- Home
- »
- Medical Devices
- »
-
U.S. Clinical Trial Equipment & Ancillary Solutions Market Report 2033GVR Report cover
![U.S. Clinical Trial Equipment & Ancillary Solutions Market Size, Share & Trends Report]()
U.S. Clinical Trial Equipment & Ancillary Solutions Market (2025 - 2033) Size, Share & Trends Analysis Report By Type (Sourcing, Supply/Logistics, Service), By Phase (Phase I, II, III, IV), And Segment Forecasts
- Report ID: GVR-4-68040-686-6
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
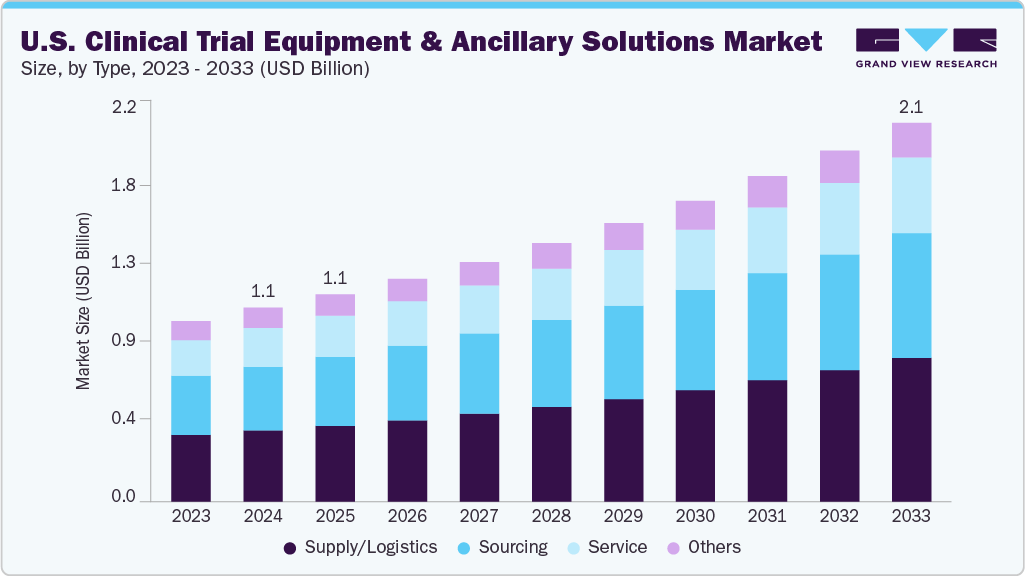
The U.S. clinical trial equipment & ancillary solutions market size was estimated at USD 1.05 billion in 2024 and is projected to reach USD 2.06 billion by 2033, growing at a CAGR of 7.83% from 2025 to 2033. The growth is due to the increasing complexity of clinical trial protocols, which has heightened the need for precision in equipment logistics, device calibration, and timely distribution of trial-related supplies. Sponsors are under growing pressure to meet tight regulatory and study timelines, which in turn has increased reliance on third-party providers specializing in ancillary supply chain management, kitting, equipment leasing, and reverse logistics.
The increasing decentralization of clinical trials in the U.S. is a major driver for the demand for equipment and ancillary solutions. Traditional centralized trials are being replaced or supplemented by hybrid and fully decentralized models, which require extensive logistical coordination and specialized equipment deployment at participants’ homes or local sites. This shift has created a need for direct-to-patient (DTP) supply chains, mobile health kits, connected medical devices, and home-use diagnostics that must be compliant with FDA standards. For instance, the rise of remote ECG monitors, wearable biosensors, and digital spirometers has necessitated ancillary providers capable of managing device calibration, patient training, and data integration remotely. The logistical complexity of supporting multiple remote sites and participants requires real-time tracking, automated resupply systems, and robust reverse logistics - all of which are fueling investments into ancillary solution providers offering end-to-end support tailored for decentralized trials.
The surge in precision medicine and complex therapeutic areas such as oncology, neurology, and gene therapy trials is also propelling the growth of the clinical trial equipment & ancillary solutions industry in the U.S. These trials often demand highly specific equipment like infusion pumps for cellular therapies, customized specimen collection kits, and temperature-controlled drug delivery devices. Ancillary providers are expected to handle specialized packaging, pre-conditioning of equipment, and maintenance protocols to preserve the integrity of temperature- and time-sensitive supplies. The increase in protocol complexity - including biomarker assessments, digital endpoints, and longitudinal sampling - has made the standardization and scalability of ancillary services critical. In response, vendors are integrating cloud-based inventory management, automated labeling, and compliance-tracking tools to streamline processes and meet the heightened demands of sponsors and CROs. This alignment with advanced therapeutic trials is expanding the market beyond basic logistics into high-value clinical trial support infrastructure.
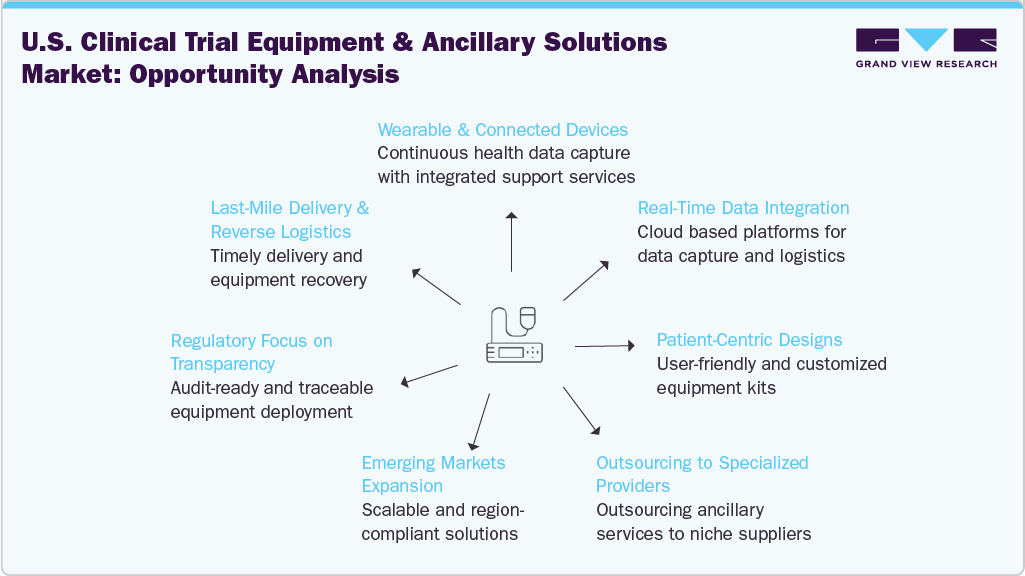
Opportunity Analysis
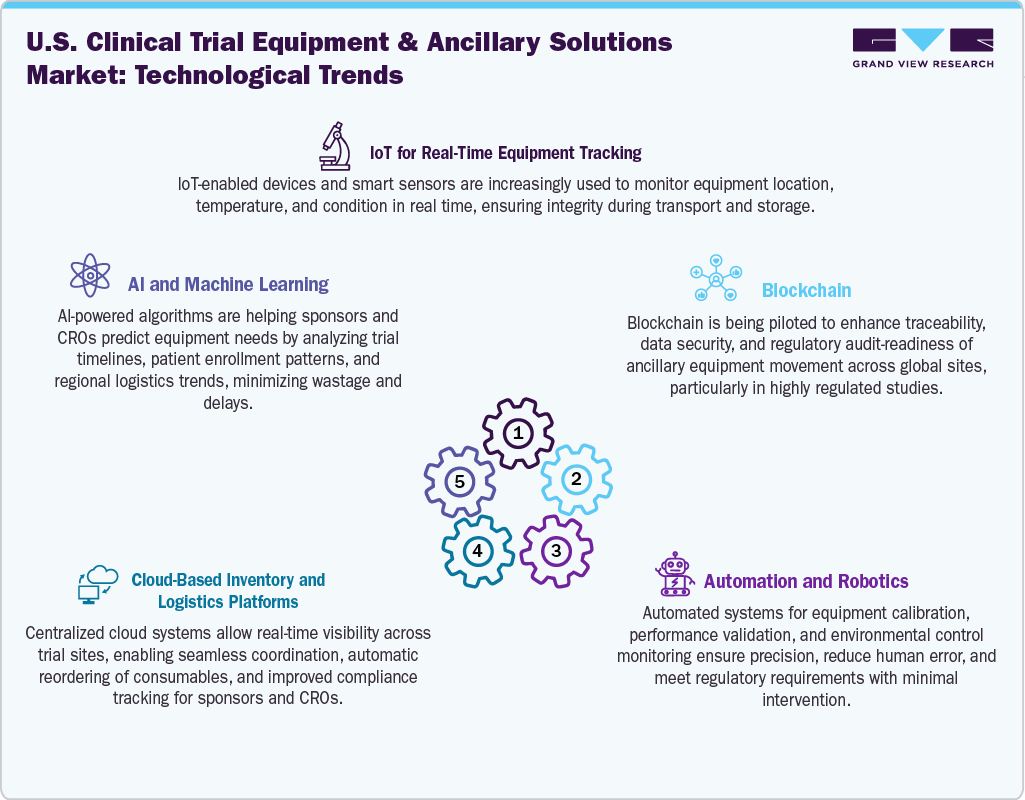
Technological Advancements
The technological landscape of the U.S. clinical trial equipment & ancillary solutions market is rapidly evolving, driven by innovations aimed at enhancing efficiency, compliance, and patient engagement. Digital integration is at the forefront, with cloud-based inventory management, IoT-enabled tracking devices, and AI-powered demand forecasting tools becoming increasingly standard. The use of wearable health tech, remote monitoring equipment, and smart kits is enabling seamless data capture and reducing the need for on-site visits in decentralized trials. Automation in equipment calibration, temperature-controlled logistics, and packaging validation is also improving accuracy and reducing manual intervention. Moreover, blockchain and RFID technologies are being explored to ensure secure, transparent, and traceable handling of critical clinical supplies across geographies. This shift toward connected, intelligent systems is not only streamlining trial operations but also opening new opportunities for vendors to offer bundled, end-to-end ancillary solutions tailored to modern trial demands.
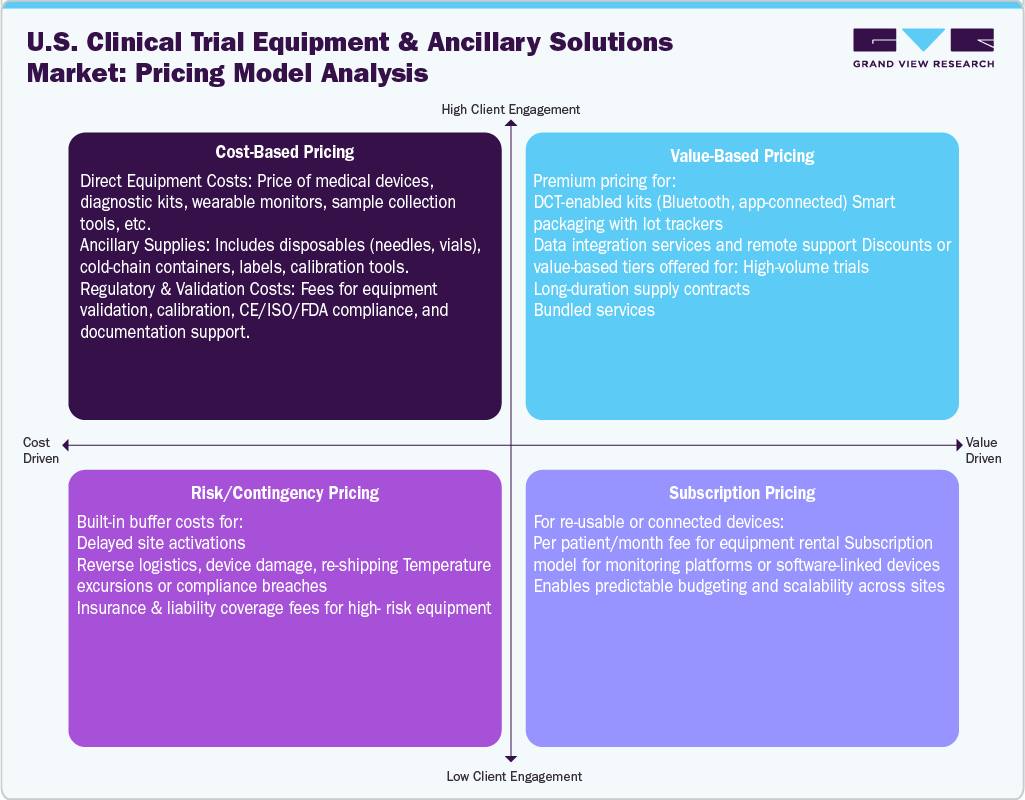
Pricing Analysis
Type Insights
Based on type, the U.S. clinical trial equipment & ancillary solutions market is classified into sourcing, supply/logistics, service, and others. The supply/logistics segment accounted for the largest revenue share of 36.7% in 2024. The growth is due to rising complexity and scale of clinical trials requiring highly coordinated distribution networks. As multi-center and decentralized trials become more prevalent, sponsors increasingly rely on specialized logistics providers to ensure timely delivery of temperature-sensitive drugs, medical devices, specimen kits, and diagnostic tools across diverse geographies.
The sourcing segment is anticipated to grow at a considerable CAGR during the forecast period. The segment growth is driven due to the rising complexity of trial protocols and the increasing diversity of required ancillary materials. Clinical trials today require a wide array of equipment, including wearable devices, point-of-care diagnostics, customized sample kits, and region-specific medical supplies-prompting sponsors and CROs to outsource sourcing functions to specialized vendors with global procurement networks.
Phase Insights
Based on phase, the U.S. clinical trial equipment & ancillary solutions industry is divided into Phase I, Phase II, Phase III, and Phase IV. The phase III segment held the largest market share in 2024, due to its expansive scale, higher patient enrollment, and greater logistical demands compared to earlier phases. Phase III trials typically involve hundreds to thousands of participants across multiple sites and often span longer durations, necessitating a broad range of equipment, diagnostic kits, wearable monitors, and cold chain logistics solutions.
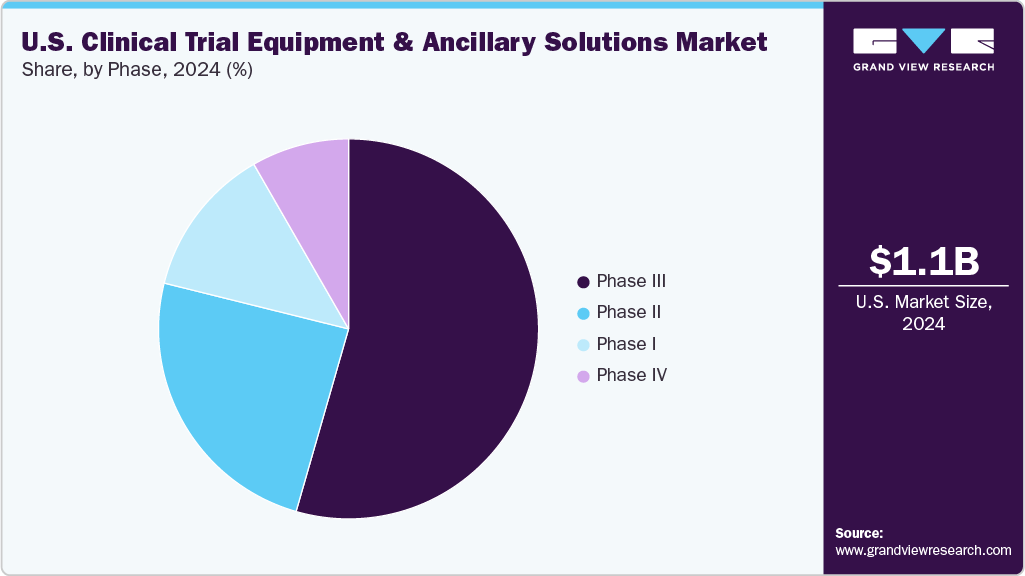
The phase I segment is anticipated to grow at the fastest CAGR over the forecast period. The growth is attributed to an increasing number of early-stage clinical trials, particularly in oncology, rare diseases, and cell and gene therapies. These trials often require specialized and customized equipment, such as first-in-human (FIH) dosing kits, rapid-response biosample collection tools, and highly controlled infusion devices, which demand high-precision sourcing and handling.
Key U.S. Clinical Trial Equipment & Ancillary Solutions Company Insights
Several key players are acquiring various strategic initiatives to strengthen their market position offering diverse services to customers. The prominent strategies adopted by companies are service launches, mergers & acquisitions/joint ventures merger, partnership & agreements, expansions, and others to increase market presence & revenue and gain a competitive edge drives the market growth.
Key U.S. Clinical Trial Equipment & Ancillary Solutions Companies:
- Ancillare, LP
- Imperial CRS, Inc.
- Woodley Equipment Company Ltd.
- Thermo Fisher Scientific, Inc.
- Parexel International (MA) Corporation
- Emsere (formerly MediCapital Rent)
- Quipment SAS
- IRM
- Marken (UPS-United Parcel Service)
- Myonex
- Yourway
Recent Developments
-
In January 2025, Ancillare announced the launch of its state-of-the-art cold chain management service capabilities for clinical trial supplies. The enhancements focus on maintaining temperature integrity, expanding global logistics capabilities, and incorporating real-time tracking and visibility tools.
-
In May 2024, Emsere entered into a partnership agreement with the Clario to enhance ophthalmic imaging capacity in clinical trials by integrating Clario’s clinical trial software with Emsere’s barcoded imaging devices. The expansion addresses needs in retina-focused ophthalmology studies, improving equipment provision and imaging workflow efficiency.
U.S. Clinical Trial Equipment & Ancillary Solutions Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 1.13 billion
Revenue forecast in 2033
USD 2.06 billion
Growth rate
CAGR of 7.83% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, phase
Country scope
U.S.
Key companies profiled
Ancillare, LP; Imperial CRS, Inc.; Woodley Equipment Company Ltd.; Thermo Fisher Scientific, Inc.; Parexel International (MA) Corporation; Emsere (MediCapital Rent); Quipment SAS; IRM; Marken (UPS-United Parcel Service); Myonex; Yourway
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Clinical Trial Equipment & Ancillary Solutions Market Report Segmentation
This report forecasts revenue growth at the country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. clinical trial equipment & ancillary solutions market report based on type, and phase:
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Sourcing
-
Procurement
-
Equipment
-
Ancillaries
-
-
Rental
-
Equipment
-
Ancillaries
-
-
-
Supply/Logistics
-
Transportation
-
Packaging
-
Others
-
-
Service
-
Calibrations
-
Equipment servicing
-
Others
-
-
Others
-
-
Phase Outlook (Revenue, USD Million, 2021 - 2033)
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
Frequently Asked Questions About This Report
b. The U.S. clinical trial equipment & ancillary solutions market size was estimated at USD 1.05 billion in 2024 and is expected to reach USD 1.13 billion in 2025.
b. The U.S. clinical trial equipment & ancillary solutions market is expected to grow at a compound annual growth rate of 7.83% from 2025 to 2033 to reach USD 2.06 billion by 2033.
b. Supply/logistics segment dominated the U.S. clinical trial equipment & ancillary solutions market with a share of 36.7% in 2024. This is attributable to rising complexity and scale of clinical trials requiring highly coordinated distribution networks.
b. Some key players operating in the U.S. clinical trial equipment & ancillary solutions market include Ancillare, LP, Imperial CRS, Inc., Woodley Equipment Company Ltd., Thermo Fisher Scientific, Inc., Parexel International (MA) Corporation, Emsere (MediCapital Rent), Quipment SAS, IRM, Marken, Myonex, Yourway
b. Key factors that are driving the market growth include increasing complexity of clinical trial protocols, which has heightened the need for precision in equipment logistics, device calibration, and timely distribution of trial-related supplies.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










