- Home
- »
- Clinical Diagnostics
- »
-
Middle East Advanced Cancer Diagnostics Market Report 2033GVR Report cover
![Middle East Advanced Cancer Diagnostics Market Size, Share & Trends Report]()
Middle East Advanced Cancer Diagnostics Market (2025 - 2033) Size, Share & Trends Analysis Report By Test (Tumor Marker Tests, Imaging-Based Diagnostics), By End Use (Hospitals & Cancer Specialty Centers, Research & Academic Institutes), By Country, and Segment Forecasts
- Report ID: GVR-4-68040-784-0
- Number of Report Pages: 130
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
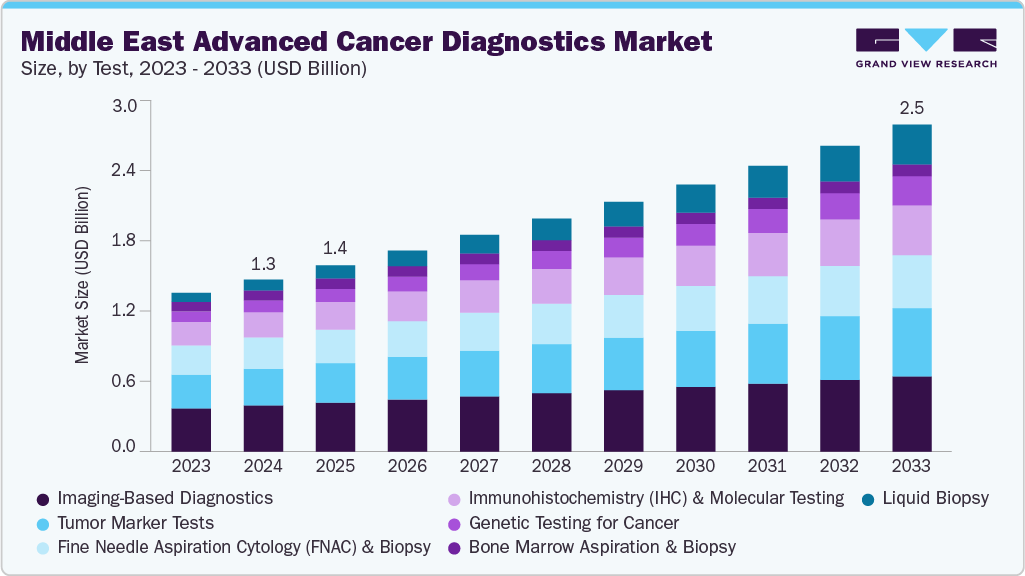
The Middle East advanced cancer diagnostics market size was estimated at USD 1.32 billion in 2024 and is projected to reach USD 2.51 billion by 2030, growing at a CAGR of 7.30% from 2025 to 2033. The market for advanced cancer diagnostics in the Middle East is driven by several strong factors such as increasing prevalence of cancer, growing demand for early detection, launch of advanced diagnostic technologies, and investment under national healthcare modernization.
Breakthroughs such as liquid biopsy, AI-enabled pathology, and biomarker-driven companion diagnostics accelerate precision oncology, enabling more personalized and effective treatment. For instance, in June 2025, M42, AstraZeneca & SOPHiA GENETICS launched UAE liquid biopsy initiative to transform cancer care. As part of the collaboration, M42 will incorporate SOPHiA GENETICS' MSK-ACCESS, which is powered by SOPHiA DDM technology, into its testing infrastructure, allowing for non-invasive genomic profiling of cancers using a simple blood draw. This approach complements, not replaces, routine screenings and is designed for patients who are unable to undergo a solid tumor biopsy, allowing them to be matched with a precision therapy tailored to their specific cancer profile.
Further, governments across the country, particularly in the UAE, Qatar, and Saudi Arabia prioritize oncology care, implement national cancer control programs, and promote advanced diagnostic technologies like liquid biopsies, next-generation sequencing (NGS), and molecular imaging. Furthermore, the increasing number of specialized cancer centers and international collaborations hastened the adoption of precision diagnostics. Also, private sector participation and partnership with Middle East diagnostic firms improve access to cutting-edge tools and expertise.
Rapid technological advancements, evolving policy frameworks, and shifting healthcare priorities influence the market dynamics for advanced cancer diagnosis. Traditionally, cancer diagnosis and treatment were primarily determined by tumor location and stage, but the advent of advanced diagnostics has altered this paradigm. These new technologies provide detailed insights into cancer's molecular, genetic, and biological underpinnings, making precision medicine a reality. Advanced diagnostics reduce the likelihood of incorrect diagnoses while allowing for more personalized treatment selection. This precision reduces patient exposure to ineffective therapies and their associated side effects, while also allowing for real-time monitoring of treatment outcomes.
Public-private partnerships and collaborations, increasing adoption of precision medicine, and favorable reimbursement policies facilitating broader adoption of advanced diagnostic technologies are fueling market's growth. For instance, in March 2025, Lunit expanded its partnership with Saudi Arabia's largest medical group to deploy an AI Chest X-Ray Solution. This latest contract expands on Lunit's previous collaboration with HMG, which introduced Lunit INSIGHT MMG to support breast cancer screening across the Kingdom. The collaboration between Lunit and INSIGHT CXR aimed to improve diagnostic accuracy and efficiency in detecting critical chest abnormalities like lung cancer, pneumonia, and tuberculosis.
Favorable reimbursement policies and regulatory alignment, such as Saudi Arabia's Vision 2030 healthcare reforms and insurance expansions, contribute to the rapid adoption of advanced diagnostics. Furthermore, PPP-funded national awareness campaigns, technology transfer initiatives improve Country access, affordability of advanced diagnostic tools, and clinician training programs. The UAE's breast cancer diagnostics industry exemplifies this by leveraging public-private partnerships, AI-powered imaging, and liquid biopsy programs to improve early detection and precision treatment.
Market Concentration & Characteristics
The Middle East advanced cancer diagnostics industry is witnessing high innovation because of adoption of next‑generation sequencing (NGS), AI‑enabled imaging and digital cytology, liquid biopsy & multi‑cancer early detection (MCED) tests, and population‑specific diagnostic developments. For instance, The Department of Health: Abu Dhabi (DoH) launched AI-powered initiatives at GITEX Global 2025, as part of its vision to help residents live longer and healthier lives. Meanwhile, university-led research collaborations have used AI to identify new immunotherapy targets for cancer by researching genes connected to blood vessel cells, opening the door to speedier and more cost-effective treatments.
The level of mergers and acquisitions is medium. It is not yet at the level seen in more mature markets, but activity is increasing and is strategically significant. To gain access to distribution, regulatory approval, or specialized diagnostic infrastructure, many international firms form partnerships or make selective acquisitions.
Regulation has a high impact owing to regulatory authorities, such as the Saudi Food and Drug Authority (SFDA) and the Ministry of Health and Prevention (MOHAP), playing significant roles such as approving tests, ensuring quality, supporting standards, integrating diagnostics into national cancer screening programs, controlling reimbursement frameworks, and lab accreditation. These regulatory factors strongly influence what diagnostics get adopted, at what pace, and where.

The substitute threat is medium because of traditional diagnostic imaging tools such as MRI, computed tomography (CT), ultrasound, biopsy + histology, and classical tumor markers are deeply entrenched, accessible, relatively inexpensive, and familiar. However, advanced diagnostics are starting to displace these substitutes for early detection, precision, and non‑invasiveness, especially where healthcare infrastructure and funding allow. Thus, the substitute threat is non‑trivial.
End user concentration is high because of a few large hospitals, specialized cancer centres, government health systems, and major diagnostic and reference labs account for the majority of demand and usage of advanced diagnostics. Smaller clinics or rural facilities are much less likely to have capacity for sophisticated tests. This concentrates market power and influence in relatively few end‑users.
Test Insights
The imaging-based diagnostics remain the largest segment in the market, contributing a share of 26.8%, supported by their central role in detection, staging, and treatment monitoring. Core modalities such as CT, PET/CT, and MRI are well-established, but the segment is evolving with advanced capabilities. For instance, in June 2025, Koning Health launched the UAE's first no-compression 3D breast imaging system. The advanced system was delivered with FourMed Medical Supplies LLC, Koning's strategic distributor in the UAE. FourMed, which is known for its commitment to service excellence and advancing Country healthcare, was instrumental in bringing this cutting-edge solution to the UAE.
In contrast, liquid biopsy is the fastest-growing segment, driven by its minimally invasive sampling and real-time insights into ctDNA, cfDNA, and circulating tumor cells. Moreover, unlike imaging, which is best suited for structural visualization, liquid biopsy enables molecular early detection, minimal residual disease tracking, and therapy resistance monitoring. Its appeal is also economic, it reduces the need for invasive biopsies and predicts relapses earlier, lowering downstream costs. Further, with accelerating clinical validation, and its transformative role in multi-cancer early detection, liquid biopsy is widely regarded as the most disruptive force in diagnostics for the next decade.
End use Insights
The hospitals and cancer specialty centers held the largest share of 45.0% in 2024, reflecting their established infrastructure for CT, MRI, PET/CT, and biopsy services, as well as their integration of advanced modalities like 3Tesla MRI and hybrid imaging. These centers lead to adoption because of multidisciplinary care pathways that directly link diagnostics with oncology treatment, and strong reimbursement systems that reinforce reliance on hospital-based platforms. For instance, in August 2024, Thumbay Healthcare, UAE’s largest private academic healthcare network, inaugurated the Thumbay Advanced Cancer Center. The new center is technologically advanced and provides a variety of specialized services, such as private chemotherapy suites, dedicated specialty clinics, and a breast cancer unit. Each aspect of the center is designed with patient-centric facilities to ensure the highest level of care and comfort.
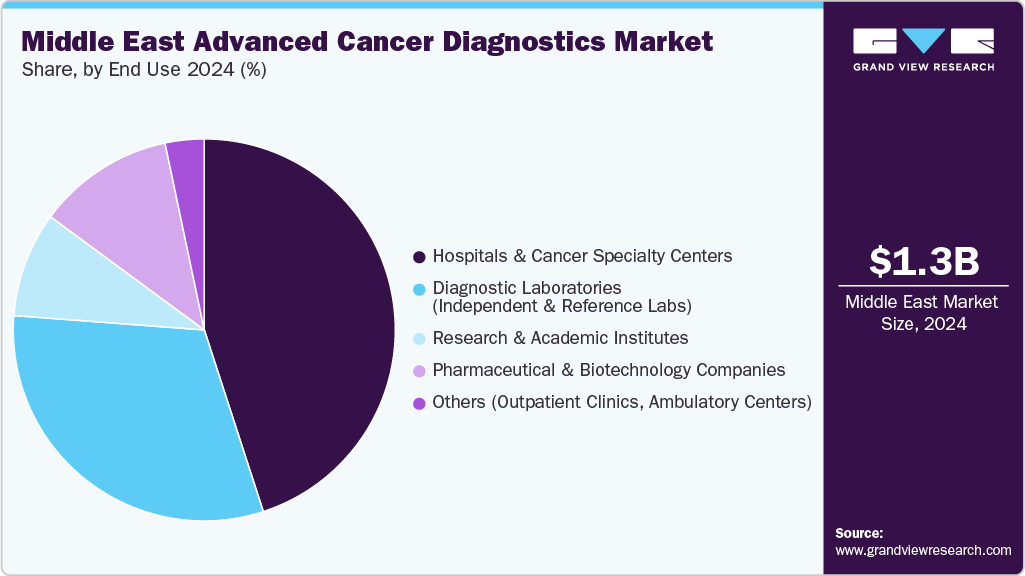
Further, diagnostic laboratories are anticipated to emerge as the fastest-growing end-use segment over the forecast period. Their expansion is driven by the rising demand for specialized, high-throughput, and cost-effective testing services, which many hospitals and smaller healthcare facilities cannot provide in-house. These laboratories frequently provide genetic testing, liquid biopsies, advanced molecular diagnostics, and biomarker-based assays. Moreover, as the region's healthcare systems evolve, there is a growing trend of outsourcing complex diagnostics to centralized, accredited labs equipped with cutting-edge technology platforms and skilled workforces. Furthermore, diagnostic laboratories are increasingly collaborating with international companies and government programs to broaden their capabilities, accelerating their rapid growth.
Country Insights
The advanced cancer diagnostics industry in Middle East is expanding. Countries such as the UAE, Kuwait, Saudi Arabia, Qatar, and Oman are witnessing an expansion of the companies and their investment in advanced diagnostic capabilities for oncology. For instance, GE Healthcare (US) and Advanced Technology Company (Kuwait) will provide healthcare IT systems, as well as CT, MRI, PET/CT, and ultrasound scanners, to Al-Razi Hospital, Sabah Hospital, Kuwait Cancer Hospital, Farwaniya Hospital, and Jahra Hospital. The five new facilities will add over 3,000 beds to Kuwait. Furthermore, the Kuwaiti government intends to invest several billion dollars over three years to build nine hospitals to improve healthcare services.
Saudi Arabia Advanced Cancer Diagnostics Market Trends
Saudi Arabia advanced cancer diagnostics industry has a strong and growing presence in the Middle East market. The breast cancer diagnostic segment is both the most significant revenue contributor and the fastest-growing subtype within that molecular diagnostics market. In addition, the increasing prevalence in Saudi Arabia is driving the growth of the market as well as boosting the opportunities for the imaging devices manufacturers to expand their presence in the country. For instance, as per the Organisation for Economic Co-operation and Development 2024 report, co-funded by the Qataran Union, it stated that cancer is the fifth leading cause of mortality in Saudi Arabia, and one in 16 premature (before the age of 75) deaths (6%) will be due to cancer between 2023 and 2050.
UAE Advanced Cancer Diagnostics Market Trends
The UAE advanced cancer diagnostics industry is growing as there is strong emphasis on early cancer detection and diagnostic quality. The Ministry of Health & Prevention runs periodic screening and early detection programs and is also involved in raising awareness and ensuring that healthcare facilities across the country are able to provide necessary diagnostic services. The UAE has developed a national cancer registry to collect and analyse data on all invasive and in situ cancer cases, tracking stage, treatment and outcomes, which supports better planning and diagnostic quality. In addition, in Abu Dhabi there is a move to introduce a blood‑based colorectal cancer screening test to complement colonoscopies, as part of a broader strategy to improve early detection.
Kuwait Advanced Cancer Diagnostics Market Trends
Kuwait's advanced cancer diagnostic industry is growing steadily as well; it has established cancer treatment centers and participates in Country health care initiatives. Moreover, the country is also establishing a new cancer unit, which is boosting the growth of the market. For instance, in October 2025, Kuwait's new unit sets the Country healthcare standard. The Kuwait Ministry of Health (MoH) has made a significant contribution to women's healthcare by establishing a new Gynecological Oncology Unit at the Kuwait Cancer Control Center (KCCC) in the Al-Sabah Specialized Medical District. This initiative is part of a larger effort to improve specialized healthcare services throughout the country. Moreover, the unit's primary goal is to improve diagnostic services in gynecological oncology. The MoH's goal in incorporating international medical standards is to improve women's health, reduce medical complications, and improve female patients' quality of life. The new unit is expected to strengthen Kuwait's position as a Country leader in this medical field.
Oman Advanced Cancer Diagnostics Market Trends
The Oman advanced cancer diagnostic industry is primarily driven by implementation of healthcare projects to detect cancer earlier and enhance diagnostic services. For instance, the government has plans to expand early detection units, support diagnostic infrastructure, and enhance screening services. While some are recent and still in the implementation phase, they signal a push toward improving diagnostic capacity, especially for common cancers among women. Moreover, the efforts include improving diagnostic centers, imaging services, and pathology labs to handle increased cancer detection and diagnosis demand.
Qatar Advanced Cancer Diagnostics Market Trends
Qatar advanced cancer diagnostic industry is emerging as it has established strong cancer detection, data gathering, and screening systems. Its “Screen for Life” national program is a structured initiative offering breast and bowel cancer screening, including home screening with accessible logistics and clinic‑based screening facilities. Moreover, the Primary Health Care Corporation (PHCC) in Qatar has recently integrated an AI‑powered mammography tool, such as Lunit INSIGHT MMG, into its national breast cancer screening workflow, making Qatar among the first in the region to deploy AI at scale in such a program. Also, the country has developed the Qatar Cancer Information Centre (QCIC), a national cancer registry dedicated to collecting reliable data for policy, planning, and improving diagnostic and screening service quality.
Key Middle East Advanced Cancer Diagnostics Company Insights
The major market players operating are focused on adopting strategic initiatives such as launches, mergers & acquisitions, partnerships, etc. Furthermore, several players are focusing on the development of testing services, thereby boosting the demand.
Key Middle East Advanced Cancer Diagnostics Companies:
- Roche Diagnostics
- Illumina, Inc.
- Thermo Fisher Scientific
- Siemens Healthineers
- GE HealthCare
- Philips Healthcare
- Canon Medical Systems
- Fujifilm Holdings Corporation
- Agilent Technologies
- Bruker Corporation
- Becton, Dickinson and Company (BD)
- Bio-Rad Laboratories
- Myriad Genetics
Recent Developments
-
In August 2025, GE HealthCare Middle East partnered with Zahrawi Group marking a significant step forward in expanding and enhancing diagnostic imaging solutions across Qatar. This partnership brings together global expertise in medical imaging and digital solutions with a strong local presence and knowledge of Qatar's healthcare sector.
-
In January 2025, Siemens Healthineers launched transformative computed tomography (CT) scanner powered by Quantum Technology and AI at Arab Health 2025.
-
In January 2024, GE HealthCare launched 19 AI-powered innovations to shape the future of healthcare at ArabHealth 2024. Emphasizing its commitment to long-term growth and driving transformational change to improve overall healthcare delivery in the Middle East. At the annual global exhibition and conference, GE HealthCare also showcased its leading imaging, ultrasound, and patient care solutions, all of which are intended to improve clinical decision-making and patient outcomes.
Middle East Advanced Cancer Diagnostics Market Report Scope
Report Attribute
Details
Market size in 2025
USD 1.43 billion
Revenue forecast in 2033
USD 2.51 billion
Growth rate
CAGR of 7.30% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in (USD billion) and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Test, end use, country
Country scope
Saudi Arabia; UAE; Kuwait; Oman; Qatar; Rest of Middle East
Key companies profiled
Roche Diagnostics; Illumina, Inc.; Thermo Fisher Scientific; Siemens Healthineers; GE HealthCare; Philips Healthcare; Canon Medical Systems; Fujifilm Holdings Corporation; Agilent Technologies; Bruker Corporation; Becton, Dickinson and Company (BD); Bio-Rad Laboratories; Myriad Genetics.
Customization scope
Free report customization (equivalent to 8 analyst’s working days) with purchase. Addition or alteration to country & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options.
Middle East Advanced Cancer Diagnostics Market Report Segmentation
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2021-2033. For this report, Grand View Research has segmented the Middle East advanced cancer diagnostics market report by test, end use, and country:
-
Test Outlook (Revenue, USD Billion, 2021 - 2033)
-
Tumor Marker Tests
-
Fine Needle Aspiration Cytology (FNAC) & Biopsy
-
Imaging-Based Diagnostics
-
Bone Marrow Aspiration & Biopsy
-
Immunohistochemistry (IHC) & Molecular Testing
-
Genetic Testing for Cancer
-
Liquid Biopsy
-
-
End Use Outlook (Revenue, USD Billion, 2021 - 2033)
-
Hospitals & Cancer Specialty Centers
-
Diagnostic Laboratories (Independent & Reference Labs)
-
Research & Academic Institutes
-
Pharmaceutical & Biotechnology Companies
-
Others
-
-
Country Outlook (Revenue, USD Billion, 2021 - 2033)
-
Saudi Arabia
-
UAE
-
Kuwait
-
Oman
-
Qatar
-
Rest of Middle East
-
Frequently Asked Questions About This Report
b. The Middle East advanced cancer diagnostics market size was estimated at USD 1.32 billion in 2024 and is expected to reach USD 1.43 billion in 2025.
b. The global renal biomarkers market is expected to grow at a compound annual growth rate of 7.30% from 2024 to 2033 to reach USD 2.51 billion by 2033.
b. Imaging-based diagnostics remain the largest segment, contributing a share of 26.8% in 2024, supported by their central role in detection, staging, and treatment monitoring.
b. Some key players operating in the renal biomarkers market include Roche Diagnostics, Illumina, Inc., Thermo Fisher Scientific, Siemens Healthineers, GE HealthCare,Philips Healthcare, Canon Medical Systems, Fujifilm Holdings Corporation, Agilent Technologies, Bruker Corporation, Becton, Dickinson and Company (BD), Bio-Rad Laboratories, Myriad Genetics.
b. Key factors that are driving the market growth are increasing prevalence of cancer, growing demand for early detection, launch of advanced diagnostic technologies, and investment under national healthcare modernization.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










