, AI-driven solutions, highlighting a market where innovation directly fuels precision oncology and patient-centered decision-making.
The level of M&A and collaborative activities is well illustrated by Lucence Health Inc.’s 2021 partnership with Waseda University. The collaboration focused on creating a high-speed liquid biopsy laser-based imaging platform for detecting circulating tumor cells and clusters. Rather than a traditional acquisition, this academic-industry tie-up underscores how partnerships remain a strategic pathway for innovation. Such activities allow companies to leverage complementary expertise, accelerate development timelines, and reduce costs, while expanding competitive advantages. This reflects an industry where alliances are as impactful as direct mergers or acquisitions.
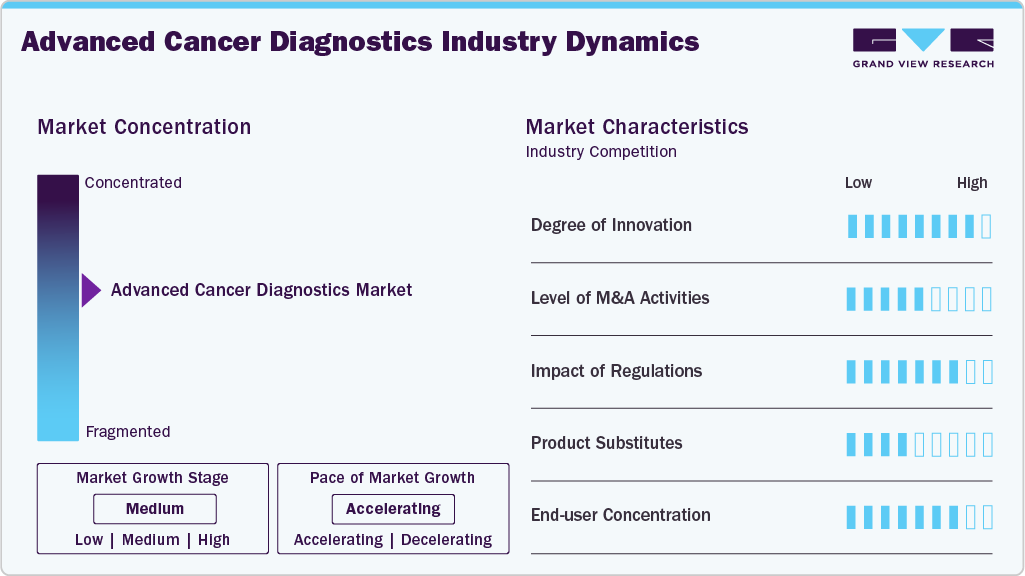
Regulation exerts a profound influence on the advanced cancer diagnostics market. Stringent frameworks such as the EU In Vitro Diagnostic Medical Devices Regulation (IVDR) and the recent EU AI Act directly shape the pace of innovation adoption. While these regulations aim to enhance safety, accuracy, and reliability, they also create bottlenecks, particularly due to shortages of notified bodies and lengthy approval processes. This can delay patient access to cutting-edge tests and restrict clinical trial availability. Companies must invest heavily in validation, compliance infrastructure, and real-world evidence to meet evolving regulatory expectations.
End Use substitutes in cancer diagnostics primarily emerge from the interplay between conventional methods and novel approaches. Traditional tissue biopsies, imaging scans, and single-biomarker tests continue to serve as gold standards. However, substitutes such as liquid biopsies, epigenomic assays, and AI-powered pathology solutions are increasingly gaining traction by offering greater sensitivity, earlier detection, and lower patient burden. These substitutes do not fully replace established modalities but act as complementary tools, creating hybrid diagnostic workflows. Their adoption illustrates a shift towards less invasive, data-rich, and patient-friendly approaches, redefining the competitive dynamics of cancer diagnostics.
End User concentration in the advanced cancer diagnostics market is characterized by a strong reliance on hospitals, cancer specialty centers, and large diagnostic laboratories, which together dominate test adoption. These institutions possess the infrastructure, expertise, and integrated care pathways needed to deploy advanced modalities such as genomic profiling, liquid biopsy, and AI-enabled imaging. While smaller outpatient clinics and academic institutes contribute to niche adoption, the market is heavily skewed toward a concentrated base of high-capacity providers. This concentration amplifies bargaining power among these end users, influencing pricing, test validation, and reimbursement negotiations.
Test Insights
In the advanced cancer diagnostics market, imaging-based diagnostics remain the largest segment contributing the share of 26.77%, supported by their central role in tumor detection, staging, and treatment monitoring. Core modalities like CT, PET/CT, and MRI are well-established, but the segment is evolving with advanced capabilities. The University of Arizona, for instance, received a USD 2 million NIH grant in June 2023 to acquire a new 3Tesla MRI with advanced diffusion and perfusion imaging, enhancing neuro-oncology research. Canon Medical reinforced the trend in April 2024 by launching its Vantage Galan 3T Supreme Edition, integrating AI-driven reconstruction and precision magnet technology to boost clarity and workflow efficiency. Mayo Clinic further advanced adoption with the clinical use of 7-Tesla MRI in October 2024, enabling unprecedented detail in neurological and oncological imaging. Siemens Healthineers built on this momentum with FDA clearance of the MAGNETOM Terra.X 7T scanner in March 2024, introducing ultra-high-field imaging into clinical practice. Similarly, the Paris Brain Institute inaugurated its own 7T MRI in November 2024, expanding Europe’s capabilities in precision neuroimaging. Extending into preclinical applications, Bruker launched 7T and 9.4T Maxwell MRI systems in May 2022, providing helium-free technology for cancer research labs. Together, these initiatives highlight why imaging remains the dominant diagnostic platform-universally adopted, heavily invested in, and continuously innovated.
In contrast, liquid biopsy is the fastest-growing segment, driven by its minimally invasive sampling and real-time insights into ctDNA, cfDNA, and circulating tumor cells. Unlike imaging, which excels at structural visualization, liquid biopsy allows for early detection, minimal residual disease tracking, and therapy resistance monitoring at a molecular level. Its appeal is also economic-reducing the need for invasive biopsies and predicting relapses earlier, thereby cutting downstream costs. With accelerating clinical validation, biopharma partnerships, and its transformative role in multi-cancer early detection, liquid biopsy is widely viewed as the most disruptive force in the next decade of diagnostics. Although conventional tissue biopsies involve invasive methods for the detection of tumor cells, liquid biopsies involve the usage of peripheral blood, which is easily accessible, allowing for more common use, mostly in patients who cannot undergo surgery. As a result, it takes less time for treatment based on tumor detection, enhancing staff & resource efficiency, and is used to screen other diseases. It can also help avoid potential complications associated with conventional biopsies, as it removes the risk of tumor spread, severe bleeding, and injury to surrounding tissue. Thus, liquid biopsy is widely accepted, which is expected to drive the market. Moreover, noninvasive technologies have been adopted in liquid biopsy, which is propelling the adoption of liquid biopsy tests. For instance, in January 2023, OncobiotaLUNG, a liquid biopsy assay, was approved by FDA. It is used for the diagnosis of lung cancer and is capable of determining risk using noninvasive blood tests.
For various applications, such as non-small cell lung cancer, breast, prostate, colorectal, and ovarian cancers, liquid biopsy is used for diagnosis & screening, making it an important tool. After various studies and evaluations, it has been derived that the liquid biopsy technique could provide an improved outcome of diagnosis. Data shows that screening techniques should be used on high-risk patients who have an ancestral history of cancer. Nowadays, community practices and academic centers use liquid biopsy as their complementary technique as compared to standard tissue biopsies, which is expected to drive the market globally. Moreover, over the past several years, studies have shown positive outcomes of liquid biopsy platforms. The government and various regulatory bodies have also shown interest in the area by promoting multiple breakthrough devices for rapid development of the technology. With high adoption of technology by various companies along with the support of government funding and initiatives, the market is expected to grow in the coming years.
End Use Insights
Under the advanced cancer diagnostics market by end use, hospitals and cancer specialty centers hold the largest share of 45.01% in 2024, reflecting their established infrastructure for CT, MRI, PET/CT, and biopsy services, as well as their integration of advanced modalities like 3Tesla MRI and hybrid imaging. These centers lead adoption because of multidisciplinary care pathways that directly link diagnostics with oncology treatment, and strong reimbursement systems that reinforce reliance on hospital-based platforms. Hospitals and cancer specialty centers are expected to dominate the market owing to high market penetration and procedure volume. Increasing favorable government initiatives to provide various services, such as reimbursement for companion diagnostics, are among the major factors anticipated to drive the market. For instance, in February 2023, Lucence Health, Inc. announced that Palmetto GBA has finalized a foundational Local Coverage Determination (LCD) for the LiquidHALLMARK assay through its laboratory technology assessment group for the Liquid Biopsy Services program (MolDX). Moreover, numerous healthcare institutions collaborate with laboratories to integrate different tests, such as NGS and PCR.
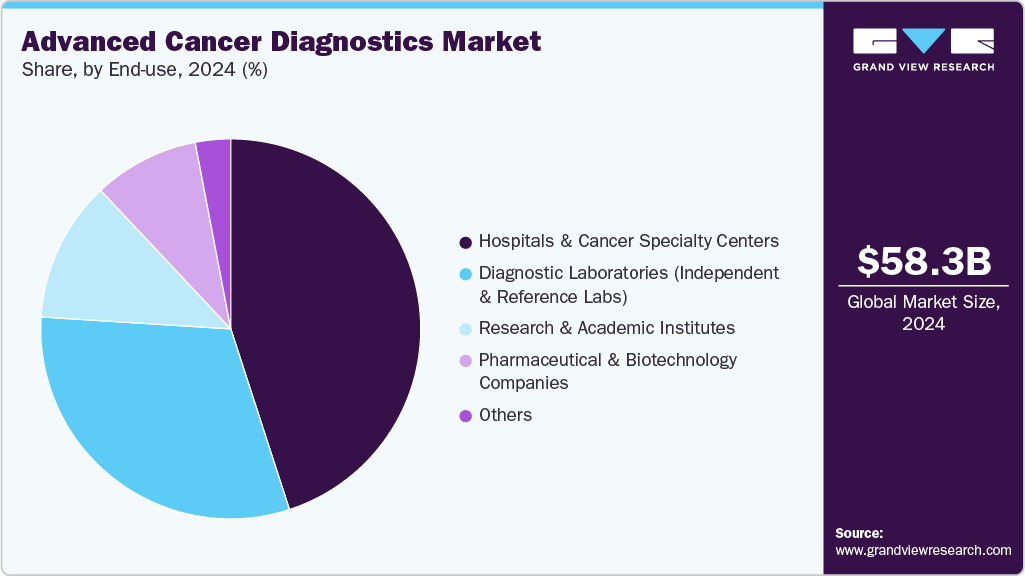
At the same time, diagnostic laboratories are projected to be the fastest-growing segment. Their expansion is fueled by the rising outsourcing of molecular and genetic testing, as hospitals increasingly collaborate with specialized labs for liquid biopsy, NGS, and companion diagnostics. With their scalability, faster turnaround times, and ability to process high test volumes across geographies, diagnostic labs are becoming indispensable to the adoption of next-generation diagnostics.
Regional Insights
North America dominated the Advanced cancer diagnostics market, accounting for 41.88% of global revenue in 2024, supported by advanced healthcare infrastructure, widespread access to MRI and PET-CT systems, and strong reimbursement frameworks.
U.S. Advanced Cancer Diagnostics Market Trends
The U.S. remains the most mature and innovation-driven market for advanced cancer diagnostics, with widespread adoption of genomic profiling, liquid biopsy, and AI-enabled imaging. Strong FDA support for companion diagnostics and early approvals like multi-cancer detection assays ensure continuous innovation. Hospitals and specialty centers dominate, while independent labs increasingly handle outsourced molecular testing, making the U.S. a global reference point for precision oncology integration.
Europe Advanced Cancer Diagnostics Market Trends
The European advanced cancer diagnostics market is expanding steadily, driven by rising cancer incidence, aging populations, and strong demand for early detection solutions. Adoption of next-generation sequencing, liquid biopsy, and AI-based imaging is accelerating, supported by precision oncology initiatives and favorable reimbursement policies in countries like Germany, the UK, and France. Collaborations between biotech firms and healthcare providers are strengthening diagnostic accessibility. However, high costs and varied regulatory frameworks pose challenges. Overall, Europe is a key growth hub, emphasizing personalized medicine and integrated cancer care.
The UK advanced cancer diagnostics market is shaped by the NHS focus on early detection and national cancer screening programs. Digital pathology rollouts and integration of AI-powered reporting systems are enhancing efficiency. Adoption of liquid biopsy platforms is growing, especially for breast and ovarian cancers, while molecular testing is central to precision medicine initiatives. Research-clinical collaborations strengthen innovation, making the UK a European leader in advanced oncology diagnostics.
The Germany advanced cancer diagnostics market leads Europe in advanced cancer diagnostics due to strong reimbursement frameworks, widespread hospital infrastructure, and rapid adoption of molecular and imaging-based tests. Clinical use of NGS and AI-driven pathology platforms is expanding, supported by collaborations with biopharma companies. Germany is also a hub for clinical trials, making it a critical market for biomarker validation and next-generation diagnostic tool deployment.
Asia Pacific Advanced Cancer Diagnostics Market Trends
Asia Pacific is witnessing the fastest growth in adoption of advanced cancer diagnostics, fueled by rising cancer prevalence, rapid healthcare infrastructure expansion, and growing government initiatives. China is advancing AI-powered cancer screening and liquid biopsy pilots, while Japan focuses on precision oncology and insurance-backed genomic testing. Cost-effective tumor marker tests remain vital in developing countries, but the region is steadily shifting toward molecular and imaging-driven approaches.
The China advanced cancer diagnostics market is emerging as a global innovation hub in advanced cancer diagnostics, with strong government-led cancer screening programs and partnerships between local biotech firms and global companies. AI-enhanced radiology, affordable liquid biopsy solutions, and tumor marker tests dominate adoption. Increasing investment in multi-cancer early detection platforms highlights the country’s ambition to lead in population-scale cancer prevention and management.
The Japan advanced cancer diagnostics market is driven by precision medicine policies, strong adoption of NGS, and nationwide insurance coverage for genomic testing. Hospitals and research institutes are deeply engaged in biomarker discovery, especially for lung, gastric, and ovarian cancers. Liquid biopsy technologies are advancing, with a growing role in therapy selection and recurrence monitoring. Japan’s expertise in high-end imaging further strengthens its position as a regional leader.
Latin America Advanced Cancer Diagnostics Market Trends
Latin America is gradually expanding its advanced diagnostics capabilities, led by Brazil and Mexico. The growing cancer burden and investment in oncology infrastructure are driving the adoption of imaging and molecular tests. While accessibility remains uneven, diagnostic laboratories are rapidly scaling genetic testing and liquid biopsy services. Public health screening initiatives and partnerships with international companies are supporting wider use of precision diagnostics across the region.
Brazil anchors Latin America’s advanced diagnostics market due to its large patient base and expanding oncology networks. Hospitals remain central to imaging-based diagnostics, while independent labs are increasingly offering molecular and liquid biopsy services. Collaborations with global diagnostic firms are helping accelerate technology transfer, and government-backed cancer screening programs are improving early detection in urban areas.
Middle East & Africa Advanced Cancer Diagnostics Market Trends
The Middle East & Africa market is developing quickly, with GCC countries driving demand through significant investments in oncology centers and imaging infrastructure. AI-based radiology and PET/CT systems are gaining traction in Saudi Arabia and the UAE, while Africa is slowly building diagnostic capacity. Regional cancer centers in Egypt and South Africa play a pivotal role in adopting advanced testing for clinical care and research.
The Saudi Arabia advanced cancer diagnostics market is expected to propel due to the country’s focus on healthcare modernization under Vision 2030 is propelling the adoption of molecular diagnostics, advanced imaging, and liquid biopsy. Hospitals are rapidly integrating genomic and imaging-based tools for precision oncology, supported by international partnerships. National cancer screening programs and infrastructure upgrades are reinforcing demand for cutting-edge cancer diagnostics.
The UAE advanced cancer diagnostics market is positioning itself as a hub for advanced cancer diagnostics in the Middle East. Strong private healthcare investment, clinical trial collaborations, and rapid adoption of genomic sequencing and digital pathology are shaping the landscape. The country is leveraging AI-powered diagnostic platforms and global partnerships to expand access, while government-driven initiatives are strengthening early detection and patient stratification programs.
Key Advanced Cancer Diagnostics Company Insights
The major market players operating are focused on adopting strategic initiatives such as launches, mergers & acquisitions, partnerships, etc. Furthermore, several players are focusing on the development of testing services, thereby boosting the demand.
Key Advanced Cancer Diagnostics Companies:
The following are the leading companies in the advanced cancer diagnostics market. These companies collectively hold the largest market share and dictate industry trends.
- Roche Diagnostics
- Illumina, Inc.
- Thermo Fisher Scientific
- Siemens Healthineers
- GE HealthCare
- Philips Healthcare
- Canon Medical Systems
- Fujifilm Holdings Corporation
- Agilent Technologies
- Bruker Corporation
- Becton, Dickinson and Company (BD)
- Bio-Rad Laboratories
- Myriad Genetics
- F. Hoffmann-La Roche Ltd.
Recent Developments
-
In March 2021, Lucence Health Inc. partnered with Waseda University to develop a high-speed liquid biopsy laser-based imaging platform for early cancer detection. The collaboration aims to enable automated identification of circulating tumor cells and clusters, advancing liquid biopsy efficiency.
-
In April 2025, Guardant Health launched Guardant360 Tissue, the first comprehensive genomic profiling test for tumor tissue that integrates DNA, RNA, AI-powered PD-L1 analysis, and genome-wide methylation data. Built on the Guardant Infinity smart liquid biopsy platform, it delivers a multiomic view to support precision oncology.
-
On June 12, 2023, the University of Arizona secured a $2 million NIH High-End Instrumentation grant to acquire an advanced 3T MRI system from Siemens Healthineers, strengthening research in brain imaging with faster, higher-resolution diagnostics.
-
In April 2024, Canon Medical released the Vantage Galan 3T / Supreme Edition MRI, integrating advanced AI solutions like AiCE and PIQE to enhance image clarity, shorten scan times, and improve productivity in high-demand oncology settings.
-
In December 2024, United Imaging showcased several breakthroughs at RSNA 2024, including AI-driven MRI, molecular imaging, and interventional X-ray systems, reinforcing its mission of “Equal Healthcare for All.”
-
In October 2024, Mayo Clinic advanced clinical adoption of 7-Tesla MRI, highlighting its role in epilepsy and neuro-oncology with unprecedented image resolution, improving diagnosis of subtle brain abnormalities.
-
In March 2024, Siemens Healthineers achieved FDA clearance for MAGNETOM Terra.X 7T MRI, the world’s first clinical eight-channel parallel transmit scanner, delivering ultra-high-resolution brain and knee imaging with deep learning reconstruction.
-
In November 2024, the Paris Brain Institute inaugurated a Siemens MAGNETOM Terra.X 7T MRI, marking France’s fifth ultra-high-field MRI and the only one embedded within a university hospital for advanced neuroscience and oncology applications.
-
In May 2022, Bruker Corporation introduced 7T and 9.4T Maxwell preclinical MRI systems, eliminating the need for helium refills and providing high-field sensitivity for oncology, neurology, and cardiology research.
Advanced Cancer Diagnostics Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 63.54 billion
Revenue forecast in 2033
USD 127.07 billion
Growth rate
CAGR of 9.05% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in (USD billion/million) and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Test, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; Germany; UK; France; Spain; Italy; Denmark; Norway; Sweden; Japan; China; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Roche Diagnostics; Illumina, Inc.; Thermo Fisher Scientific; Siemens Healthineers; GE HealthCare; Philips Healthcare; Canon Medical Systems; Fujifilm Holdings Corporation; Agilent Technologies; Bruker Corporation; Becton, Dickinson and Company (BD); Bio-Rad Laboratories; Myriad Genetics, F. Hoffmann-La Roche Ltd.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Global Advanced Cancer Diagnostics Market Report Segmentation
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2021-2033. For the purpose of this report, Grand View Research has segmented the global advanced cancer diagnostics market report on the basis of test, end use, and region:
-
Test Outlook (Revenue, USD Billion, 2021 - 2033)
-
Tumor Marker Tests
-
Fine Needle Aspiration Cytology (FNAC) & Biopsy
-
Imaging-Based Diagnostics
-
Bone Marrow Aspiration & Biopsy
-
Immunohistochemistry (IHC) & Molecular Testing
-
Genetic Testing for Cancer
-
Liquid Biopsy
-
-
End Use Outlook (Revenue, USD Billion, 2021 - 2033)
-
Hospitals & Cancer Specialty Centers
-
Diagnostic Laboratories (Independent & Reference Labs)
-
Research & Academic Institutes
-
Pharmaceutical & Biotechnology Companies
-
Others
-
-
Regional Outlook (Revenue, USD Billion, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global advanced cancer diagnostics market size was estimated at USD 58.30 billion in 2024 and is expected to reach USD 63.54 billion in 2025.
b. The global advanced cancer diagnostics market is expected to grow at a compound annual growth rate of 9.05% from 2025 to 2033 to reach USD 127.07 billion by 2033.
b. North America dominated the advanced cancer diagnostics market with a share of 41.88% in 2024. This is attributable to advanced healthcare infrastructure, widespread access to MRI and PET-CT systems, and strong reimbursement frameworks.
b. Some key players operating in the advanced cancer diagnostics market include Roche Diagnostics, Illumina, Inc., Thermo Fisher Scientific, Siemens Healthineers, GE HealthCare,Philips Healthcare, Canon Medical Systems, Fujifilm Holdings Corporation, Agilent Technologies, Bruker Corporation , Becton, Dickinson and Company (BD), Bio-Rad Laboratories, and Myriad Genetics.
b. Key factors that are driving the market growth include rising global cancer burden and technological developments such as liquid biopsy, AI-enabled pathology, and biomarker-driven companion diagnostics which are accelerating precision oncology, and enabling more personalized and effective treatment
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










