- Home
- »
- Clinical Diagnostics
- »
-
France In Vitro Diagnostics Market, Industry Report, 2033GVR Report cover
![France In Vitro Diagnostics Market Size, Share & Trends Report]()
France In Vitro Diagnostics Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Instruments, Reagents, Software), By Test Location (Point of Care, Home-care), By End-use (Hospitals, Laboratory), By Application, By Technology, And Segment Forecasts
- Report ID: GVR-4-68040-858-9
- Number of Report Pages: 320
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
France In Vitro Diagnostics Market Summary
The France in vitro diagnostics market size was estimated at USD 4.37 billion in 2025 and is projected to reach USD 6.93 billion by 2030, growing at a CAGR of 5.86% from 2026 to 2033. Some of the key growth factors driving the market growth include the increasing prevalence of chronic and infectious diseases, advancements in diagnostic technologies, and the shift toward value-based healthcare emphasizing early detection and preventive care.
Key Market Trends & Insights
- By product, the reagents segment led the market with the largest revenue share of 66.39% in 2025.
- By technology, the molecular diagnostics-based products segment led the market with the largest revenue share of 24.97% in 2025.
- Based on application, the infectious diseases segment led the market with the largest revenue share of 38.11% in 2025.
- Based on test location, the other location segment led the market with the largest revenue share of 56.50% in 2025.
- Based on end use, the hospitals segment led the market with the largest revenue share of 48.09% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 4.37 Billion
- 2033 Projected Market Size: USD 6.93 Billion
- CAGR (2026-2033): 5.86%
The growth of the market is attributed to the rising global demand for accurate, rapid, and personalized diagnostic solutions. The integration of digital health, AI, and data analytics into diagnostic platforms enhances test accuracy and clinical decision-making. In addition, the expansion of point-of-care testing, personalized medicine, and emerging markets, coupled with heightened pandemic preparedness, continues to drive market penetration, creating significant opportunities for industry stakeholders.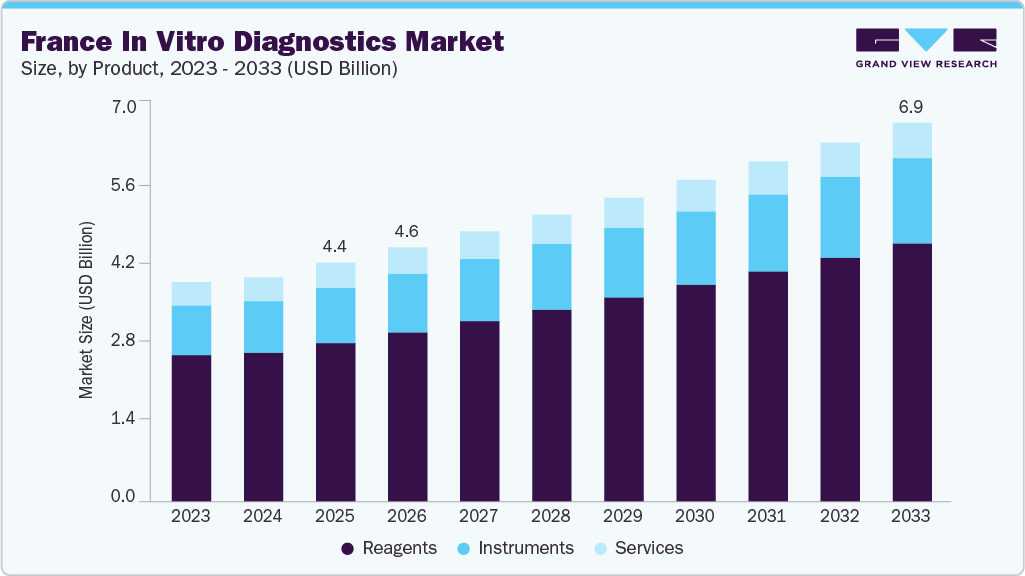
The France in vitro diagnostics industry is evolving rapidly, driven by technological advancements, rising disease prevalence, and government initiatives to strengthen healthcare infrastructure. Major global players, including Roche, Thermo Fisher Scientific, Qiagen, Danaher, and Hologic, have established a strong presence in France, continuously expanding their molecular diagnostics portfolios with real-time PCR systems and high-throughput sequencing tools.
Furthermore, the French molecular diagnostic market would thus be directly impacted by this acquisition, as companies expand their capabilities and presence in this growing sector. In January 2025, bioMérieux acquired SpinChip Diagnostics ASA (SpinChip). This small benchtop analyzer is designed for near-patient testing, delivering high-sensitivity results from whole blood samples within 10 minutes-a level of performance previously available only in laboratory settings.
Health spending as a share of GDP highlights the priority France assigns to healthcare within its overall economy, and it is a critical structural indicator for understanding long-term demand in the IVD market. In 2023, health expenditure in France accounted for 11.5% of GDP, making it the second highest in the European Union after Germany (11.7%) and well above the EU average of around 10%.
Furthermore, France has significantly strengthened its national laboratory and research capacity through large investments in artificial intelligence (AI), high-performance computing, and biosafety infrastructure, particularly in response to pandemic preparedness needs. One of the key drivers has been the National AI Strategy, which allocated around USD 1.62 billion (EUR 1.5 billion) between 2018 and 2022 and was later expanded under the broader France 2030 initiative. These investments aim to support advanced data analysis, medical research, and innovation across healthcare and diagnostics. Based on 2024-2025 data, France possesses a high-capacity, largely consolidated, and technologically advanced national diagnostic laboratory infrastructure, with over 1,100 research laboratories and over 800 million annual clinical tests.
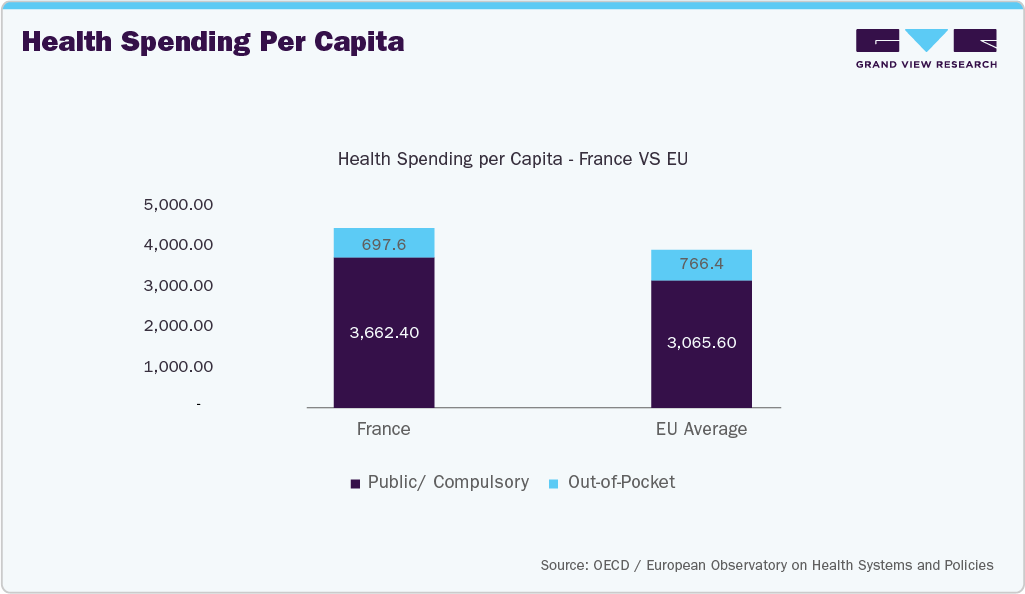
France has one of the lowest OOP burdens in Europe, which directly supports higher diagnostic test utilization:

A defining strength of the French system lies in its financing structure, with public and compulsory health insurance schemes funding 84.4% of total health expenditure, compared with an EU average of 80.0%. As a result, the out-of-pocket (OOP) share is only 9.3%, among the lowest in the European Union, while voluntary health insurance accounts for a further 6.3%. This financing mix is particularly advantageous for the IVD market because diagnostic tests are routinely prescribed within standardized clinical pathways and reimbursed through national mechanisms. Physicians and laboratories therefore face minimal financial friction when ordering tests, supporting widespread utilization across preventive screening, chronic disease monitoring, and hospital-based diagnostics. Diagnostics are consequently structurally embedded in routine medical decision-making, rather than being selectively used based on patient affordability.
Product Insights
The reagent segment led the market with the largest revenue share of 66.39% in 2025, the European Commission’s historical scrutiny of consolidation involving diagnostic consumables continues to influence supplier strategies in France, encouraging portfolio divestments and multi-supplier procurement models to preserve competition. Reagents represent the most structurally resilient product segment in the France IVD market due to their direct linkage to testing volumes and their recurring nature. Even as post-pandemic testing normalized, national data from Assurance Maladie show that total laboratory test volumes in 2023-2024 remained materially above pre-COVID levels, sustaining baseline reagent consumption across public and private laboratories.
The instruments segment is anticipated to grow at the fastest CAGR over the forecast period. The growth of the segment is fueled by advancements in technology and partnerships among major players. In January 2025, bioMérieux acquired SpinChip Diagnostics ASA (SpinChip). This small benchtop analyzer is designed for near-patient testing, delivering high-sensitivity results from whole blood samples within 10 minutes of a level of performance previously available only in laboratory settings. According to bioMérieux’s Chief Medical Officer, point-of-care (POC) diagnostics play a critical role in improving patient outcomes and reducing healthcare costs.
Technology Insights
The molecular diagnostics segment led the market with the largest revenue share of 24.97% in 2025. Molecular diagnostic tools are essential for identifying biomarkers linked to chronic diseases at early stages, enabling timely interventions and improved patient outcomes. The segment is shaped by major global players like Roche, Thermo Fisher Scientific, Qiagen, Danaher, and Hologic, which have a strong presence in France. These companies are expanding their portfolios with innovations such as real-time PCR systems and high-throughput sequencing tools. Furthermore, the French molecular diagnostic market would thus be directly impacted by this acquisition, as companies expands its capabilities and presence in this growing sector. For instance, In May 2024, Bruker Corporation successfully completed its acquisition of ELITechGroup for USD 915.77 million in cash, excluding the clinical chemistry division of ELITech. ELITech is a high-growth, profitable company specializing in systems and assays for molecular diagnostics (MDx), biomedical systems, and microbiology.
The coagulation segment is anticipated to grow at the fastest CAGR over the forecast period, driven by increasing demand for precise diagnostics to assess hemostasis, a process that prevents excessive bleeding by enabling blood clot formation. Coagulation tests are essential in detecting abnormalities in blood clotting components, making them vital for patients with cardiovascular conditions, blood disorders, and autoimmune diseases. Major companies, including Abbott, Siemens Healthcare GmbH, and Beckman Coulter, Inc., are actively innovating in this field, creating advanced IVD instruments that streamline and improve coagulation testing. The COVID-19 pandemic also brought significant attention to coagulation diagnostics.
Application Insights
The infectious diseases segment accounted for the largest market revenue share in 2025. One major trend is the shift toward multiplex tests that detect multiple respiratory pathogens like Flu A/B, COVID-19, and RSV in a single panel. While the expansion into multiplex respiratory testing initially met market needs, the space has quickly become saturated with similar products. As a result, companies need to avoid "me-too" products and instead emphasize unique product features to secure a foothold in labs and clinics that may be consolidating equipment after pandemic-driven over-purchasing. Innovation from leading manufacturers continues to strengthen France’s infectious-disease testing ecosystem. For instance, in April 2025, bioMérieux introduced WATCHFIRE, a molecular testing solution featuring a 22-target respiratory panel designed for integration with the BIOFIRE platform and FIREWORKS software.
The oncology segment is anticipated to grow at the fastest CAGR over the forecast period, driven by the increasing prevalence of cancer and the demand for advanced diagnostic solutions. Cancer remains a significant health burden, as it is the second leading cause of death in the U.S., with common types including colorectal, prostate, breast, and lung cancer. The rising importance of early cancer detection is likely to propel the demand for cancer-related IVD tests, especially as AI and digital pathology streamline workflows and improve diagnostic accuracy. The adoption of comprehensive, multi-biomarker profiling tests and non-invasive liquid biopsies will continue to grow, meeting the demand for precise and patient-friendly cancer diagnostics.
Test Location Insights
The others segment accounted for the largest market revenue share in 2025. This includes traditional clinical laboratories, hospital diagnostic departments, reference and centralized testing facilities, specialized public health labs, and other institutional diagnostic settings that support high-throughput and complex assays. While PoC and home testing have grown rapidly due to convenience and decentralization, the others segment remains a key revenue driver because it houses a significant volume of routine, specialized, and confirmatory testing that cannot be entirely replaced by decentralized formats.
The homecare segment is expected to grow at the fastest CAGR during the forecast period. The France in vitro diagnostics industry has seen a marked increase in the adoption of homecare testing, driven by the need for accessible and reliable testing solutions. In July 2025, the French social-security system expanded its national STI screening programme by offering free at-home test kits for sexually transmitted infections (STIs) to women aged 18-25, with plans to extend the service to men in the same age group later in the year. This initiative builds on the Mon test IST programme launched in response to rising STI rates post-pandemic which already allows anyone enrolled in the French healthcare system to get STI tests in medical laboratories without a GP prescription, reimbursed fully for those under 26 and at 60 % for older patients.
End-use Insight
The hospitals segment accounted for the largest market revenue share in 2025, owing to increased hospitalization, as doctors require diagnostic interpretation for further treatment. Most often, diagnostic centers operate in collaboration with hospitals; hence, hospitals have their own diagnostic set up. Furthermore, ongoing development of healthcare infrastructure is anticipated to enhance the existing hospital facilities. Thus, the demand for hospital-based IVD tests is increasing. Most of the IVD devices are purchased by hospitals and are used in significant volumes.

The others segment is expected to grow at the fastest CAGR over the forecast period, due increasing demand for molecular diagnosis,this segment includes primary care settings, pharmacies, occupational health providers, contract research organizations (CROs), and public-health agencies. In primary care and pharmacy settings, diagnostic use is centered on screening, chronic disease monitoring, and rapid tests, particularly glucose testing, infectious disease screening, and select POCT applications. These settings benefit from national initiatives aimed at decentralizing care and improving early diagnosis outside hospitals.
Key France In Vitro Diagnostics Company Insights
Some of the key players operating in the France in vitro diagnostics industry include Abbott, bioMérieux, F. Hoffmann-La Roche Ltd., Siemens Healthineers AG, Thermo Fisher Scientific, Inc., Bio-Rad Laboratories, Inc. among others. Mature players heavily invest in state-of-the-art technology and infrastructure, allowing them to efficiently process & analyze a large volume of data. Market players in the France IVD market are adopting various market strategies such as new product launches, mergers & acquisitions, and partnerships to strengthen their product portfolios, offer diverse technologically advanced & innovative products, expand their product portfolios, and improve competency.
ARUP Laboratories, RayBiotech, Inc., Randox Laboratories Ltd., Novacyt Group, Acobiom, Poly-Dtech, and Biosynex are some of the emerging market participants in the France in vitro diagnostics industry. Emerging players put greater focus on gaining market penetration through product differentiation. These companies are actively involved in developing novel and accurate IVD testing products to improve overall health services. Moreover, these companies are collaborating with research institutes, government bodies, and global leaders to increase the range of their products in potential markets.
Key France In Vitro Diagnostics Company:
- Eurobio Scientific
- Servibio
- CliniSciences
- Avantor, Inc.
- QIAGEN
- BD
- bioMérieux SA
- F. Hoffmann-La Roche AG
- QuidelOrtho Corporation
- Abbott
- Agilent Technologies, Inc.
- Siemens Healthineers AG
- Bio-Rad Laboratories, Inc.
- Danaher
- Sysmex Corporation
- Charles River Laboratories
- Quest Diagnostics Incorporated
- Novacyt Group
- Acobiom
- Poly-Dtech
- Biosynex.
Recent Developments
-
In January 2026, Charles River announced plans to acquire PathoQuest SAS from SHS Capital, combining PathoQuest’s proprietary NGS biosafety testing capabilities with Charles River’s global scale to accelerate adoption of advanced in-vitro sequencing methods and strengthen its France operations in quality-control and diagnostic-related testing for biopharma.
-
In December 2025, bioMérieux announced the launch of a new immunoassay endocrinology testing portfolio for equine health under the VIDAS platform, expanding its in vitro diagnostics offerings in niche clinical markets that intersect veterinary labs and specialized diagnostics segments used by some French diagnostic providers.
-
In November 2025, bioMérieux collaboration with Oxford Nanopore Technologies, bioMérieux launched AmPORE-TB, a sequencing-based research use only solution for rapid characterization of drug-resistant tuberculosis, supporting advanced molecular diagnostic workflows that laboratories in France and Europe use for infectious disease control.
-
In January 2025, bioMérieux strengthened its point-of-care diagnostics presence by acquiring SpinChip, an immunoassay platform company, supporting future IVD product development including high-sensitivity POC immunoassays. The integration complements bioMérieux’s VIDAS and BIOFIRE portfolios and targets enhanced rapid diagnostics solutions that French clinical laboratories will adopt once commercialized.
France In Vitro Diagnostics Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 4.65 billion
Revenue forecast in 2033
USD 6.93 billion
Growth rate
CAGR of 5.86% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, Volume (number of instruments in thousands, reagents in number of tests in million), and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Products, technology, application, test location, end-use
Country scope
France
Key companies profiled
Eurobio Scientific; Servibio; CliniSciences; Avantor, Inc.; QIAGEN; BD; bioMérieux SA; F. Hoffmann-La Roche AG; QuidelOrtho Corporation; Abbott; Agilent Technologies, Inc.; Siemens Healthineers AG; Bio-Rad Laboratories, Inc.; Danaher; Sysmex Corporation; Charles River Laboratories; Quest Diagnostics Incorporated; Novacyt Group; Acobiom; Poly-Dtech; Biosynex.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
France In Vitro Diagnostics Market Report Segmentation
This report forecasts revenue growth at country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the France in vitro diagnostics market report based on products, technology, application, test location, and end-use:
-
Product Outlook (Revenue, USD Billion, 2021 - 2033) (Volume, Number of Instruments, in Thousands, Volume, Reagents, Number of Tests in Million)
-
Instruments
-
Reagents
-
Software
-
-
Technology Outlook (Revenue, USD Billion, 2021 - 2033) (Volume, Number of Instruments, in Thousands, Volume, Reagents, Number of Tests in Million)
-
Immunoassay
-
Instruments
-
Reagents
-
Others
-
-
Hematology
-
Instruments
-
Reagents
-
Others
-
-
Clinical Chemistry
-
Instruments
-
Reagents
-
Others
-
-
Molecular Diagnostics
-
Instruments
-
Reagents
-
Others
-
-
Coagulation
-
Instruments
-
Reagents
-
Others
-
-
Microbiology
-
Instruments
-
Reagents
-
Others
-
-
Others
-
Instruments
-
Reagents
-
Others
-
-
-
Application Outlook (Revenue, USD Billion, 2021 - 2033)
-
Infectious Diseases
-
Diabetes
-
Oncology
-
Cardiology
-
Nephrology
-
Autoimmune Diseases
-
Drug Testing
-
Other applications
-
-
Test Location Outlook (Revenue, USD Billion, 2021 - 2033)
-
Point of Care
-
Home-care
-
Others
-
-
End-use Outlook (Revenue, USD Billion, 2021 - 2033) (Volume, Number of Instruments, in Thousands, Volume, Reagents, Number of Tests in Million)
-
Hospitals
-
Public
-
Private
-
-
Laboratory
-
Public
-
Private
-
-
Others
-
Frequently Asked Questions About This Report
b. The France in vitro diagnostics market size was estimated at USD 4.37 billion in 2025 and is expected to reach USD 4.65 billion in 2026.
b. The France in vitro diagnostics market is expected to grow at a compound annual growth rate of 5.86% from 2026 to 2033 to reach USD 6.93 billion by 2033.
b. The reagents segment accounted for largest market share of 66.37% in 2025. This is attributed to their substantially elevated risk of progression from latent infection to active TB disease.
b. Some key players operating in the France in vitro diagnostics market include Eurobio Scientific, Servibio, CliniSciences, Avantor, QIAGEN, BD, bioMérieux, Roche, QuidelOrtho, Abbott, Agilent, Siemens Healthineers, Bio-Rad, Danaher, Sysmex, Charles River Laboratories, Quest Diagnostics, Novacyt Group, Acobiom, Poly-Dtech, and Biosynex.
b. Key factors that are driving the market growth include the increasing prevalence of chronic and infectious diseases, advancements in diagnostic technologies, and the shift toward value-based healthcare emphasizing early detection and preventive care.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










