- Home
- »
- Clinical Diagnostics
- »
-
U.S. Allergy And Autoimmune Disease Diagnostics Market 2033GVR Report cover
![U.S. Allergy And Autoimmune Disease Diagnostics Market Size, Share & Trends Report]()
U.S. Allergy And Autoimmune Disease Diagnostics Market (2025 - 2033) Size, Share & Trends Analysis Report By Product & Services (Allergy Diagnostics, Autoimmune Disease), By Test Type, By Diagnostics Type, By End-use, And Segment Forecasts
- Report ID: GVR-4-68040-695-1
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
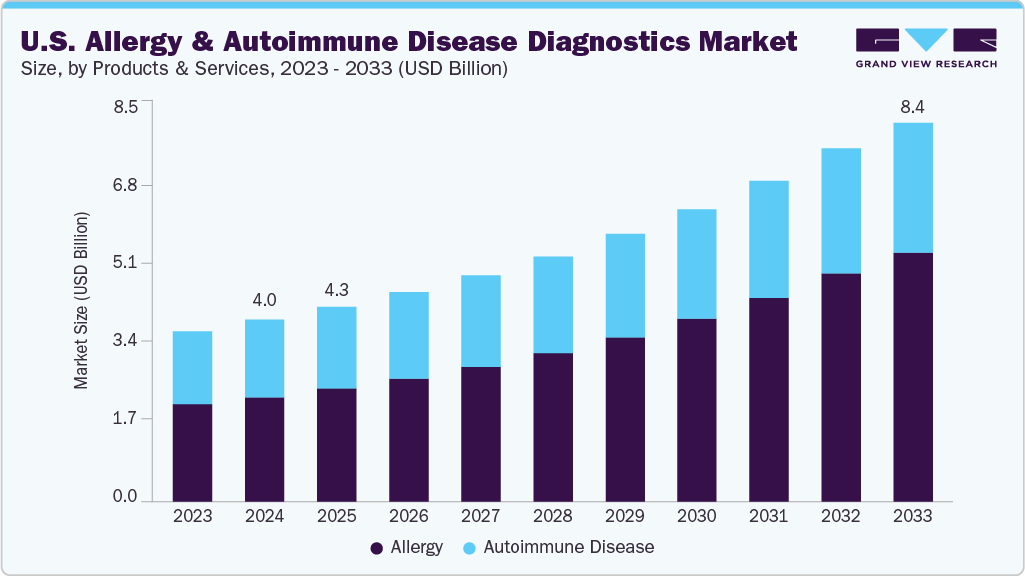
The U.S. allergy and autoimmune disease diagnostics market size was estimated at USD 4.03 billion in 2024 and is projected to reach USD 8.40 billion by 2033, growing at a CAGR of 8.67% from 2025 to 2033. The market is primarily driven by the rising incidence of allergic and autoimmune disorders, increasing patient awareness, and improved access to specialized diagnostic testing. Allergy diagnostics are widely used to identify specific IgE-mediated reactions, while autoimmune disease diagnostics support early detection and differentiation of complex conditions such as rheumatoid arthritis, systemic lupus erythematosus, and multiple sclerosis.
In addition, the growing volume of diagnostic testing for allergy and autoimmune conditions has prompted significant improvements in sample handling, assay throughput, and data reporting. These enhancements support more efficient laboratory operations and expand access to specialized tests. Laboratories are increasingly adopting automation and integrated platforms to manage rising demand, streamline workflows, and maintain accuracy in high-complexity testing.
Digitized reporting systems and connectivity with electronic health records further improve coordination between diagnostic teams and clinicians. These tools reduce turnaround times and support timely clinical decision-making in managing chronic allergic and autoimmune conditions.
The U.S. market is experiencing continuous innovation, driven by research-focused developments and a shift toward more targeted diagnostic solutions. In April 2025, Beckman Coulter Life Sciences introduced its Next-Generation Basophil Activation Test (BAT) to support allergy research. Although designed for research use only, the test reflects a growing emphasis on functional assays that offer deeper insights into immune cell behavior and allergic triggers.
In a related development, WellTheory launched an enterprise solution in October 2023 to address autoimmune care through employer and payer networks. This platform focuses on structured, lifestyle-based management of autoimmune symptoms, underscoring a broader market trend toward integrating diagnostics with ongoing patient support. These efforts reflect how diagnostics are increasingly paired with care delivery models to improve long-term outcomes for individuals with chronic immune conditions.
The U.S. market is evolving toward more specialized, data-driven approaches that align diagnostics with personalized care. As testing technologies become more sophisticated and accessible, the market is expected to benefit from stronger clinical integration, improved patient engagement, and broader adoption across traditional healthcare settings and emerging care platforms.
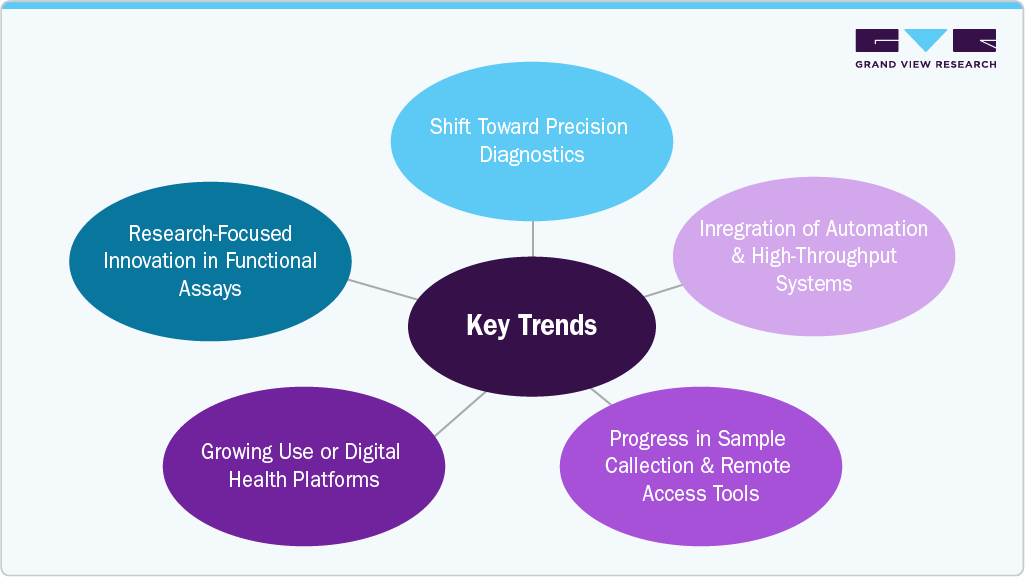
Key Trends Shaping the U.S. Allergy & Autoimmune Disease Diagnostics Market
- Shift Toward Precision Diagnostics:
There is a growing demand for diagnostic tools that provide more accurate, personalized insights into allergic and autoimmune conditions. Biomarker-based assays and multiplex platforms enable earlier detection and more specific differentiation between overlapping immune disorders.
- Integration of Automation and High-Throughput Systems:
Laboratories are increasingly adopting automated analyzers and integrated workflow solutions to improve efficiency and scale testing capacity. This trend supports faster turnaround times and reduces manual errors in complex immunological testing.
- Progress in Sample Collection and Remote Access Tools:
Laboratories and diagnostic developers are improving sample collection methods to support remote allergy and autoimmune testing access. Dried blood spot (DBS) sampling and mail-in collection kits are gaining traction for preliminary screening and longitudinal disease monitoring. These approaches support patient-centric care and increase access in underserved or non-urban areas.
- Growing Use of Digital Health Platforms:
Diagnostic insights are being increasingly integrated into digital care models. Platforms that combine diagnostic results with patient-reported outcomes and care coordination tools are gaining traction in autoimmune disease management.
- Research-Focused Innovation in Functional Assays:
Ongoing research is driving the development of novel functional diagnostics, such as basophil activation tests (BAT) and T-cell response assays. These tools aim to provide more clinically relevant insights into immune activity and hypersensitivity mechanisms.
Market Concentration & Characteristics
The U.S. allergy and autoimmune disease diagnostics industry is marked by continuous innovation driven by expanding immunological research, new biomarker discovery, and evolving testing technologies. Advances in multiplex immunoassays, molecular diagnostics, and lab automation are enhancing diagnostic accuracy and throughput. Functional assays, such as basophil activation tests and T-cell response profiling, are gaining interest for their ability to capture complex immune dynamics. These innovations align diagnostics with precision medicine goals and improve clinical decision-making in allergy and autoimmune care.
Mergers and acquisitions in the U.S. allergy and autoimmune disease diagnostics industry are increasingly focused on expanding specialized testing services, integrating digital platforms, and improving access to immune profiling solutions. Companies target laboratories with expertise in autoantibody panels, allergen-specific IgE testing, and remote diagnostic infrastructure. Strategic partnerships and acquisitions also support adoption within chronic disease management programs. In September 2024, Quest Diagnostics completed the acquisition of select laboratory assets from Allina Health in Minnesota and western Wisconsin, strengthening its ability to deliver comprehensive autoimmune and allergy testing services across a wider regional network. This trend highlights the push to consolidate resources and broaden access to advanced immune diagnostics.
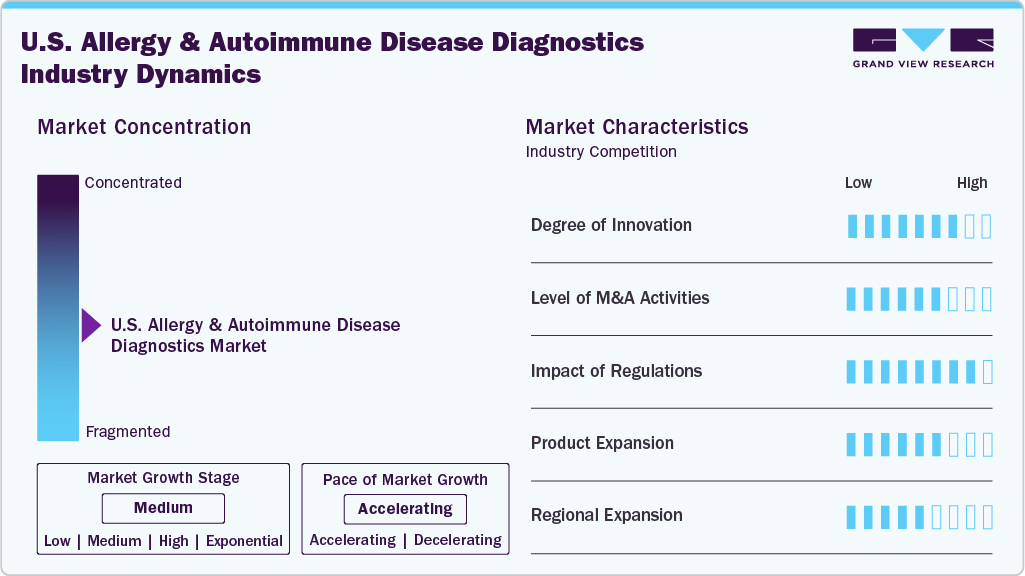
Regulatory oversight is critical in shaping the allergy and autoimmune disease diagnostics industry. Testing services are primarily governed by CLIA regulations, with additional oversight from bodies such as the FDA and CAP for test validation, accuracy, and lab accreditation. The market is also influenced by evolving reimbursement policies for serological and molecular testing under CMS. As demand grows for innovative and multi-analyte assays, companies must navigate complex regulatory pathways to ensure clinical utility, data transparency, and compliance with quality standards.
Manufacturers are broadening their diagnostic product lines to address the growing complexity of allergy and autoimmune testing. The portfolio now features advanced molecular diagnostic kits, multiplex antibody panels, and high-sensitivity assays for early disease detection. Companies are also introducing integrated platforms capable of running combined allergy and autoimmune tests, reducing turnaround time and improving workflow efficiency. These expanded product categories support precision diagnostics and enable laboratories to deliver more comprehensive and accurate results for diverse patient needs.
Regional expansion efforts are focused on increasing access to specialized immune diagnostics across non-metropolitan areas through mail-in collection kits, telehealth integration, and partnerships with regional healthcare providers. As allergy and autoimmune conditions require timely diagnosis and monitoring, expanding infrastructure to underserved areas has become a key market priority. This includes deploying remote testing solutions and digital platforms that allow patients to engage in care without relying solely on centralized lab facilities.
Products & Services Insights
Based on products and services, allergy diagnostics accounted for the largest market share of 57.45% in 2024. The high national burden of allergic conditions such as asthma, allergic rhinitis, and food allergies supports this dominance. Demand for accessible and accurate testing methods has increased across clinical, outpatient, and home-based settings. The segment continues to benefit from rising public awareness, wider test availability, and improvements in in-vitro diagnostic platforms. In addition, the expansion of decentralized care models and the growing use of at-home allergy test kits contribute to segment growth by enabling early detection and monitoring in more convenient, cost-effective formats. In June 2024, Kenota Health received FDA 510(k) clearance and a CLIA waiver for its Point-of-Care Allergy Test System, which allows rapid and accurate allergy testing outside traditional laboratory environments, reinforcing the trend toward decentralized diagnostics. The allergy diagnostics segment is also projected to register the fastest CAGR over the forecast period, driven by continued innovation in self-testing solutions and the increasing integration of allergy testing into digital health platforms and remote care workflows.
The autoimmune disease diagnostics segment is expected to register notable growth during the forecast period, supported by the rising incidence of chronic immune-mediated conditions such as rheumatoid arthritis, systemic lupus erythematosus, and type 1 diabetes in the U.S. This growth is driven by increasing public and clinical awareness, improved access to autoantibody-based testing, and the availability of more comprehensive disease-specific diagnostic panels. Early and accurate detection is becoming increasingly important for guiding long-term disease monitoring and individualized treatment strategies. Advancements in molecular diagnostics, biomarker discovery, and improvements in digital immunoassay platforms are contributing to higher test accuracy and broader adoption across hospitals, specialty clinics, and independent diagnostic laboratories. With autoimmune conditions often presenting overlapping or nonspecific symptoms, the demand for precise diagnostics continues to grow. This segment is gaining traction for its role in enabling more timely diagnoses and expanding clinical options in managing complex autoimmune disorders.
Test Type Insights
The allergy segment held the largest share based on test type in 2024, accounting for 57.45% of the market. This is primarily due to the widespread use of allergy diagnostics to detect reactions to common triggers such as pollen, specific foods, and contact allergens. Commonly used methods-such as specific IgE blood tests, skin prick tests, and patch testing-remain popular across clinical settings because they are easy to perform, reliable, and suitable for routine care. According to the CDC, over 50 million individuals in the U.S. experience allergies annually, making it one of the most common chronic health concerns. With growing awareness, greater interest in early detection, and the emergence of consumer-directed testing models, this segment continues to lead the market. The use of well-established testing protocols and the demand for rapid, straightforward diagnostic results reinforce its strong position.
The autoimmune disease segment is expected to grow steadily over the forecast period due to rising awareness and more cases of long-term immune-related conditions in the U.S. There is also growing demand for early and reliable testing, as many autoimmune diseases have symptoms that overlap with other disorders. Tests such as antinuclear antibody (ANA) panels, autoantibody screens, and molecular-based diagnostics are often used to support accurate diagnosis. In January 2025, Exagen Inc. enhanced its AVISE CTD platform by launching seven new biomarker assays-TC4d, TIgG, TIgM, and anti-RA33 in IgA, IgG, and IgM variants, plus anti-CarP-to improve detection accuracy for systemic lupus erythematosus (SLE) and rheumatoid arthritis (RA), marking a significant product advancement in the autoimmune diagnostics space. Improvements in test accuracy, faster lab systems, and access to specialized testing further support this growth. As interest in personalized care increases, more hospitals, specialty labs, and outpatient centers offer autoimmune diagnostics. Ongoing research in immunology and biomarkers is also helping to expand the range and usefulness of these tests, making this segment an important part of the U.S. diagnostics market.
Diagnostics Type Insights
The allergy diagnostics segment had the highest revenue share in 2024 and is expected to grow the fastest during the forecast period. This growth is supported by greater awareness of allergic conditions and the need for quick and accurate testing. The segment benefits from regular tests like skin prick testing, specific IgE blood tests, and immunoassays, which are known for being fast, easy to use, and reliable in clinical practice. Allergy tests are used in large laboratories and smaller settings like outpatient clinics, and some are now available for home use. New technologies-such as automated allergen panels, better test sensitivity, and digital result tools-are also helping the segment grow. In August 2024, scientists introduced a real-time inflammation monitoring device that tracks immune response through a skin patch, highlighting the trend toward continuous and non-invasive diagnostic tools that can support allergy and immune-related condition management. As more people look to identify allergy triggers early and use at-home options, the demand for allergy diagnostics continues to rise. The combination of simplicity, speed, and flexibility makes this segment a major part of the overall market.
The autoimmune disease diagnostics segment is expected to grow steadily over the forecast period, supported by rising demand for early and accurate detection of complex immune-related conditions. Many of these diseases, such as lupus, rheumatoid arthritis, and celiac disease, have symptoms that are hard to tell apart, making timely diagnosis important for proper treatment. The wider use of advanced testing methods like multiplex immunoassays, indirect immunofluorescence, and molecular diagnostics drives this growth. These tests help find specific autoantibodies and genetic markers linked to autoimmune diseases. New developments-such as high-speed screening tools, AI-based result interpretation, and digital testing systems-are improving how quickly and accurately these conditions can be diagnosed. As healthcare providers focus more on long-term monitoring and personalized treatment plans, the role of autoimmune diagnostics in the overall market continues to grow.
End Use Insights
The allergy segment dominated the U.S. market by end use in 2024 and is expected to grow fastest during the forecast period. Key end users include hospitals and clinics, diagnostic laboratories, and research centers, all of which play important roles in testing and long-term care. Hospitals and clinics remain the primary locations for allergy diagnostics because they serve large patient populations and have access to advanced testing tools. For instance, in June 2025, Mount Sinai Health System received a USD 5 million gift to expand its eczema and allergic conditions services. The funding grows diagnostic capabilities, brings in more clinical staff, and supports allergy-related research. This shows how leading U.S. hospitals are improving their capacity to meet rising demand for allergy testing. Diagnostic laboratories continue to offer high-volume and specialized testing, while research institutions focus on developing more accurate test methods. These end users support the segment’s strong position and continued growth.
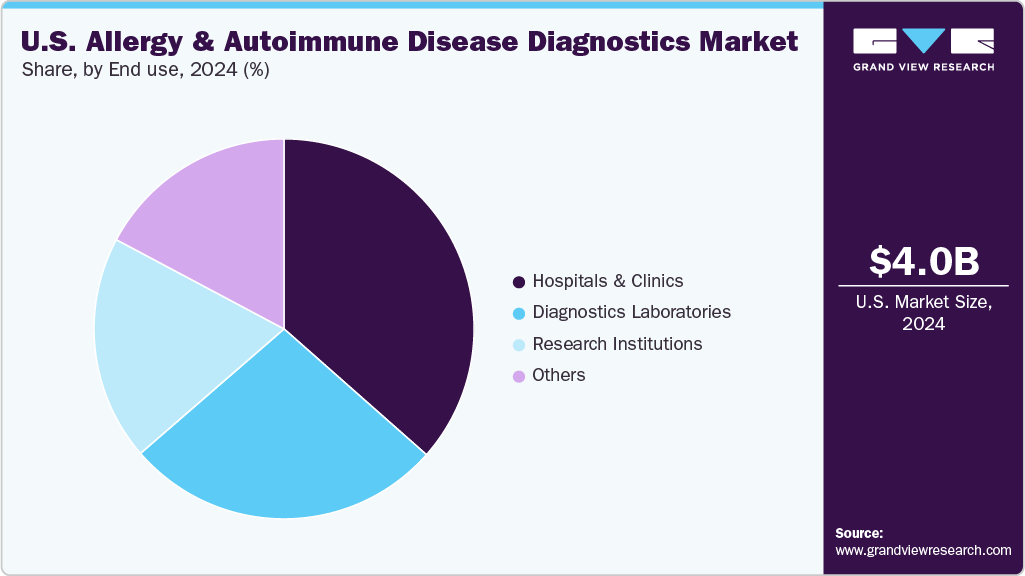
Autoimmune disease is expected to grow strongly over the forecast period by end use. This is mainly due to increasing awareness of immune-related conditions and the need for early and accurate testing. Key end users-hospitals, clinics, diagnostic labs, and research centers-are using more autoimmune tests to improve diagnosis and long-term care. Hospitals and clinics depend on these tests to help identify conditions like rheumatoid arthritis and lupus early, which can lead to better treatment results. Diagnostic labs carry out large volumes of tests using special panels and molecular tools, while research centers help by finding new biomarkers and improving test methods. As testing technology improves and more focus is placed on personalized care, autoimmune diagnostics is expected to grow across all these settings.
Key U.S. Allergy And Autoimmune Disease Diagnostics Company Insights
Some key players in the market focus on innovation, expanding their test offerings, and improving accuracy to stay competitive. These companies are working toward regulatory approvals like FDA clearance to meet standards and increase market access.
They also invest in biomarker research, digital diagnostic tools, and partnerships to support new product development. These efforts aim to boost the use of their tests in both clinical settings and decentralized care environments, such as home testing and outpatient services.
Key U.S. Allergy And Autoimmune Disease Diagnostics Companies:
- Thermo Fisher Scientific, Inc.
- HYCOR Biomedical
- EUROIMMUN Medizinische Labordiagnostika AG (PerkinElmer, Inc.)
- Omega Diagnostics Group PLC
- Lincoln Diagnostics, Inc.
- AESKU.GROUP GmbH
- Minaris Medical America, Inc.
- HOB Biotech Group Corp., Ltd.
- DASIT Group SPA
- R-Biopharm AG
- bioMérieux
- Siemens Healthcare GmbH
- Hoffmann-La Roche Ltd
- Abbott
- Beckman Coulter, Inc.
- Danaher Corporation
- Quest Diagnostics
- Nova Diagnostics Pte Ltd.
Recent Developments
-
In April 2025, U.S.-based Beckman Coulter Life Sciences announced the launch of its Next-Generation BAT to support allergy diagnostics. Designed for research use only, the test offers a safer, non-invasive alternative to traditional oral food challenges by detecting allergen-induced basophil activation through a simple blood draw. This development highlights the growing focus on functional immunoassays in the U.S., supporting improved understanding of allergic responses and paving the way for more precise diagnostic tools in clinical settings.
-
In April 2025, AliveDx submitted a U.S. FDA 510(k) premarket notification for its MosaiQ AiPlex CTDplus multiplex microarray assay, specifically targeting autoimmune connective tissue diseases, such as SLE, RA, systemic sclerosis, Sjögren’s syndrome, and idiopathic inflammatory myopathies. The assay detects up to 1,275 biomarkers per hour and had already earned CE-IVDR certification earlier that year. This submission represents a next-generation, high-throughput tool for more efficient and accurate autoimmune disease diagnosis in U.S. clinical labs.
-
In January 2025, Werfen received FDA 510(k) clearance for its Aptiva APS IgG and IgM reagents, a diagnostic tool designed for detecting antiphospholipid syndrome (APS). Leveraging advanced PMAT (particle-based multi-analyte technology), these reagents allow semi-quantitative detection of anti-cardiolipin and anti-β₂ glycoprotein I antibodies. The system delivers up to 960 test results per eight-hour shift with minimal manual input, marking a significant expansion of the U.S. autoimmune diagnostic reagent offerings.
U.S. Allergy And Autoimmune Disease Diagnostics Market Report Scope
Report Attribute
Details
Revenue forecast in 2033
USD 8.40 billion
Growth rate
CAGR of 8.67% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Products & services, test type, diagnostic type, end use
Key companies profiled
Thermo Fisher Scientific, Inc.; HYCOR Biomedical; EUROIMMUN Medizinische Labordiagnostika AG (PerkinElmer, Inc.); Omega Diagnostics Group PLC; Lincoln Diagnostics, Inc.; AESKU.GROUP GmbH; Minaris Medical America, Inc.; HOB Biotech Group Corp., Ltd.; DASIT Group SPA; R-Biopharm AG; bioMérieux; Siemens Healthcare GmbH; Hoffmann-La Roche Ltd; Abbott; Beckman Coulter, Inc.; Danaher Corporation; Quest Diagnostics; Nova Diagnostics Pte Ltd.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Allergy And Autoimmune Disease Diagnostics Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. allergy and autoimmune disease diagnostics market report based on products & services, diagnostic type, test type, and end use:
-
Products & Services Outlook (Revenue in USD Million, 2021 - 2033)
-
Allergy Diagnostics
-
Instruments
-
Consumables
-
Services
-
-
Autoimmune Disease Diagnostics
-
Instruments
-
Consumables
-
Services
-
-
-
Test Type Outlook (Revenue in USD Million, 2021 - 2033)
-
Allergy Diagnostics
-
In vivo Test
-
Skin Prick Test
-
Intradermal Test
-
Patch Test
-
-
In vitro Test
-
-
Autoimmune Disease Diagnostics
-
Antinuclear Antibody Tests
-
Autoantibody Tests
-
C-reactive Protein (CRP)
-
Complete Blood Count (CBC)
-
Urinalysis
-
Others
-
-
-
Diagnostics Type Outlook (Revenue in USD Million, 2021 - 2033)
-
Allergy Diagnostics
-
Food
-
Dairy Products
-
Poultry Product
-
Tree Nuts
-
Peanuts
-
Shellfish
-
Wheat
-
Soys
-
Other Food Allergens
-
-
Inhaled
-
Drug
-
Other Allergens
-
-
Autoimmune Disease Diagnostics
-
Systemic Autoimmune Disease Diagnostics
-
Rheumatoid Arthritis
-
Ankylosing Spondylitis
-
Systemic Lupus Erythematosus (SLE)
-
Others
-
-
Localized Autoimmune Disease Diagnostics
-
Multiple Sclerosis
-
Type 1 Diabetes
-
Hashimoto's Thyroiditis
-
Idiopathic Thrombocytopenic Purpura
-
Others
-
-
-
-
End Use Outlook (Revenue in USD Million, 2021 - 2033)
-
Allergy Diagnostics
-
Hospitals & Clinics
-
Diagnostics Laboratories
-
Research Institutions
-
Others
-
-
Autoimmune Disease
-
Hospitals & Clinics
-
Diagnostics Laboratories
-
Research Institutions
-
Others
-
-
Frequently Asked Questions About This Report
b. The global U.S. allergy and autoimmune disease diagnostics market size was estimated at USD 4.03 billion in 2024 and is expected to reach USD 5.27 billion in 2025.
b. The global U.S. allergy and autoimmune disease diagnostics market is expected to grow at a compound annual growth rate of 8.67% from 2025 to 2033 to reach USD 8.40 billion by 2027.
b. Based on products and services, the allergy diagnostics segment accounted for the largest market share of 57.45% in 2024 in the U.S. allergy and autoimmune disease diagnostics market. This dominance is supported by the high national burden of allergic conditions such as asthma, allergic rhinitis, and food allergies.
b. Some key players operating in the U.S. allergy and autoimmune disease diagnostics market include Thermo Fisher Scientific, Inc.; HYCOR Biomedical; EUROIMMUN Medizinische Labordiagnostika AG (PerkinElmer, Inc.); Omega Diagnostics Group PLC; Lincoln Diagnostics, Inc.; AESKU.GROUP GmbH; Minaris Medical America, Inc.; HOB Biotech Group Corp., Ltd.; DASIT Group SPA; R-Biopharm AG; bioMérieux; Siemens Healthcare GmbH; Hoffmann-La Roche Ltd; Abbott; Beckman Coulter, Inc.; Danaher Corporation; Quest Diagnostics; Nova Diagnostics Pte Ltd.
b. Key factors that are driving the market growth include rising incidence of allergic and autoimmune disorders, increasing patient awareness, and improved access to specialized diagnostic testing.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










