- Home
- »
- Biotechnology
- »
-
Residual DNA Testing Market Size, Industry Report, 2033GVR Report cover
![Residual DNA Testing Market Size, Share & Trends Report]()
Residual DNA Testing Market (2026 - 2033) Size, Share & Trends Analysis Report By Product & Service (Instruments), By Test Type (Raw Material Testing, Bulk Testing), By Technology (PCR) By Application (Monoclonal Antibodies), By End-user, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-886-1
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
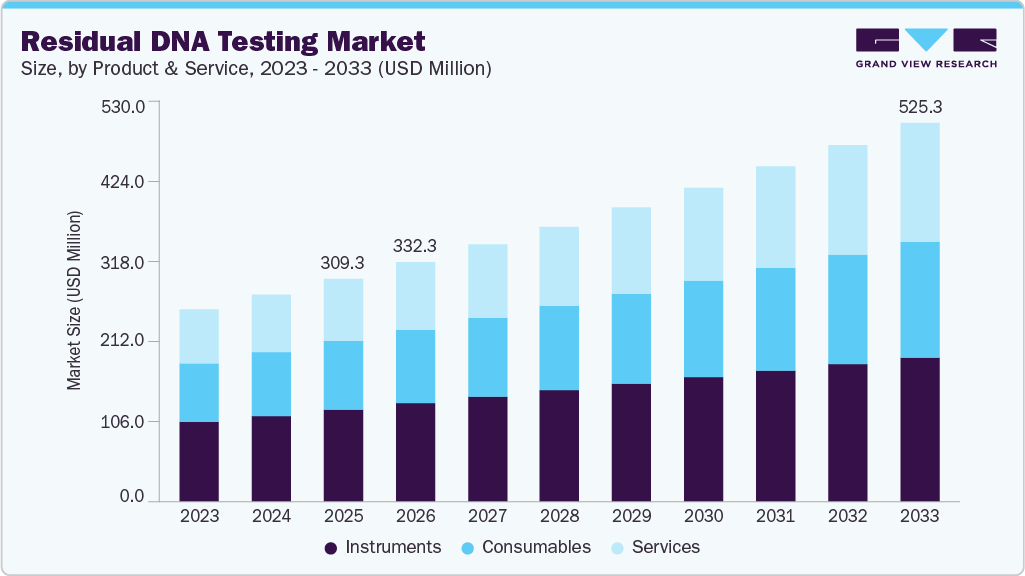
Residual DNA Testing Market Summary
The global residual DNA testing market size was estimated at USD 309.3 million in 2025 and is projected to reach USD 525.3 million by 2033, growing at a CAGR of 6.8% from 2026 to 2033. Growth is driven by stricter regulatory requirements, rising biologics and gene therapy production, and advancements in sensitive DNA detection technologies.
Key Market Trends & Insights
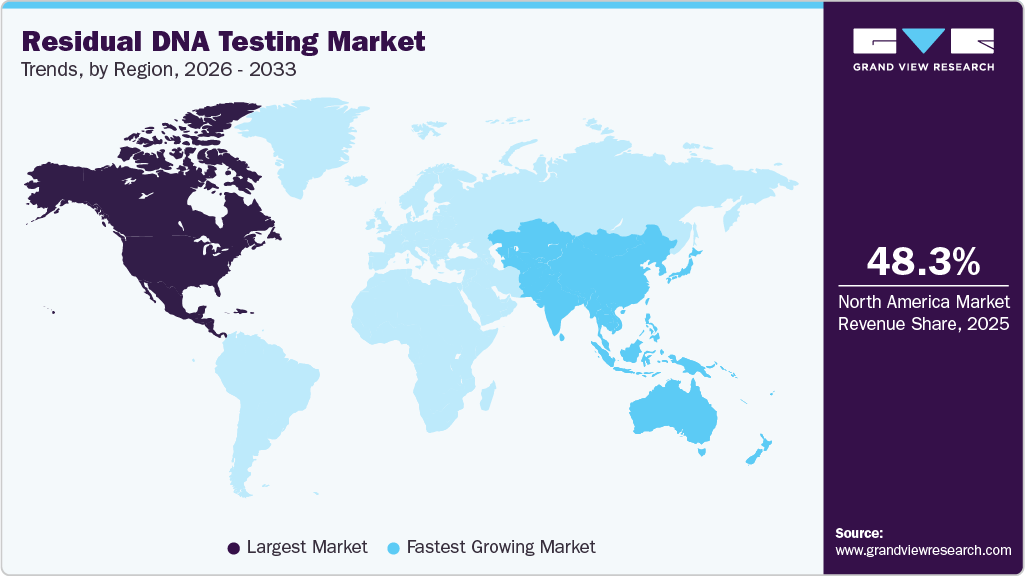
- North America residual DNA testing industry held the largest global market share of 48.3% in 2025.
- The residual DNA testing industry in the U.S. is expected to grow over the forecast period due to the expanding aging population.
- Based on product & service, the consumables segment dominated the market with a share of 41.1% in 2025.
- Based on technology, the PCR segment led the residual DNA testing market in 2025, accounting for the largest revenue share of 42.4%.
- Based on application, the monoclonal antibodies segment led the residual DNA testing market in 2025, accounting for a share of 45.1%.
Market Size & Forecast
- 2025 Market Size: USD 309.3 Million
- 2033 Projected Market Size: USD 525.3 Million
- CAGR (2026-2033): 6.8%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Growth of Biologics and Gene Therapies
The rapid expansion of biologics and gene therapy pipelines is a key driver of the residual DNA testing market. Products such as monoclonal antibodies, vaccines, and especially gene therapies (including genetically modified cell therapies) rely on host systems, increasing the risk of residual DNA contamination and the need for highly sensitive testing to ensure product safety.
Gene therapies (including genetically modified cell therapies)
Product Name
Originator Company
Modality
Disease(s)
Year First Approved
Locations Approved
Vyjuvek
Krystal Biotech
HSV-1 gene therapy
Epidermolysis bullosa
2023
US
Adstiladrin
Merck
Adenoviral gene therapy
Bladder cancer
2022
US
Hemgenix
uniQure
AAV5 gene therapy
Hemophilia B
2022
US, EU, UK
Roctavian
BioMarin
AAV5 gene therapy
Hemophilia A
2022
EU, UK
Upstaza
PTC Therapeutics
AAV2 gene therapy
AADC deficiency
2022
EU, UK
Carvykti
Legend Biotech
CAR-T
Myeloma
2022
US, EU, UK, Japan
Delytact
Daiichi Sankyo
Oncolytic virus
Brain cancer
2021
Japan
Benoda
JW Therapeutics
CAR-T
B-cell lymphoma
2021
China
Abecma
Bluebird Bio
CAR-T
Myeloma
2021
US, EU, UK, Canada, Japan
Breyanzi
Bristol-Myers Squibb
CAR-T
B-cell lymphoma
2021
US, EU, UK, Japan, Canada, Switzerland
Source: Molecular Therapy, Secondary Research, Primary Interviews, Grand View Research
At the same time, rising clinical and commercial-scale manufacturing has strengthened quality control requirements. Regulatory bodies such as the U.S. Food and Drug Administration and European Medicines Agency mandate strict DNA limits, driving adoption of advanced methods like qPCR and NGS, particularly for gene therapies (including genetically modified cell therapies).
Advancements in Detection Technologies
Advancements in detection technologies are significantly driving the residual DNA testing market by enhancing sensitivity, specificity, and throughput. Techniques such as real-time PCR (qPCR), digital droplet PCR (ddPCR), and next-generation sequencing (NGS) enable accurate detection of trace DNA levels in complex biologics and gene therapies. These methods offer precise quantification, faster turnaround times, and improved reproducibility, making them essential for regulatory compliance and product safety.
Moreover, innovations in automation, high-throughput platforms, and advanced data analysis are improving workflow efficiency and reducing errors. Technologies like NGS and emerging PCR-based methods allow large-scale, rapid DNA analysis, expanding applications across biopharmaceutical manufacturing. This improves scalability, lowers operational costs, and supports broader adoption in quality control, further driving market growth.
Market Concentration & Characteristics
The residual DNA testing industry exhibits a high degree of innovation, driven by advancements in sensitive detection technologies such as digital PCR (dPCR) and next-generation sequencing (NGS), which enable precise quantification of trace DNA impurities in complex biologics and gene therapies. Innovation is further supported by automation, high-throughput platforms, and AI-based data analysis, improving efficiency, accuracy, and scalability. For instance, in February 2026, in the United States, NeoGenomics launched RaDaR ST, a ctDNA-based MRD assay for solid tumors, enabling earlier residual disease detection, recurrence monitoring, and supporting personalized treatment decisions across clinical oncology workflows.
The residual DNA testing industry shows a moderate to high level of M&A activity, primarily driven by leading life sciences companies seeking to expand technological capabilities and service offerings. Key players such as Thermo Fisher Scientific, Eurofins Scientific, and Bio-Rad Laboratories активно pursue strategic acquisitions to strengthen portfolios, particularly in advanced platforms like digital PCR and sequencing. Overall, M&A remains focused on capability enhancement and market expansion rather than aggressive consolidation.
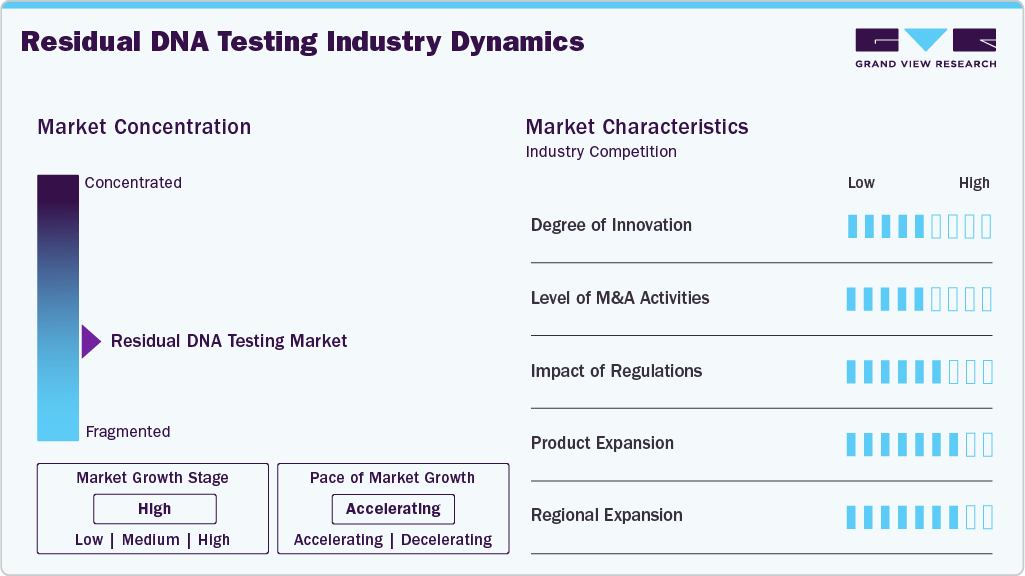
Regulations have a high impact on the residual DNA testing industry, with agencies such as the U.S. Food and Drug Administration and European Medicines Agency enforcing strict limits on residual DNA in biologics and gene therapies. This drives demand for sensitive, validated testing methods and makes regulatory compliance a key market growth driver.
Product expansion in the residual DNA testing industry is steadily increasing, with companies broadening their portfolios to include advanced kits, reagents, and integrated testing solutions. Key players are focusing on developing ready-to-use, high-sensitivity assays and scalable platforms tailored for biologics and gene therapies. This expansion enhances workflow efficiency, supports diverse applications, and strengthens market competitiveness.
Regional expansion in the residual DNA testing industry is gaining momentum as companies extend their footprint into high-growth biopharma hubs such as China and India. This expansion is driven by increasing outsourcing of biologics manufacturing, rising clinical trial activity, and supportive government initiatives. Market players are strengthening local capabilities through facility expansion and strategic collaborations, enabling better access to cost-efficient services and faster turnaround times.
Product & Service Insights
In 2025, the consumables segment led the residual DNA testing market with a 41.1% share, driven by the high and recurring demand for reagents, assay kits, and sample preparation materials required for routine DNA quantification workflows. The increasing volume of biologics, vaccines, and cell and gene therapy production has significantly elevated testing frequency, thereby boosting consumables usage. Moreover, the growing adoption of advanced techniques such as qPCR and next-generation sequencing (NGS) has further strengthened demand for specialized, high-sensitivity consumables, ensuring consistent segment dominance.
The services segment is the fastest-growing in the residual DNA testing market, driven by increasing outsourcing to CROs/CDMOs, rising complexity of biologics and cell & gene therapies, and demand for specialized, regulatory-compliant testing solutions.
Test Type Insights
The final product testing segment led the residual DNA testing market in 2025 with the largest revenue share of 45.0% and is expected to grow at the fastest CAGR. This growth is driven by regulatory requirements, increasing biologics and gene therapy production, and rising adoption of sensitive detection technologies for product safety and compliance.
The bulk testing segment is projected to grow significantly over the forecast period, driven by increasing upstream biologics production, higher demand for process monitoring, and the need for consistent quality control during large-scale manufacturing.
Technology Insights
The PCR segment led the residual DNA testing market in 2025, accounting for the largest revenue share of 42.4%. The demand is driven by its high sensitivity, specificity, and widespread adoption for accurate quantification of residual DNA in biopharmaceutical products. Its dominance is further supported by advancements in real-time and digital PCR technologies, enabling precise and rapid detection. In addition, increasing regulatory emphasis on stringent quality control and the growing pipeline of biologics and gene therapies continue to drive segment growth.
The next-generation sequencing segment is expected to grow at the fastest rate over the forecast period, driven by its ability to provide comprehensive and high-throughput analysis of residual DNA. Increasing adoption in complex biologics and gene therapy workflows, along with advancements in sequencing technologies and declining costs, is further accelerating segment growth.
Application Insights
The monoclonal antibodies segment led the residual DNA testing market in 2025, accounting for a share of 45.1%, driven by the widespread use of mAbs in oncology, autoimmune, and infectious disease therapies. The increasing volume of mAb production and stringent regulatory requirements for residual DNA quantification are further supporting segment dominance.
The cell & gene therapy segment is expected to grow at the fastest rate over the forecast period, driven by the increasing commercialization of advanced therapies and rising investment in biomanufacturing infrastructure. The complexity of these therapies necessitates highly precise residual DNA testing, further supporting segment growth.
End-user Insights
The pharmaceutical & biotechnology companies segment dominated the residual DNA testing market in 2025, accounting for a revenue share of 59.6%, driven by extensive biologics and vaccine production activities. Increasing R&D investments and stringent regulatory requirements for product safety and quality further support the segment’s leading position.
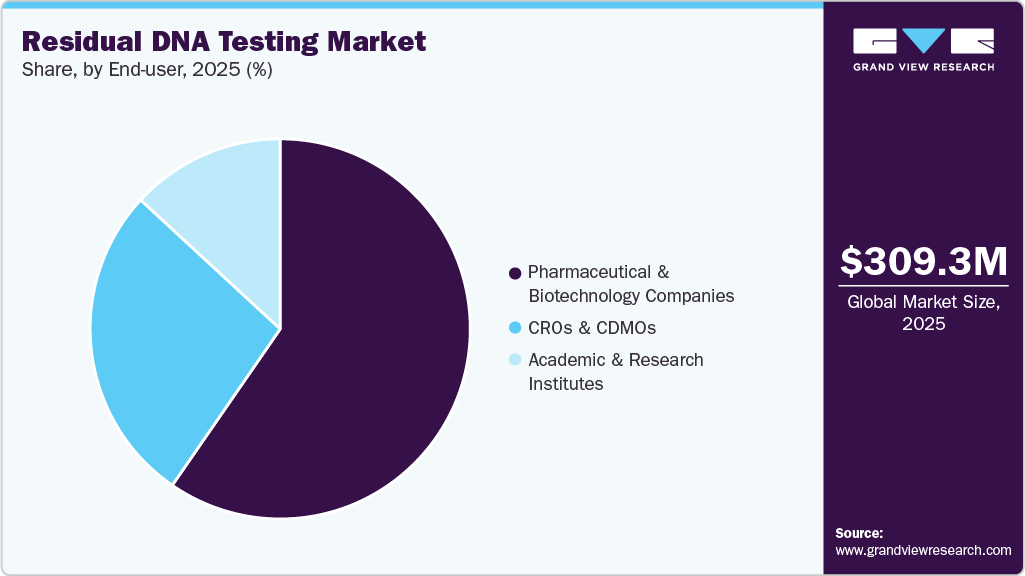
CROs & CDMOs are expected to witness significant growth from 2026 to 2033, driven by increasing outsourcing of biopharmaceutical development and manufacturing activities. Rising demand for specialized testing services, cost efficiency, and scalability offered by these organizations is further accelerating segment growth.
Regional Insights
North America residual DNA testing market dominated the global market and accounted for a 48.3% share in 2025, driven by a strong presence of biopharmaceutical companies, advanced manufacturing infrastructure, and stringent regulatory oversight from agencies. The region benefits from early adoption of advanced analytical technologies and a robust pipeline of gene and cell therapies, supporting consistent demand for high-sensitivity testing solutions.
U.S. Residual DNA Testing Market Trends
The residual DNA testing industry in the U.S. is expected to grow over the forecast period due to the expanding aging population. The U.S. represents the most advanced market, driven by strong innovation in gene and cell therapies, along with widespread adoption of cutting-edge analytical platforms, continues to drive demand for highly sensitive and compliant testing solutions.
Europe Residual DNA Testing Market Trends
Europe represents a mature market characterized by well-established regulatory frameworks led by the European Medicines Agency and increasing focus on quality assurance in biologics production. Growth is supported by rising investments in biosimilars and advanced therapies, along with strong research collaboration across countries such as Germany and UK, driving the adoption of standardized and compliant testing practices.
The UK market demand is driven by robust research infrastructure and growing investments in life sciences and clinical development. Increasing collaboration between academia and industry is fostering innovation in biologics, thereby supporting the need for advanced residual DNA testing solutions.
Germany plays a key role in Europe’s residual DNA testing market, backed by a strong industrial base in biologics manufacturing and a focus on precision and quality standards. The country’s emphasis on biosimilars and advanced therapeutics supports the adoption of reliable and standardized testing methodologies.
Asia Pacific Residual DNA Testing Market Trends
The residual DNA testing industry in Asia Pacific is anticipated to witness the fastest growth at a CAGR of 9.9% from 2026 to 2033, fueled by expanding biomanufacturing capabilities, cost advantages, and increasing clinical trial activity in countries like China, India, and South Korea. The region is also witnessing growing government support and foreign investments, making it a key hub for outsourcing and driving demand for efficient and scalable testing solutions.
China is emerging as a major growth hub, driven by rapid expansion of biopharma manufacturing, increasing clinical trials, and strong government support. The rising presence of domestic biotech firms and cost-efficient production capabilities is accelerating demand for scalable testing solutions.
Japan is witnessing growth due to its well-regulated pharmaceutical sector and focus on high-quality manufacturing practices. The country’s advancements in regenerative medicine and biologics production are contributing to increased adoption of precise DNA impurity testing technologies.
MEA Residual DNA Testing Market Trends
The MEA residual DNA testing industry is emerging, supported by gradual improvements in healthcare infrastructure and increasing focus on biotechnology development in regions like UAE and South Africa. While still at a nascent stage, the market is benefiting from rising awareness of biologics safety and growing investments in pharmaceutical manufacturing, creating opportunities for future growth.
Kuwait represents a developing market, with growth supported by gradual healthcare modernization and increasing interest in pharmaceutical quality standards. While still nascent, rising investments in healthcare infrastructure are expected to create opportunities for specialized testing services.
Key Residual DNA Testing Company Insights
The residual DNA testing industry is moderately consolidated, with global leaders competing on technology, portfolio depth, and regulatory expertise. Key players such as Thermo Fisher Scientific Inc., Merck KGaA, and QIAGEN N.V. hold notable shares due to their strong offerings in reagents, kits, and advanced analytical platforms, supported by extensive R&D and global reach.
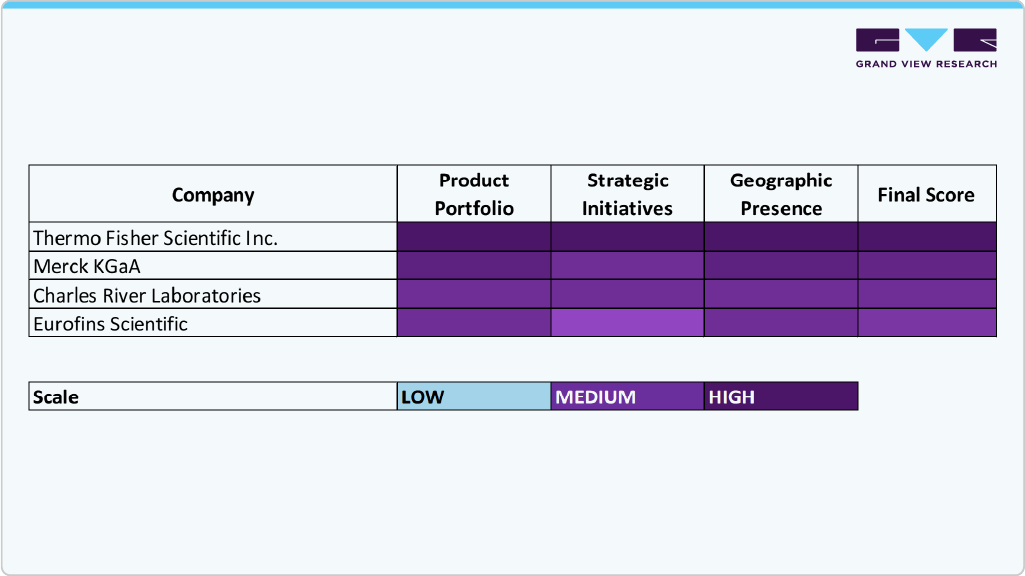
Moreover, companies such as Charles River Laboratories, Eurofins Scientific, and Bio-Rad Laboratories, Inc. contribute through specialized testing services and PCR technologies. Other players including F. Hoffmann-La Roche Ltd., Maravai LifeSciences, FUJIFILM Corporation, and Sartorius AG are expanding capabilities, intensifying competition through innovation and integrated solutions.
Key Residual DNA Testing Companies:
The following key companies have been profiled for this study on the residual DNA testing market.
- Thermo Fisher Scientific Inc.
- Merck KGaA
- Charles River Laboratories
- Eurofins Scientific
- Bio-Rad Laboratories, Inc.
- QIAGEN N.V.
- F. Hoffmann-La Roche Ltd.
- Maravai LifeSciences
- FUJIFILM Corporation
- Sartorius AG
Recent Developments
-
In June 2025, QIAGEN expanded its oncology diagnostics portfolio through strategic partnerships to advance MRD testing capabilities using digital PCR and NGS for clinical and companion diagnostic applications.
-
In May 2024, in the United States, Tempus clinically launched its MRD testing portfolio, including xM MRD and NeXT Personal Dx assays, to enable early cancer recurrence detection and residual disease monitoring.
Residual DNA Testing Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 332.3 million
Revenue forecast in 2033
USD 525.3 million
Growth rate
CAGR of 6.8% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product & service, test type, technology, application, end user, and region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; Thailand; South Korea; Brazil; Argentina; South Africa; Saudi Arabia, UAE; Kuwait
Key companies profiled
Thermo Fisher Scientific Inc.; Merck KGaA; Charles River Laboratories; Eurofins Scientific; Bio-Rad Laboratories, Inc.; QIAGEN N.V.; F. Hoffmann-La Roche Ltd.; Maravai LifeSciences; FUJIFILM Corporation; Sartorius AG
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Global Residual DNA Testing Market Report Segmentation
This report forecasts revenue growth and provides an analysis on the latest trends in each of the sub-segments from 2021 to 2033. For this report, Grand View Research has segmented the residual DNA testing market on the basis of product & service, test type, technology, application, end-user, and region.
-
Product & Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Instruments
-
Consumables
-
Services
-
-
Test Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Raw Material Testing
-
Bulk Testing
-
Final Product Testing
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
PCR
-
Threshold Assays
-
DNA Probe Hybridization
-
Next-Generation Sequencing
-
Others
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Monoclonal Antibodies
-
Vaccines
-
Cell & Gene Therapy
-
Others
-
-
End-user Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical and Biotechnology Companies
-
Academic & Research Institutes
-
CROs & CMOs
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
Japan
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
MEA
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global residual DNA testing market size was estimated at USD 309.3 million in 2025 and is expected to reach USD 332.3 million in 2026.
b. The global residual DNA testing market is expected to grow at a compound annual growth rate of 6.8% from 2026 to 2033 to reach USD 525.3 million by 2033.
b. North America residual DNA testing market dominated the global market and accounted for a 48.3% share in 2025, driven by a strong presence of biopharmaceutical companies, advanced manufacturing infrastructure, and stringent regulatory oversight from agencies.
b. Thermo Fisher Scientific Inc.; Merck KGaA; Charles River Laboratories; Eurofins Scientific; Bio-Rad Laboratories, Inc.; QIAGEN N.V.; F. Hoffmann-La Roche Ltd.; Maravai LifeSciences; FUJIFILM Corporation; Sartorius AG
b. The market is driven by stricter regulatory requirements, rising biologics and gene therapy production, and advancements in sensitive DNA detection technologies.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










