- Home
- »
- Clinical Diagnostics
- »
-
At-home Chronic Disease Biomarker Testing Market Report, 2033GVR Report cover
![At-home Chronic Disease Biomarker Testing Market Size, Share & Trends Report]()
At-home Chronic Disease Biomarker Testing Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Test Kits/Devices, Consumables & Reagents), By Application (Diabetes, Cardiovascular Diseases), By Technology, By Distribution Channel, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-885-5
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2026 - 2033
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
At-home Chronic Disease Biomarker Testing Market Summary
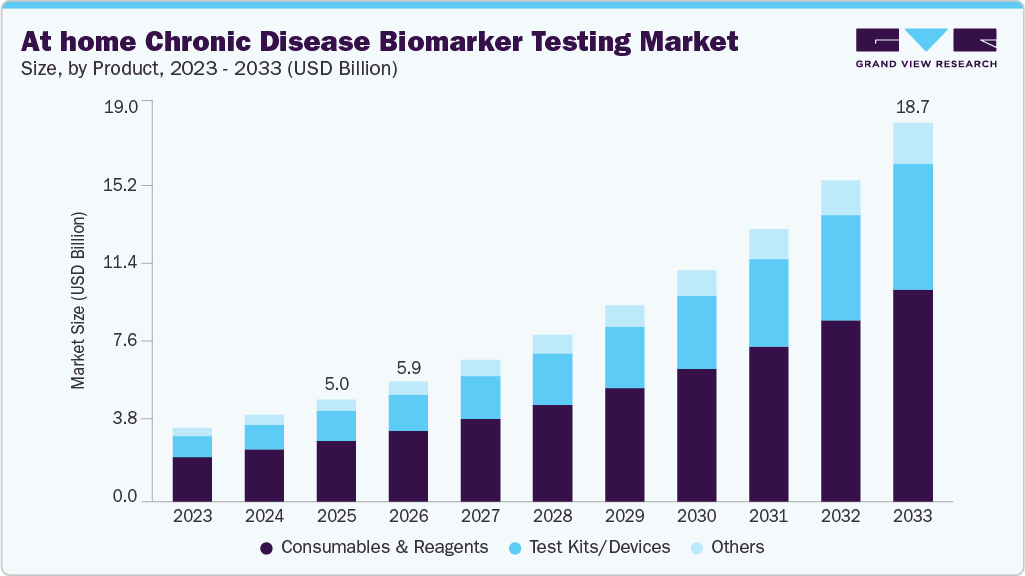
The global at-home chronic disease biomarker testing market size was estimated at USD 5.04 billion in 2025 and is projected to reach USD 18.68 billion by 2033, growing at a CAGR of 17.79% from 2026 to 2033. This growth is driven by the rising prevalence of chronic diseases, an increasing emphasis on preventive and personalized healthcare, and the rapid adoption of decentralized diagnostic solutions.
Key Market Trends & Insights
- North America's at-home chronic disease biomarker testing industry dominated the global market and accounted for the largest revenue share of 44.07% in 2025.
- The U.S. led the North American market and held the largest revenue share in 2025.
- By product, the consumables and reagents segment dominated the global market and accounted for the largest revenue share of 59.51% 2025.
- By application, the diabetes segment held the largest revenue share of 39.47% in 2025.
- By technology, the lateral flow assays (LFA) segment held the largest revenue share of 49.20% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 5.04 Billion
- 2033 Projected Market Size: USD 18.68 Billion
- CAGR (2026-2033): 17.79%
- North America: Largest market in 2025
In addition, ongoing advancements in wearable technologies and digital biomarkers are further accelerating market demand. For instance, a 2023 study developed a microfluidics-based POCT platform integrated with an oxygen-sensitive membrane and smartphone detection for simultaneous measurement of multiple chronic disease biomarkers. The system demonstrated clinical-grade sensitivity, with results consistent with commercial laboratory kits.The growth of the at-home chronic disease biomarker testing industry is driven by the rising prevalence of chronic diseases. Chronic conditions require continuous monitoring, creating a strong demand for accessible diagnostic solutions that patients can use at home. For instance, according to the World Health Organization (WHO), chronic diseases account for the majority of global deaths, and premature mortality from these conditions continues to rise due to population growth and aging. Globally, more than 830 million were living with diabetes in the year 2022, a number that has more than doubled over the past 30 years, reflecting the rapid expansion of chronic diseases across both developed and developing regions. The economic burden of chronic diseases further fuels demand for home-based testing, as these conditions require long-term treatment, frequent hospital visits, and ongoing diagnostic monitoring, all of which increase healthcare costs. Home-based biomarker testing enables early detection, continuous monitoring, and better disease management, helping reduce hospitalizations and overall expenses. In India, programs such as the National Program for Prevention and Control of Cancer, Diabetes, Cardiovascular diseases (lipids, CRP), and Stroke (NPCDCS) and investments in digital health infrastructure are further supporting the adoption of at-home testing solutions, making them a practical and cost-effective alternative to traditional healthcare delivery models.
The growing emphasis on preventive and personalized healthcare is a major factor driving the expansion of the market. Healthcare is shifting from a reactive, treatment-focused approach to a proactive model that prioritizes early detection, risk assessment, and individualized care. Preventive healthcare encourages regular monitoring of key biomarkers, enabling patients and healthcare providers to identify potential health issues before they progress to severe conditions. Personalized healthcare further enhances this trend by tailoring treatment and monitoring plans to an individual’s unique physiological, genetic, and lifestyle profile. Recent initiatives, including digital health programs and wearable device integration, support continuous health monitoring and data-driven personalized care. For instance, according to a recent article, in 2025, remote patient monitoring platforms will provide actionable insights from home-based biomarker data, empowering both patients and clinicians. This combination of preventive and personalized strategies increases patient engagement, reduces complications, and lowers healthcare costs, thereby accelerating the adoption of at-home biomarker testing solutions and significantly expanding the market.
The rapid adoption of decentralized diagnostic solutions is a key driver accelerating the growth of the market. Decentralized diagnostics shift healthcare delivery from hospitals and laboratories directly to patients’ homes, enabling convenient, timely, and continuous monitoring of chronic conditions. This approach reduces dependency on centralized healthcare facilities, which often face capacity constraints, long waiting times, and accessibility challenges, particularly in rural or underserved areas. At-home biomarker testing kits and remote monitoring devices allow patients to measure critical health parameters such as glucose levels, blood pressure, heart rate, and respiratory function without visiting a clinic, making disease management more efficient and patient-centric. The trend is further reinforced by advancements in digital health platforms and telemedicine, which integrate test results into real-time monitoring systems, enabling healthcare providers to track patient health remotely and adjust treatment plans promptly. Government initiatives and private investments are supporting the expansion of decentralized healthcare infrastructure, making at-home testing solutions more reliable, secure, and widely available. By improving accessibility, convenience, and care efficiency, decentralized diagnostic solutions are driving greater adoption of home-based biomarker testing, reducing healthcare costs, and enabling proactive management of chronic diseases, ultimately fueling sustained market growth.
Ongoing advancements in wearable technologies and digital biomarkers are expanding the at‑home chronic disease biomarker testing industry by enabling continuous, real‑time health monitoring outside clinical settings. Wearable biosensorssuch as smartwatches and patchesnow collect physiological data, including ECG, heart rate, and interstitial glucose, that can be translated into actionable digital biomarkers for chronic disease management. For Instance, the FDA has authorized multiple sensor‑based devices for at‑home use, including Dexcom’s G7 Continuous Glucose Monitoring System and Apple’s Hypertension Notification Feature, underscoring regulatory support for clinical‑grade wearables that empower patients to track health indicators at home. These technologies support early detection, personalized care, and remote clinician engagement, driving broader adoption of at‑home testing.
Market Concentration & Characteristics
The at-home chronic disease biomarker testing industry demonstrates a high degree of innovation, driven by advancements in wearable technology, digital biomarkers, and remote monitoring platforms. For instance, modern devices, such as the Dexcom G7 Continuous Glucose Monitoring System and the Apple Watch ECG feature, enable real-time, clinical-grade monitoring of chronic conditions from home. These wearables leverage sensor miniaturization, microfluidics, and optical technologies to capture vital health data non-invasively, improving patient compliance and comfort. Collected data generates digital biomarkers, which, combined with AI-powered analytics, provide predictive insights, early detection of complications, and personalized treatment guidance. Integration with telemedicine platforms allows clinicians to remotely monitor patient health, deliver timely interventions, and reduce unnecessary hospital visits. FDA approvals of such devices demonstrate regulatory validation of both accuracy and safety for home use. By combining innovative hardware, digital intelligence, and connectivity, the market is redefining chronic disease management, empowering patients, and fostering preventive, personalized healthcare.
Mergers, acquisitions, and strategic partnerships in the market space are moderate, driving growth and innovation in the market by enabling companies to expand product portfolios, integrate technologies, and scale operations. For instance, Glooko’s acquisition of Monarch Medical Technologies strengthened its remote diabetes management capabilities, bridging hospital and home care. Strategic deals are also evident at the industry level, such as Danaher’s $9.9 billion acquisition of Masimo, enhancing non-invasive patient monitoring and diagnostics. Rising M&A activity in digital health and connected care reflects the market’s focus on consolidation, innovation, and the development of integrated at-home solutions for chronic disease management.

The regulatory impact on the at-home chronic disease biomarker testing industry is high. Regulations play a critical role in shaping the market by ensuring the safety, accuracy, and reliability of diagnostic devices. Regulatory approvals, such as those from the U.S. Food and Drug Administration (FDA) and the European CE marking, confirm that at-home devices meet clinical performance and data-accuracy standards, which builds patient and clinician trust. FDA-authorized devices like Dexcom G7 Continuous Glucose Monitor and Apple Watch ECG demonstrate regulatory support for home-based chronic disease monitoring, facilitating market adoption. Compliance with data privacy laws, such as HIPAA in the U.S. and GDPR in Europe, ensures secure handling of sensitive health information collected by connected wearables and apps. In addition, evolving guidelines on digital biomarkers and telehealth integration encourage innovation while safeguarding patient safety. Overall, regulatory frameworks act as both enablers and quality checkpoints, accelerating the adoption of at-home biomarker testing while maintaining high standards of care.
Product expansion in the at‑home chronic disease biomarker testing industry is intensifying as companies introduce innovative tools that broaden diagnostic capabilities and user accessibility. For Instance, in Oct 2025, ProDx Health launched a nationwide hormone testing service as a wearable. ProDx Health’s nationwide hormone testing service, which enables consumers to collect samples at home and receive clinical hormone biomarker resultsincluding testosterone, cortisol, and thyroid levels—within 48 hours, without needing a doctor’s appointment or insurance authorization. This expansion bridges the gap between wearable health data and actionable clinical testing by offering rapid, comprehensive biomarker insights that complement consumer devices like smartwatches and fitness trackers, which often monitor trends but do not provide causal biochemical analysis. Meanwhile, wearable technology firms are broadening offerings with advanced sensors capable of tracking lactate, glucose trends, and environmental health parameters in daily life, signaling a shift toward multifunctional at‑home diagnostics. These product extensions enhance patient engagement, support preventive care, and drive wider adoption of home‑based chronic disease management tools.
The regional expansion of the at-home chronic disease biomarker testing industry is moderate, with strong adoption in North America and Europe and growing uptake in Asia-Pacific and the Middle East, driving growth in the market as companies enter emerging markets with rising chronic disease prevalence. For example, Roche and Abbott have expanded their home diagnostics and remote monitoring solutions in India, Brazil, and Southeast Asia, targeting diabetes and cardiovascular disease management. These initiatives increase accessibility, address local healthcare gaps, and tap into the growing demand for home-based testing solutions in underserved regions.
Product Insights
The consumables and reagents segment dominated the market, accounting for the largest revenue share of 59.51% in 2025. The segment’s dominance is primarily attributed to the high frequency of testing, as consumables such as test kits, reagents, lancets, and collection materials are used repeatedly in at-home chronic disease biomarker testing. In addition, ongoing innovations in assay chemistry and user-friendly sample collection systems have increased adoption across both D2C and clinical channels. Strong demand for reliable, validated reagents from established laboratories and telehealth providers further supports revenue growth. The segment also benefits from the expansion of subscription-based home testing models and preventive care programs, which drive recurring purchases of consumables. Overall, the critical role of these materials in enabling accurate, convenient, and scalable at-home diagnostics underpins the segment’s leading market position.
The test kits/devices segment is projected to grow at the fastest rate of 19.68% during the forecast period. These kits now cover a wider range of biomarkers, assessing multiple chronic disease indicators via blood, saliva, and urine samples, reflecting advances in biotechnology and the integration of digital health. Providers are increasingly integrating AI-powered analytics and telehealth services to offer predictive insights and personalized care recommendations, enhancing the clinical value of at-home devices. The shift toward more comprehensive, user-friendly kits with better result interpretation and broader distribution is a key driver of this rapid growth. For instance, platforms like FutureWell offer customizable self-testing solutions that track up to 17 biomarkers across metabolic and chronic disease areas, delivering richer health insights from a single device. This exemplifies the innovation fueling the accelerated expansion of the test kits/devices segment.
Application Insights
In 2025, the adult diabetes segment accounted for the largest market share of 39.47% and is anticipated to grow over the forecast period. The segment’s dominance is attributed to rising global diabetes prevalence and increasing demand for regular monitoring and early detection. According to the World Health Organization (WHO), 830 million people were living with diabetes in 2022. Diabetes diagnostic tools, such as HbA1c tests, are key drivers of this dominance, with the global HbA1c testing market poised to expand significantly as technologies like point‑of‑care and automated analyzers improve accessibility and accuracy in both clinical and at‑home settings. Recent health initiatives promoting regular diabetes screening and self-management, including expanded guidelines for frequent glycemic monitoring and support for home testing devices, are indeed accelerating the adoption of digital diabetes technologies.
The cancer (tumor markers) segment is likely to grow fastest over the forecast period. Growth is driven by the rising global incidence of cancer, growing demand for early detection and preventive screening, and technological advancements in high-sensitivity assays and at-home testing kits. Awareness campaigns and government initiatives promoting regular cancer screening are encouraging wider adoption of home-based diagnostics. Furthermore, integrating digital health platforms, AI-powered predictive analytics, and multi-marker testing panels enhances the accuracy, convenience, and clinical relevance of tumor marker tests. As patients increasingly prioritize early intervention and personalized monitoring, the cancer biomarker segment is projected to surpass other chronic disease categories in both revenue growth and market penetration, reflecting a shift toward proactive, technology-enabled cancer management.
Technology Insights
In 2025, the independent lateral flow assays (LFA) segment led the market, accounting for 49.20% of revenue, and is expected to grow steadily over the forecast period. The segment’s dominance is primarily driven by the simplicity, rapid results, affordability, and ease of use of LFA-based devices, which allow consumers to perform tests at home without specialized training or equipment. LFA kits are widely used for monitoring chronic conditions such as diabetes, cardiovascular disease, and infectious complications, offering convenience and real-time feedback. The market is further supported by technological advancements improving assay sensitivity, multiplexing capabilities, and integration with digital health platforms for tracking and teleconsultation. In addition, government initiatives promoting preventive care and early detection, along with the rising adoption of OTC and point-of-care diagnostics, are driving growth in the LFA segment, making it a critical driver of the overall at-home biomarker testing market.
The Next-Generation Sequencing (NGS) segment is expected to register the fastest CAGR over the forecast period. This rapid expansion is driven by the growing demand for comprehensive genomic profiling, early disease detection, and personalized medicine, particularly in chronic diseases and oncology. NGS enables simultaneous analysis of multiple biomarkers, providing deeper insights into disease risk, progression, and treatment response that are not possible with traditional single-marker tests. Advances in high-throughput sequencing technologies, cost reduction, and integration with AI-driven data analysis are further enhancing its accessibility and clinical utility. In addition, increasing adoption of NGS in at-home and decentralized testing platforms, coupled with supportive regulatory frameworks in North America, Europe, and parts of the Asia Pacific, is fueling market growth. As consumers and healthcare providers seek more precise, predictive, and actionable health insights, the NGS segment is poised to outpace other biomarker testing technologies in both revenue and adoption.
Distribution Channel Insights
In 2025, the retail pharmacies segment accounted for 40.11% of the market and is expected to grow steadily over the forecast period. The segment’s dominance is driven by the widespread availability of pharmacies, consumer trust, and the convenience of in-store purchases, making it a preferred channel for at-home testing kits and related products. Pharmacies not only provide physical access but also support (OTC) distribution, product demonstrations, and pharmacist guidance, enhancing consumer confidence in self-testing. The growth of subscription-based home testing kits and increasing partnerships between pharmacies, telehealth providers, and diagnostic companies are further bolstering adoption. In addition, government initiatives promoting preventive healthcare, chronic disease management, and early detection are increasing the demand for easily accessible testing solutions. The integration of pharmacies with digital health platforms and app-based result delivery is also driving segment growth, ensuring that retail pharmacies remain a key distribution channel for at-home biomarker testing.
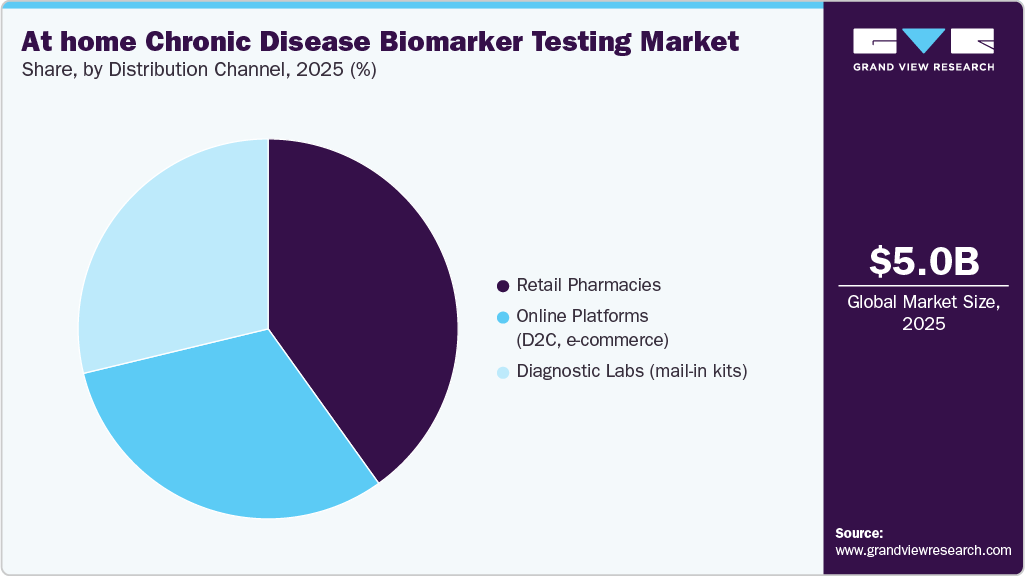
The online platforms segment is anticipated to grow at the fastest rate over the forecast period. This rapid growth is driven by the increasing preference for convenience, contactless, and discreet access to at-home chronic disease biomarker testing kits. Consumers can easily order tests online, receive kits at home, and access results digitally, often accompanied by telehealth consultations and AI-driven insights. The expansion of e-commerce infrastructure, mobile health applications, and subscription-based testing models is further fueling adoption. In addition, the COVID-19 pandemic accelerated consumer comfort with online healthcare solutions, normalizing the use of digital channels for preventive health and chronic disease management. Online platforms also enable broader geographic reach, including underserved or remote areas, and facilitate partnerships with laboratories and diagnostics providers. As patients increasingly prioritize convenience, rapid results, and integrated digital support, online channels are expected to outpace traditional distribution channels such as retail pharmacies, making them a key growth driver for the at-home biomarker testing market.
Regional Insights
North America at- home, chronic disease biomarker testing industry dominated the global market and accounted for 44.07% of revenue share in 2025. Growth in the region is supported by a combination of high disease burden, strong consumer readiness, advanced infrastructure, and supportive regulation. The prevalence of diabetes, cardiovascular disease, and cancer drives demand for tests like HbA1c and lipid panels, enabling continuous monitoring and preventive care models. Consumers are comfortable with self-testing and value convenience, privacy, & rapid results. A robust ecosystem, supported by the Food and Drug Administration and Health Canada, ensures high trust through strict validation standards, while insurance and employer programs integrate testing into routine care. Recent initiatives reinforce this leadership. For instance, in 2026, the FDA promoted expanded OTC access and improved usability, and in March 2025, the FDA approved the Teal Wand, the first at-home cervical cancer screening test, highlighting strong regulatory alignment with innovation.
U.S. At home Chronic Disease Biomarker Testing Market Trends
The U.S. at- home, chronic disease biomarker testing industry held the largest share of the North American market in 2025, driven by its high burden of chronic diseases, strong consumer adoption of self-testing, and a well-established digital health ecosystem. Advanced healthcare infrastructure and widespread integration of telehealth platforms enable seamless use of at-home diagnostics. Supportive regulatory oversight from the Food and Drug Administration ensures high-quality, reliable testing, while expanding OTC approvals further boost accessibility. In addition, strong insurance coverage and employer-sponsored wellness programs subsidize testing, encouraging preventive care. The presence of leading diagnostics and digital health companies also accelerates innovation.
Europe At home Chronic Disease Biomarker Testing Market Trends
The Europe at-home chronic disease biomarker testing industry is experiencing significant growth, driven by the rising burden of diabetes, cardiovascular diseases, and cancer, as well as a strong emphasis on preventive healthcare within public systems. Adoption is supported by rising demand for convenient, private testing, though it remains more cautious than in North America. A highly regulated environment under the European Medicines Agency and strict IVDR guidelines ensures high standards of safety, accuracy, and reliability, building strong patient trust. For instance, BD’s December 2025 expansion of IVDR‑certified assays strengthens Europe’s dominance by increasing access to regulatory-compliant, high-quality tests, boosting trust and adoption within public healthcare systems, and reinforcing the region’s focus on clinical rigor and integrated, standardized at-home diagnostics.
The UK industry for at-home chronic disease biomarker testing is expanding due to rising rates of diabetes and cardiovascular conditions, growing consumer demand for convenient testing, and integration with digital health and telemedicine. NHS-backed preventive programs, private healthcare, and employer wellness initiatives support adoption. Home HbA1c kits allow individuals to collect finger-prick samples at home, send them to UKAS-accredited labs, and receive digital results, enabling continuous glucose monitoring without GP visits. This trend highlights the UK’s shift toward patient-centered, technology-enabled chronic disease management.
Germany’s at‑home chronic disease biomarker testing industry is growing as chronic conditions like diabetes and cardiovascular diseases become more prevalent and preventive care gains priority. High consumer health awareness and strong digital health adoption support the uptake of home testing solutions, particularly for markers such as HbA1c, lipids, and metabolic panels. Germany’s robust healthcare infrastructure and universal insurance coverage facilitate integration of at‑home tests with clinical care pathways, often through physician‑ordered kits processed by accredited laboratories. Supportive regulatory frameworks, including the Federal Institute for Drugs and Medical Devices and IVDR compliance, ensure quality and safety, while collaborations between diagnostics firms and healthcare providers enhance accessibility. This positions Germany as one of Europe’s most advanced markets for reliable, regulated at‑home chronic disease
Asia Pacific At home Chronic Disease Biomarker Testing Market Trends
The Asia Pacific at- home, chronic disease biomarker testing industry is projected to witness the fastest growth over the forecast period, due to the rising prevalence of diabetes, cardiovascular diseases, and other lifestyle-related chronic conditions. Rapid urbanization, increasing health awareness, and growing adoption of digital health technologies and telemedicine are driving demand for convenient home-based diagnostics. Expanding middle-class populations with higher disposable incomes and a willingness to pay for preventive care further accelerate market growth. In addition, governments in countries such as China, India, and Japan are promoting early-detection programs and supporting regulatory frameworks for home testing. Limited access to traditional healthcare in rural areas also makes at-home testing an attractive solution, positioning Asia Pacific as the fastest-growing and most opportunity-rich market for chronic disease biomarker testing globally.
China’s at-home chronic disease biomarker testing industry is expanding due to rapid urbanization, rising prevalence of diabetes and cardiovascular diseases, increasing health awareness, growing disposable incomes, and widespread adoption of digital health and telemedicine solutions, making home-based diagnostics more accessible and convenient.
Latin America At home Chronic Disease Biomarker Testing Market Trends
The Latin America at‑home chronic disease biomarker testing industry is growing as the prevalence of diabetes, hypertension, and cardiovascular diseases increases across the region. Rising health awareness, expanding mobile and internet penetration, and a growing middle class are driving demand for convenient and affordable home‑based testing solutions. Limited access to healthcare in rural and underserved areas encourages the adoption of at‑home diagnostics, while telemedicine platforms are supporting remote monitoring and follow‑up care. Regulatory improvements and partnerships between local labs and digital health companies are also enabling broader availability of home testing kits, positioning Latin America as an emerging growth market in chronic disease management.
Middle East and Africa At home Chronic Disease Biomarker Testing Market Trends
The Middle East & Africa market for at‑home chronic disease biomarker testing is expanding due to rising rates of diabetes, cardiovascular diseases, and obesity, coupled with increasing health awareness and demand for preventive care. Growing mobile and internet penetration support the adoption of digital health and telemedicine, making home‑based diagnostics more accessible. Governments in the region are investing in healthcare infrastructure and health technology initiatives, while private healthcare providers and telehealth platforms are partnering to offer convenient home testing solutions. Limited access to traditional healthcare in rural areas further drives interest in at‑home testing, positioning the Middle East & Africa as an emerging growth region for chronic disease monitoring and management.
Saudi Arabia’s at‑home chronic disease biomarker testing industry is expanding as the prevalence of diabetes, cardiovascular disease, and obesity rises across the population. Growing health awareness, increased smartphone and internet use, and strong government support for digital health initiatives under Vision 2030 are driving demand for convenient home‑based diagnostics. Telemedicine adoption is increasing, and partnerships between private healthcare providers and digital health platforms are making it easier for individuals to access and use home testing kits. In addition, investments in healthcare infrastructure and preventive care programs are encouraging early disease detection and long‑term monitoring, positioning Saudi Arabia as one of the fastest‑growing markets for at‑home chronic disease testing in the Middle East.
Key At-home Chronic Disease Biomarker Testing Company Insights
The competitive landscape of the at-home chronic disease biomarker testing industry is characterized by a mix of established diagnostics companies, direct-to-consumer (D2C) brands, tech-enabled device makers, and telehealth platforms. Global leaders such as Abbott Laboratories, Roche Diagnostics, and Thermo Fisher Scientific leverage their extensive lab networks and regulatory expertise to offer reliable chronic disease panels. D2C companies like Everlywell and PrivaPath Diagnostics focus on convenience, fast digital results, and subscription models. Tech-enabled players, including Dexcom and Withings, provide continuous monitoring and AI-driven insights, while telehealth providers such as Roman Health integrate testing with virtual physician support. Regional players in Europe, Asia, and Latin America are expanding via local labs and digital platforms. Competitive differentiation hinges on regulatory compliance, data-driven insights, integration with care pathways, and user experience, making innovation, trust, and hybrid distribution strategies critical for market leadership.
Key At-home Chronic Disease Biomarker Testing Companies:
The following key companies have been profiled for this study on the at-home chronic disease biomarker testing market.
- Everlywell
- SiPhox, Inc.
- PrivaPath Diagnostics
- Thorne
- Quest Diagnostics
- Labcorp
- Thermo Fisher Scientific Inc.
- 23andMe
- Segterra, Inc. (InsideTracker)
- AMR Labs SA
- Hims & Hers Health, Inc.
- niahealth
- FutureWell
Recent Developments
-
In October 2025, a landmark advancement in diabetes prevention was achieved by PocDoc, an UK-based digital health diagnostics company, with the launch of the world’s first smartphone-based test for assessing type 2 diabetes risk, incorporating the gold-standard blood biomarker, HbA1c.
-
In September 2025, Hims & Hers agreed to acquire YourBio Health, developer of the TAP bladeless micro‑needle blood sampling device for at‑home use, enhancing consumer access to biomarker testing without venipuncture.
-
In September, 2025, Glooko acquired Monarch Medical Technologies, integrating the FDA‑cleared EndoTool glycemic management system into its digital diabetes care platform strengthening its hospital‑to‑home chronic disease management offerings.
At-home Chronic Disease Biomarker Testing Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 5.93 billion
Revenue forecast in 2033
USD 18.68 billion
Growth rate
CAGR of 17.79% from 2026 to 2033
Actual Data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, application, technology, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Norway; Denmark; Sweden; China; Japan; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Everlywell; SiPhox, Inc; PrivaPath Diagnostics; Thorne; Quest Diagnostics; Labcorp; Thermo Fisher Scientific Inc.; 23andMe; Segterra, Inc. (InsideTracker); AMR Labs Sa; him’s & Hers Health, Inc.; niahealth; FutureWell
Customization scope
Free report customization (equivalent up to 8 analyst working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global At-home Chronic Disease Biomarker Testing Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis on the latest industry trends and opportunities in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the at-home chronic disease biomarker testing market report based on product, application, technology, distribution channel, and region.
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Test Kits/Devices
-
Consumables & Reagents
-
Others
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Diabetes
-
Cardiovascular diseases (lipids, CRP)
-
Cardiovascular diseases (tumor markers)
-
Others
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Immunoassays
-
Polymerase Chain Reaction (PCR)
-
Next-Generation Sequencing (NGS)
-
Lateral Flow Assays (LFA)
-
Others
-
-
Distribution Channel Outlook (Revenue, USD Million, 2021 - 2033)
-
Online platforms
-
Retail pharmacies
-
Diagnostic labs
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Norway
-
Denmark
-
Sweden
-
Norway
-
Rest of Europe
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
Rest of Asia Pacific
-
-
Latin America
-
Brazil
-
Argentina
-
Rest of Latin America
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Rest of Middle East & Africa
-
Frequently Asked Questions About This Report
b. The global at-home chronic disease biomarker testing market size was estimated at USD 5.04 billion in 2025 and is expected to be USD 5.93 billion in 2026.
b. The global at-home chronic disease biomarker testing market, in terms of revenue, is expected to grow at a compound annual growth rate of 17.79% from 2026 to 2033 to reach USD 18.68 billion by 2033.
b. North America's at-home chronic disease biomarker testing market dominated the global market and accounted for the largest revenue share of 44.07% in 2025.
b. Global leaders such as Abbott Laboratories, Roche Diagnostics, and Thermo Fisher Scientific leverage their extensive lab networks and regulatory expertise to offer reliable chronic disease panels. D2C companies like Everlywell and PrivaPath Diagnostics focus on convenience, fast digital results, and subscription models.
b. The growth is driven by the rising prevalence of chronic diseases, an increasing emphasis on preventive and personalized healthcare, and the rapid adoption of decentralized diagnostic solutions.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










