- Home
- »
- Medical Devices
- »
-
China Continuous Glucose Monitoring Devices Market, 2033GVR Report cover
![China Continuous Glucose Monitoring Devices Market Size, Share & Trends Report]()
China Continuous Glucose Monitoring Devices Market (2025 - 2033) Size, Share & Trends Analysis Report By Product (Standalone CGM Devices, Integrated CGM-Insulin Pump Systems), By Connectivity (Bluetooth, 4G/Cellular), By Indication, By End Use, And Segment Forecasts
- Report ID: GVR-4-68040-656-9
- Number of Report Pages: 75
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
China CGM Devices Market Size & Trends
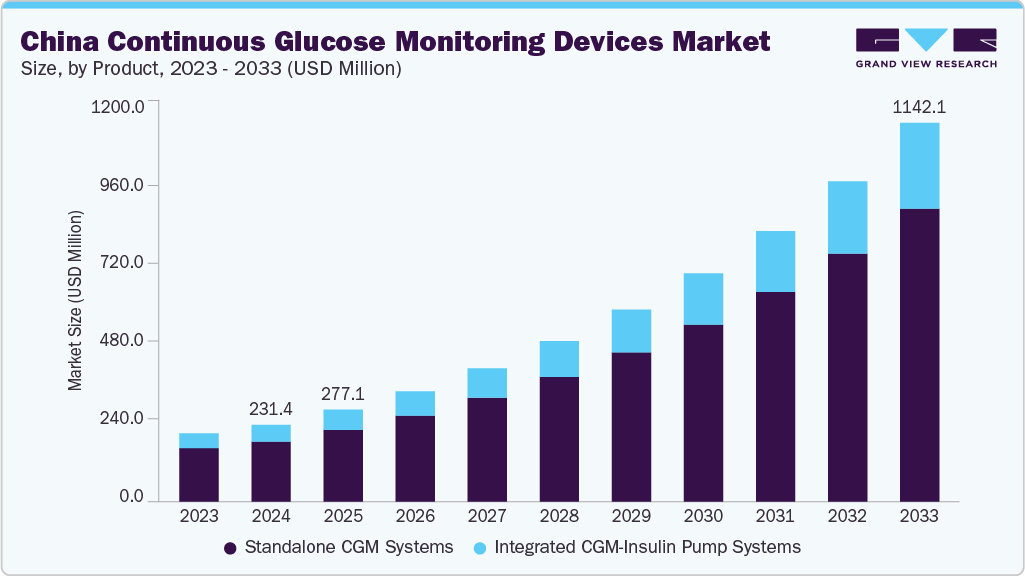
The China continuous glucose monitoring devices market size was estimated at USD 231.4 million in 2024 and is projected to grow at a CAGR of 19.4% from 2025 to 2033 to reach a value of USD 1,142.1 million in 2033. The China continuous glucose monitoring (CGM) devices industry is expanding due to the sharp rise in diabetes cases, driven by urbanization, sedentary habits, and dietary shifts. A 2023 study shows 233 million diabetic patients in China is up 163% since 2005, with prevalence rising from 7.53% to 13.7%.
This growing burden, along with enhanced public health awareness, improved healthcare infrastructure, and government support for advanced monitoring tools, is fueling CGM adoption. Integration with smartphones and wearables adds to user convenience. Manufacturers are investing in cost-effective innovations, and the rising demand for real-time glucose tracking is further boosting market growth across healthcare settings.
Initially developed for managing type 1 and type 2 diabetes, CGMs have gained widespread popularity beyond traditional use, particularly among young individuals seeking better lifestyle and health monitoring. Videos tagged “sugar control” on Douyin, China’s version of TikTok, have garnered over 20.8 billion vie, indicating massive interest. The wearable nature and real-time tracking capabilities of CGM devices have made them appealing to younger, tech-savvy users. Users now rely on CGMs for weight management and dietary planning, illustrating the market's shift toward preventive health behaviors.
According to Sinocare’s Director of CGM Consumer Business for China, sales of CGM needles surged by 63% year-on-year in H2 2024, while Sinocare’s sales doubled, growing by 104% on major platforms like T-mall and JD.com. This reflects both growing public interest and market penetration, especially among youth newly diagnosed or at risk for diabetes. Expert voices add credibility to this trend.
Affiliation
Key Insight
China-Japan Friendship Hospital
CGMs are useful for high-risk individuals, but they advise against unnecessary use by healthy people.
Shanghai Tenth People’s Hospital
Observed an increase in young patients with elevated blood glucose due to poor diets and inactivity.
Sinocare Health
Reinforces the value of CGMs for early diagnosis and urges balanced, periodic use.
Product innovation is another critical growth driver. The iCan CGM system, developed by Sinocare Meditech Inc., recently received regulatory clearance in China and the EU. In a multicenter clinical study involving 60 adults with type 1 and type 2 diabetes, the iCan CGM system demonstrated high accuracy with a Mean Absolute Relative Difference (MARD) of 8.88% and a wear life of 14.76 days. It showed 100% of readings in safe zones on the Surveillance Error Grid, confirming reliability.
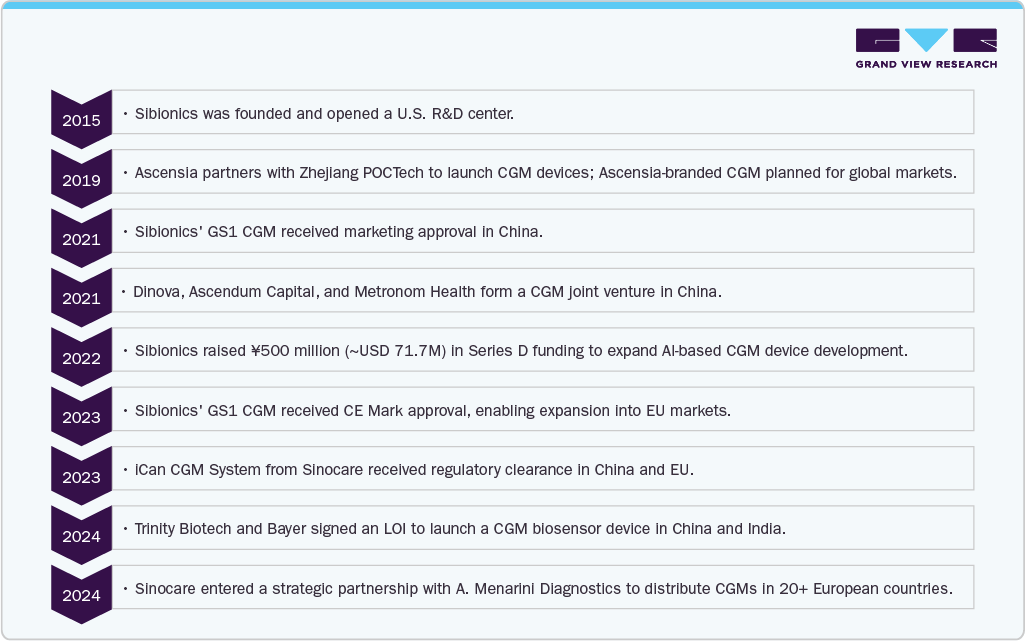
The continuous glucose monitoring (CGM) devices industry in China has witnessed significant growth and strategic developments over the past few years, reflecting a robust trajectory in innovation, market penetration, and international collaboration. Companies like Sibionics and Sinocare have achieved important regulatory milestones, while strategic partnerships with global firms such as Bayer and A. Menarini Diagnostics are enabling expansion into Europe and beyond. These advancements have been fueled by China's rising diabetes prevalence, increasing digital health adoption, and demand for cost-effective, minimally invasive monitoring solutions. With enhanced sensor technologies and international funding, China’s CGM landscape is becoming a global innovation hub. Below are the key past developments since the inception of continuous glucose monitoring (CGM) technologies in China:
The CGM market in China is fueled by a diverse product portfolio from both domestic and international companies. These firms offer a range of devices featuring varied wear times, sensor technologies, and smartphone compatibility to meet growing demand across demographics, enhancing diabetes management through innovation, accessibility, and real-time glucose tracking.
Product Portfolio of Leading CGM Companies in China
Company
Key CGM Device(s)
Launch Year (China)
Key Features
Abbott Laboratories
FreeStyle Libre
2020
Factory-calibrated, 14-day wear, app-based data
Dexcom, Inc.
Dexcom G6, G7
G6: 2021, G7: 2024
Real-time alerts, Apple Watch compatibility
Sinocare Inc.
iCan CGM
2023
15-day wear, high accuracy, CE & China NMPA approved
Medtronic plc
Guardian Connect
N/A
SmartGuard technology, predictive alerts
Ascensia Diabetes Care
POCTech CGM (co-branded)
2019
CE-marked, Bluetooth connectivity
Trinity Biotech
Waveform CGM (planned launch)
2024 (planned)
Biosensor, low-cost, real-time tracking
MicroTech Medical
CGM System (clinical stage)
In trials
Cloud-linked, long-wear design
Meiqi Medical
CGM (under development)
N/A
Focus on affordability and bulk procurement
Yuwell
Yuwell CGM
2022
App-connected, real-time glucose tracking
i-SENS Inc.
CGM under development
To be announced (TBA)
Miniaturized sensor, expanding into China
Over the past two years, China’s CGM (Continuous Glucose Monitoring) market has undergone a major transformation, driven by competitive pricing and aggressive market penetration strategies from domestic manufacturers. In 2022, CGM products in China were typically priced between ¥400 to ¥600 (USD $57-$86), making them less accessible to the broader population. However, by 2023, this pricing landscape changed dramatically as companies like Sinocare and Yuyue Medical slashed prices by nearly 50%, especially during key events like the Double 11 shopping festival. During that period, CGM devices were offered for as low as ¥100 to ¥200 (USD $14-$29), boosting both sales volume and brand visibility.
This strategic pricing approach was coupled with substantial investments in targeted advertising, including elevator media and digital campaigns. As a result, Sinocare reported selling over 100,000 boxes, generating ¥210 million (USD $30 million) in online revenue. Yuyue Medical also saw strong growth, with CGM sales in the millions and a 30% year-on-year increase in glucose meter sales. These aggressive moves disrupted the hold of international brands like Abbott and Medtronic, increasing domestic adoption and localization.
Following this phase of price-led expansion, the market began to stabilize by early 2024, as companies reached an informal consensus to avoid further price wars. Now, leading Chinese CGM manufacturers are shifting focus toward global expansion, particularly in Europe, where several have already obtained CE and MDR certifications. Events like the EASD 2023 became launchpads for these brands to secure partnerships and distributors. This transition signals China's ambition to become a dominant player in global CGM markets through a combination of affordability, innovation, and regulatory compliance.
CGM Device Pricing Trends in China (2022-2023)
Year
Average Price Range (¥ / USD)
Key Players
Notable Strategy/Event
2022
¥400-¥600 (USD 57-USD 86)
Sinocare, Yuyue
Baseline market prices
2023
¥100-¥200 (USD 14- USD 29) - Double 11
Sinocare, Yuyue
Aggressive discounts, online ads
Late 2023
~¥200-¥300 (USD 29-USD 43) - Stabilized
All major domestic firms
Price stabilization post-market consolidation
Market Concentration & Characteristics
The China continuous glucose monitoring devices industry is moderately concentrated, with key players such as Abbott, Dexcom, and Sinocare leading the market. The industry is characterized by rapid technological innovation, rising health awareness, and strong e-commerce adoption. Increasing diabetes prevalence and a growing younger population concerned with wellness are accelerating demand. Domestic companies are emerging with affordable, wearable CGM technologies, intensifying competition. Regulatory support and advancements in sensor accuracy, smartphone integration, and data analytics further shape the market. The sector is evolving quickly, offering significant opportunities for both global and local firms targeting real-time, user-friendly glucose monitoring solutions.
China’s CGM devices industry is experiencing a strong wave of innovation. Researchers have developed a coin-sized CGM sensor using organic electrochemical transistors (OECT), offering enhanced anti-noise ability, sensitivity, and stability in September 2024. This wearable device includes a minimally invasive microneedle array and hydrogel interface, ensuring comfort and performance. In trials, it showed comparable results to the Dexcom G6. The technology operates with low power in aqueous environments, making it ideal for long-term use. Institutions like HKU, Zhejiang University, and Guangzhou Medical University are advancing clinical applications. Chinese firms like SiBionics and POCTech also contribute to this competitive innovation landscape.

China’s continuous glucose monitoring devices industry has witnessed moderate merger and acquisition (M&A) activity, driven by the need for market expansion and technological advancement. In 2023, Abbott strengthened its local presence through a regional acquisition. Trinity Biotech entered a partnership with Bayer after acquiring Waveform’s CGM assets to launch biosensors in China. Earlier, a joint venture involving Dinova Medtech and Metronom Health aimed to introduce innovative CGM technologies. Sinocare also expanded internationally through strategic partnerships. These developments reflect a growing trend of consolidation and collaboration as companies aim to enhance distribution, innovation, and competitiveness in China’s expanding CGM market.
China’s regulatory environment for the continuous glucose monitoring industry is increasingly supportive, promoting innovation while ensuring safety. The National Medical Products Administration (NMPA) requires clinical trials and device registration, classifying CGMs as medical devices. Recent reforms have introduced fast-track approvals and streamlined evaluations for innovative technologies. Regulations also enforce strict quality control standards aligned with international norms and mandate post-market surveillance, including adverse event reporting. In addition, data protection laws ensure patient privacy and cybersecurity compliance. These measures encourage domestic and international manufacturers to bring advanced CGM systems to market efficiently, boosting growth while maintaining high safety and performance standards.
In China, traditional finger-prick blood glucose meters remain the primary substitute for CGM systems, offering a low-cost and widely accessible alternative. Flash monitoring systems like Abbott’s FreeStyle Libre serve as a middle-ground option, providing periodic glucose readings without requiring continuous wear. Insulin pumps with integrated glucose sensing offer a semi-automated substitute, often used by Type 1 patients seeking tighter control. In addition, emerging non-invasive glucose devices-such as optical sensors-are gaining interest, although they’re not yet mainstream. Finally, smartphone health apps combined with periodic lab tests can partially replace CGMs, especially where cost or access is a concern.
The China CGM devices industry is witnessing notable growth with Spirit Health’s announcement to launch its CareSens Air CGM in February 2025. This user-friendly, 15-day wearable sensor sends glucose readings directly to a smartphone every five minutes. Designed for simplicity and affordability, it features an ergonomic, all-in-one applicator that ensures painless attachment and minimal discomfort. The integrated mobile app offers real-time trend visualization, personalized alerts, and lifelog tracking for effective glucose management. It also supports remote monitoring by caregivers and healthcare professionals, enhancing data-driven care. This regional launch reinforces Spirit Health’s commitment to accessible and connected diabetes solutions.
Product Insights
The standalone CGM systems segment dominated the market in 2024 due to growing preference for real-time, non-invasive glucose tracking, especially among type 2 diabetes patients. These devices, operating independently of insulin pumps, gained popularity for their cost-effectiveness, user-friendliness, and broad availability through hospitals and e-commerce platforms. Government support for digital healthcare and rising awareness of CGM benefits further fueled adoption. Innovations by local companies and competitive pricing enhanced access and drove leadership in the segment. In addition, companies like Goodix advanced the sector with compact, power-efficient CGM solutions, contributing to rapid product development and supporting the broader growth of smart health wearables in China.
The integrated CGM-insulin pump systems segment is projected to experience the fastest CAGR in 2024 during the forecast period, driven by the rising adoption of automated diabetes management technologies. These systems offer real-time glucose monitoring with automatic insulin delivery, improving glycemic control and reducing patient burden. Growing prevalence of Type 1 diabetes, increasing awareness of closed-loop systems, and expanding access to advanced medical devices support this trend. Technological advancements, government initiatives promoting digital health, and partnerships between CGM and insulin pump manufacturers are also fueling demand. Their ability to enhance patient outcomes is making them a preferred choice for intensive diabetes therapy.
Connectivity Insights
The bluetooth segment dominated the market with the largest revenue share in 2024 due to its widespread integration in wearable health technologies. Bluetooth-enabled CGMs offer seamless real-time glucose data transmission to smartphones and monitoring apps, improving convenience and user engagement. The growing popularity of mobile health platforms, coupled with consumer preference for wireless, easy-to-use devices, has fueled adoption. Manufacturers increasingly incorporate Bluetooth for compatibility with digital health ecosystems. In addition, advancements in low-power Bluetooth chipsets have enhanced battery life and accuracy, making them ideal for continuous monitoring. These advantages have solidified Bluetooth’s dominance in the CGM connectivity segment.
The near field communication (NFC) segment is projected to witness the fastest CAGR during the forecast period due to its simplicity, cost-effectiveness, and compatibility with smartphones. NFC-enabled CGMs allow users to scan sensors with mobile devices for instant glucose readings without continuous connectivity, appealing to budget-conscious consumers. Its low energy consumption and user-friendly design make it suitable for large-scale adoption, especially in regions with limited digital infrastructure. As Chinese manufacturers focus on affordability and accessibility, NFC technology is emerging as a preferred solution for diabetes management, supporting broader market penetration and driving rapid segment expansion.
Indication Insights
The type 1 diabetes segment dominated the market the largest revenue share in 2024. This growth is driven by the increasing prevalence of type 1 diabetes in children and adolescents. According to the IDF, China has around 116,588 individuals aged 0-19 with type 1 diabetes. The high need for accurate and continuous glucose monitoring in this group has fueled CGM device adoption. Parents prefer CGMs for their non-invasive, real-time tracking benefits. Pediatric healthcare improvements, enhanced awareness, and supportive government policies have further boosted demand.
The type 2 diabetes segment is projected to grow at the fastest CAGR over the forecast period due to rising diabetes prevalence, lifestyle changes, and an aging population. With over 140 million people living with diabetes in China mostly type 2, the need for effective monitoring tools has surged. Increased awareness, government initiatives promoting digital health, and affordability of CGMs have accelerated adoption. In addition, the integration of CGM data with mobile apps for personalized care and remote monitoring appeals to elderly and tech-savvy users.
End Use Insights
The pharmacies segment dominated the market in 2024, accounting for the largest revenue share due to widespread accessibility, consumer trust, and expanded product availability. Pharmacies serve as convenient points of purchase for diabetes patients, especially in urban and semi-urban areas. Increased collaboration between CGM manufacturers and retail pharmacy chains has improved device visibility and affordability. In addition, many pharmacies offer in-store consultations and digital health guidance, encouraging CGM adoption. As awareness of continuous glucose monitoring grows among the diabetic population, pharmacies play a pivotal role in driving sales, making them a dominant distribution channel in the Chinese market.
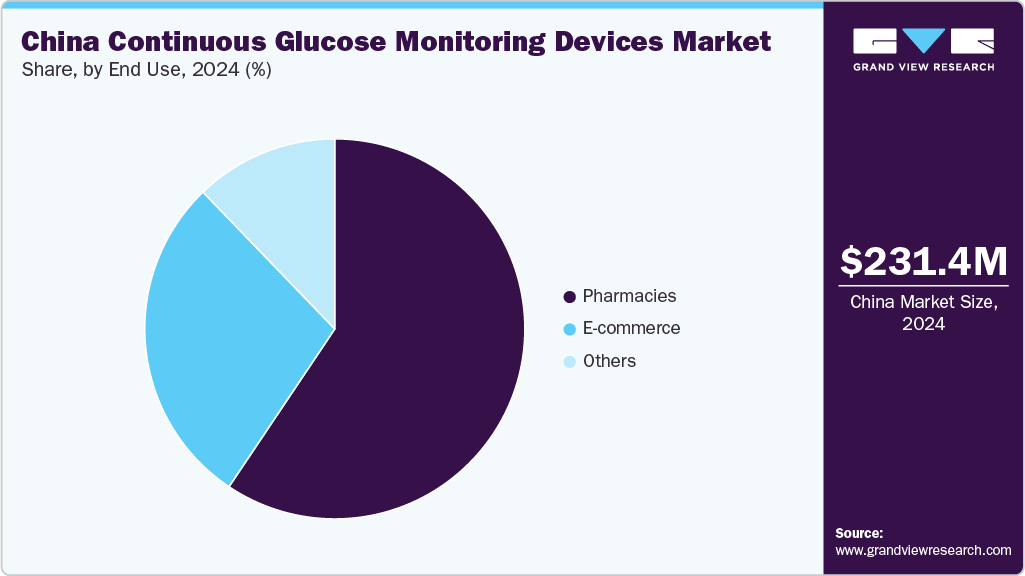
The e-commerce segment is expected to grow at the fastest CAGR over the forecast period due to rising internet penetration, consumer preference for convenience, and digital health trends. Online platforms like JD.com and Tmall offer a wide selection of CGM devices, competitive pricing, and home delivery, enhancing accessibility for users across regions. Manufacturers leverage these platforms to launch promotions, reach wider audiences, and gather customer feedback. In addition, increased health awareness and integration of online medical consultations further support e-commerce growth. As digital ecosystems evolve, e-commerce is poised to become a primary channel for CGM device distribution in China.
Key China Continuous Glucose Monitoring Devices Company Insights
Leading market players in China’s CGM devices industry include Abbott, Dexcom, and Sinocare Inc., each playing a pivotal role in expanding access to glucose monitoring technology. These companies have leveraged advanced sensor technologies, digital integration, and cost-effective pricing to gain competitive advantages. Abbott and Dexcom continue to lead with established global trust and product performance, while Sinocare’s local presence and affordability cater to the growing domestic demand. Their focus on enhancing user convenience, accuracy, and availability through pharmacies, e-commerce platforms, and hospitals has driven adoption across urban and rural areas, positioning them at the forefront of China’s rapidly growing CGM market.
Key China Continuous Glucose Monitoring Devices Companies:
- Abbott Laboratories
- Dexcom, Inc.
- Sinocare Inc.
- Medtronic plc
- Ascensia Diabetes Care
- Trinity Biotech
- MicroTech Medical
- Meiqi Medical
- Yuwell
- i-SENS Inc.
Recent Developments
-
In January 2025, Abbott expressed strong demand for its FreeStyle Libre glucose monitors and new device launches to offset challenges from China's procurement policies and currency headwinds in 2025. Despite a cautious first-quarter outlook, medical device sales exceeded expectations, with the company forecasting annual profits.
-
In January 2024, Dexcom launched the Dexcom ONE+ CGM in the China. Featuring the latest G7 sensor, it offers real-time glucose monitoring with smartphone and smartwatch compatibility. Designed for insulin-treated Type 1 and Type 2 diabetes, ONE+ enhances ease of use and tracks lifestyle-related glucose impacts.
China Continuous Glucose Monitoring Devices Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 277.1 million
Revenue forecast in 2033
USD 1,142.1 million
Growth rate
CAGR of 19.4% from 2025 to 2033
Actual Data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, connectivity, indication, end use
Regional scope
China
Key companies profiled
Abbott Laboratories; Dexcom, Inc.; Sinocare Inc.; Medtronic plc; Ascensia Diabetes Care; Trinity Biotech; MicroTech Medical; Meiqi Medical; Yuwell; i-SENS Inc.
Customization scope
Free report customization (equivalent upto to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
China Continuous Glucose Monitoring Devices Market Report Segmentation
This report forecasts revenue growth at country levels and analyzes the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the China continuous glucose monitoring devices market report based on product, connectivity, indication and end use.
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Standalone CGM Devices
-
Devices
-
Consumables (Receivers, Sensors, and Transmitters)
-
-
Integrated CGM-Insulin Pump Systems
-
-
Connectivity Outlook (Revenue, USD Million, 2021 - 2033)
-
Bluetooth
-
4G/Cellular
-
NFC (Near Field Communication)
-
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Type 1 Diabetes
-
Type 2 Diabetes
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmacies
-
E-commerce
-
Others
-
Frequently Asked Questions About This Report
b. The China continuous glucose monitoring devices market size was valued at USD 231.4 million in 2024 and is expected to reach a value of USD 277.1 million in 2025.
b. The China continuous glucose monitoring devices market is expected to grow at a compound annual growth rate of 19.4% from 2025 to 2033 to reach USD 1,142.1 million by 2033.
b. The standalone CGM systems segment dominated the China market and accounted for the largest revenue share of around 78.3% in 2024, owing to their widespread availability, ease of use, and growing adoption among insulin-dependent patients.
b. Some key players operating in the China CGM devices market includes Abbott Laboratories, Dexcom, Inc., Sinocare Inc., Medtronic plc, Ascensia Diabetes Care, Trinity Biotech, MicroTech Medical, Meiqi Medical, Yuwell, i-SENS Inc.
b. Key factors driving the growth of the China CGM devices market include the increasing prevalence of diabetes, growing awareness of continuous glucose monitoring benefits, expanding healthcare infrastructure, and supportive government initiatives. Additionally, technological advancements and rising demand for real-time, accurate glucose monitoring are fueling adoption, especially among insulin-treated patients.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










