- Home
- »
- Clinical Diagnostics
- »
-
Asia Pacific Oncology Based Molecular Diagnostics Market 2033GVR Report cover
![Asia Pacific Oncology Based Molecular Diagnostics Market Size, Share & Trends Report]()
Asia Pacific Oncology Based Molecular Diagnostics Market (2025 - 2033) Size, Share & Trends Analysis Report By Type (Breast Cancer, Lung Cancer), By Product (Instruments, Reagents), By Technology (PCR, Sequencing), By Region, And Segment Forecasts
- Report ID: GVR-4-68040-201-5
- Number of Report Pages: 136
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
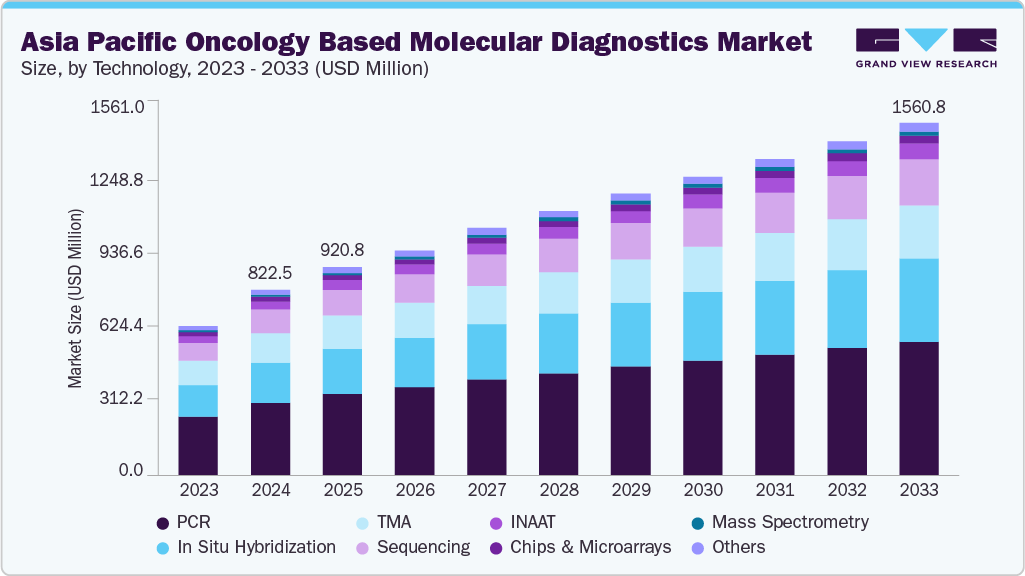
The Asia Pacific oncology based molecular diagnostics market size was estimated at USD 822.5 million in 2024 and is projected to reach USD 1,560.75 million by 2033, growing at a CAGR of 6.8% from 2025 to 2033. Rising cancer burden across breast, prostate, and colon, along with the aging population, is driving the demand for effective diagnostic solutions in the region. Advancements in technology, the growing adoption of point-of-care testing (POCT), and developments in biomarker identification systems are supporting market expansion. In addition, government initiatives aimed at cancer screening and early detection, as well as increased healthcare investments across emerging economies, are contributing to the rising demand for molecular diagnostics.
The increasing requirement for accurate and rapid testing methods and the growing integration of Next-Generation Sequencing (NGS) techniques for oncology diagnostics further support the market. Awareness of personalized medicine and the adoption of targeted treatment strategies are also influencing market trends. Expanding research and development efforts and collaborations to develop innovative molecular diagnostic solutions are expected to strengthen the regional market outlook through 2033.
Growing healthcare infrastructure, particularly in countries like China, India, and Japan, is boosting the accessibility and adoption of advanced diagnostic technologies. Rising partnerships between diagnostic laboratories and healthcare institutions and the focus on early cancer detection programs are fostering regional growth. Furthermore, the increasing shift toward non-invasive testing methods, such as liquid biopsies, and the availability of advanced genetic testing platforms are expected to create significant opportunities for the market in the coming years.

Overall, the Asia Pacific market is evolving with innovations like liquid biopsies, advanced NGS platforms, AI-driven analytics, and portable molecular testing devices that make cancer detection faster and more accurate. Introducing multi-omics approaches and strategic collaborations further supports personalized treatment and early detection initiatives. However, factors such as high costs of advanced technologies, uneven access across rural areas, and regulatory complexities may influence the pace of widespread adoption in the region.
Market Concentration & Characteristics
Innovation is high in the Asia Pacific oncology based molecular diagnostics industry, focusing on early detection, personalized treatment, and non-invasive diagnostic approaches. Advancements in next-generation sequencing (NGS), digital PCR, and AI-powered data analysis improve diagnostic precision and turnaround times. Technologies such as liquid biopsy, RNA sequencing, and tumor mutation burden profiling are gaining traction across various cancer types. Integrating molecular diagnostics with digital health platforms and clinical decision support tools further enhances real-time clinical applicability.
M&A activity in the region is moderate, with larger diagnostics and life sciences companies acquiring smaller firms specializing in bioinformatics, liquid biopsy, and tumor profiling. Collaborations between diagnostic developers and pharmaceutical companies are increasing, particularly for companion diagnostics that support targeted therapies. These strategic deals aim to broaden testing capabilities, strengthen oncology-focused portfolios, and accelerate regulatory and geographic expansion across Asia Pacific.
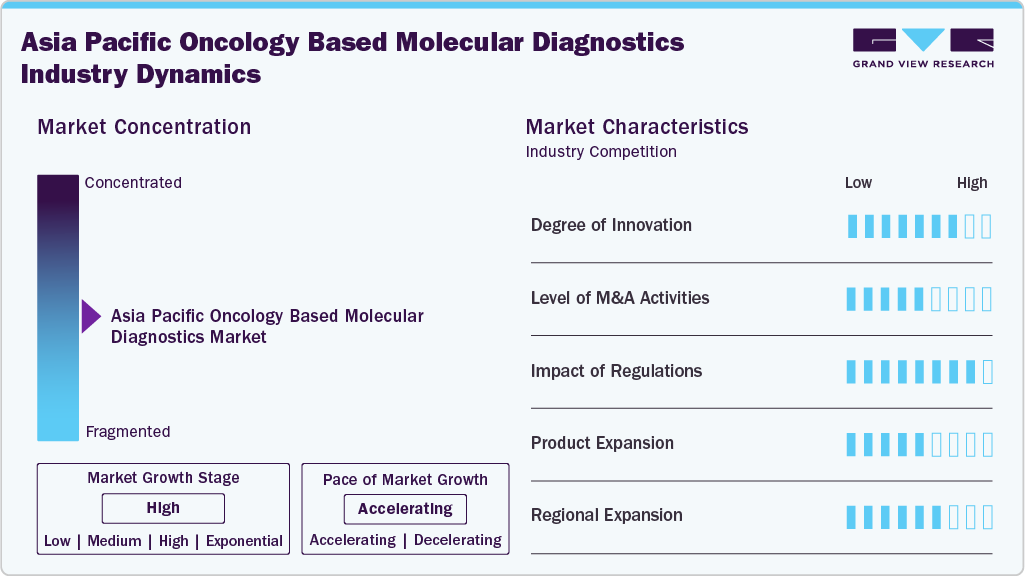
Regulatory frameworks across countries like Japan, China, and Australia are becoming more structured, with stringent requirements for analytical performance and clinical validation of oncology diagnostics. While approval processes can be complex, government initiatives and faster regulatory pathways for innovative tests, including liquid biopsies and companion diagnostics, support market growth. Emphasis on clinical utility and real-world evidence remains key for regulatory approvals in this sector. Product expansion is robust, with companies introducing tumor-specific genomic panels, multi-gene testing solutions, and companion diagnostics tailored for cancer therapies. Innovations in liquid biopsy platforms and minimal residual disease (MRD) monitoring tools are also advancing. Automation of testing workflows, improved turnaround times, and integration with electronic health records (EHRs) enhance operational efficiency for laboratories and hospital-based diagnostic facilities.
Regional expansion is active across high-growth markets such as China, India, and Southeast Asia, supported by partnerships with local diagnostic laboratories, research institutes, and healthcare providers. Rising healthcare expenditure, cancer screening programs, and increasing adoption of precision oncology are creating opportunities for broader test availability. While developed markets like Japan lead in adoption, emerging economies in the region are witnessing accelerated uptake due to improved healthcare infrastructure and awareness campaigns.
Type Insights
On the basis of type, breast cancer accounted for the largest market share at 19.64% in 2024. This leadership is driven by the rising cancer burden across the Asia Pacific and a growing emphasis on personalized oncology care. Molecular diagnostics-particularly tests identifying biomarkers such as HER2 and BRCA1/2 mutations-play a critical role in guiding targeted therapies. Expanding national screening programs, broader insurance coverage, and a preference for minimally invasive testing further bolster this segment.
In 2025, Asia Pacific clinical experts issued a consensus on improving diagnostic standards for HER2‑negative early breast cancer, emphasizing the need to strengthen genetic testing and counseling infrastructure across the region. This initiative reflects a coordinated regional effort to advance molecular diagnostics for breast cancer-from advanced laboratories in Japan and South Korea to emerging testing centers in India and Southeast Asia-highlighting their growing importance in early detection and treatment decision-making throughout Asia Pacific.
On the other hand, liver cancer and prostate cancer diagnostics markets are also anticipated to witness notable growth, alongside colorectal cancer, owing to the rising incidence rates and increasing awareness of early screening across the Asia Pacific. Molecular diagnostics are essential in identifying gene mutations, tumor markers, and other molecular signatures, pivotal in guiding personalized treatment strategies.
Broad opportunities exist for developing molecular-level diagnostic technologies that support precision medicine and accelerate new drug discovery. The growing focus on biomarker-driven therapies, combined with initiatives aimed at improving access to advanced diagnostic solutions in emerging markets, is expected to expand the adoption of oncology-based molecular diagnostics for these cancer types further.
Technology Insights
The polymerase chain reaction (PCR) technology segment held the highest revenue share of 39.42% in 2024, owing to its proven effectiveness in detecting cancer-specific genetic mutations with high sensitivity and speed. PCR continues to be widely utilized in oncology diagnostics due to its cost efficiency, quick turnaround time, and ability to amplify even trace amounts of DNA or RNA from tumor samples. It is key in early detection, treatment monitoring, and minimal residual disease assessment across various cancer types. Despite the rising adoption of advanced technologies such as next-generation sequencing (NGS), PCR maintains its dominance in clinical workflows because of its broad accessibility, scalability, and seamless integration into routine diagnostic processes across advanced and resource-limited healthcare settings.
The sequencing segment is expected to register a strong growth rate during the forecast period, driven by its capability to deliver comprehensive genomic insights for cancer diagnosis, treatment planning, and disease monitoring. Next-generation sequencing (NGS) supports simultaneous analysis of multiple cancer-associated genes with high sensitivity, enabling more precise and personalized therapeutic approaches. This growing adoption of precision oncology is transforming cancer care workflows across the Asia Pacific.
Rapid advances in national precision medicine programs are accelerating regional NGS deployment. For example, Singapore’s National Precision Medicine program aims to sequence 100,000 genomes by 2026 using Illumina NovaSeq platforms, enhancing cancer diagnostics. These initiatives highlight how public‑private collaborations across Asia Pacific are embedding NGS into oncology diagnostics and strengthening regional capacity for precision medicine. Ongoing improvements in sequencing speed, accuracy, and data analytics-alongside increasing reimbursement coverage in countries like South Korea and Australia-are positioning sequencing as a key growth driver for the oncology-based molecular diagnostics market in Asia Pacific through 2033.
Product Insights
The reagents segment held the highest share in 2024, driven by their essential role in molecular diagnostic workflows across diverse cancer types within the Asia Pacific. Reagents-such as enzymes, primers, probes, and buffers-are foundational components for techniques like PCR, next-generation sequencing (NGS), and in situ hybridization, all widely utilized in cancer diagnostics across the region. The rising cancer burden, growing diagnostic test volumes in clinical laboratories, and the increasing adoption of personalized oncology approaches significantly boost the demand for reagents in Asia.
Reagents ensure tests' accuracy, sensitivity, and reproducibility, which are critical for cancer detection and monitoring. With laboratories across countries like China, Japan, and India adopting automated and high-throughput platforms, the demand for specialized and compatible reagent kits continues to grow. Ongoing innovations in reagent formulations-supporting multiplex assays, faster reaction times, and improved stability-strengthen the segment's growth and cement reagents as a core revenue contributor.
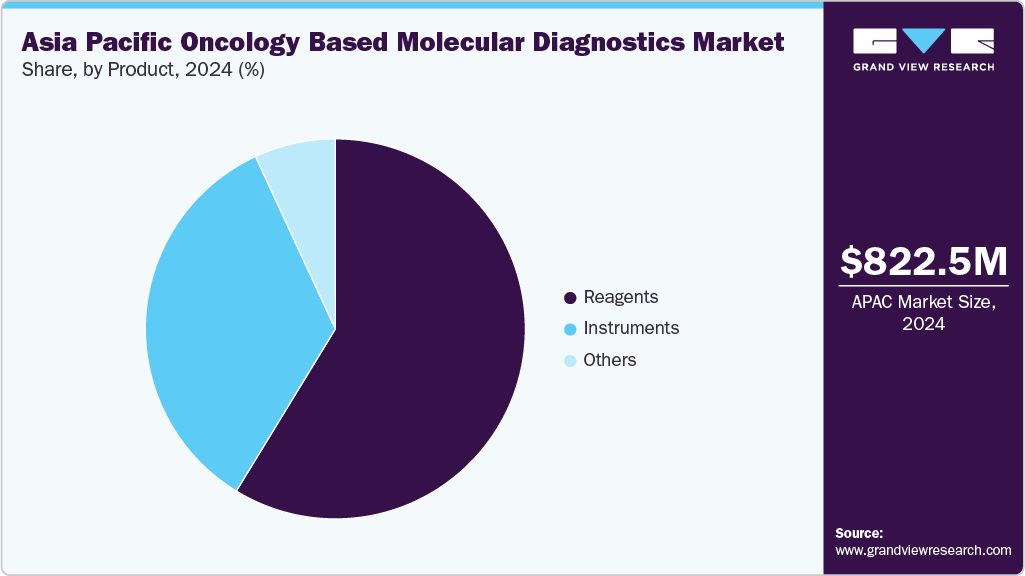
The reagents segment is also anticipated to expand at a significant CAGR over the forecast period, supported by the increasing number of oncology diagnostic tests and the rising adoption of molecular assays across Asia. Growing preference for high-performance reagents compatible with PCR and next-generation sequencing (NGS) platforms is fueling demand, particularly as laboratories focus on achieving accurate and rapid cancer detection. Continuous advancements in reagent formulations-improving efficiency, stability, and multiplexing capabilities-further strengthen the segment’s role in supporting personalized oncology diagnostics and high-throughput testing environments.
Country Insights
Japan oncology based molecular diagnostics market accounted for a revenue share of 22.96% in 2024, propelled by the country’s advanced healthcare infrastructure, rising cancer burden, and early adoption of precision medicine. Molecular diagnostic tools are strongly utilized for identifying genetic mutations, tumor profiling, and guiding targeted therapies. Government initiatives supporting early cancer screening and favorable reimbursement frameworks for molecular testing are further enhancing market momentum.
A key example of Japan’s leadership in genomics-driven oncology is the LC‑SCRUM‑Asia network, initiated initially as LC‑SCRUM‑Japan in 2013 and expanded to a 150‑hospital national lung cancer genomic screening and clinical development initiative in 2024 through the LC‑SCRUM‑CD program. This network has facilitated genomic profiling for thousands of lung cancer patients and played a pivotal role in biomarker‑matched clinical trials and regulatory approvals for novel cancer drugs and diagnostics in Japan. Ongoing government-industry collaboration and continuous integration of advanced platforms like PCR and NGS position Japan to maintain a leading edge in the Asia Pacific’s molecular diagnostics landscape.
India Oncology Based Molecular Diagnostics Market Trends
The oncology based molecular diagnostics market in India is expected to grow significantly, driven by the rising cancer burden, increasing awareness of early detection, and expanding access to advanced diagnostic technologies. The adoption of molecular diagnostic tools for identifying genetic mutations and tumor markers and guiding targeted therapies is gaining momentum due to the growing demand for personalized treatment. Government initiatives to enhance cancer screening programs and develop advanced healthcare infrastructure further support market expansion.
In May 2025, the Telangana health department launched an AI-based cancer screening pilot targeting oral, breast, and cervical cancers across three districts. The initiative integrates high-resolution imaging with AI diagnostics to detect early-stage cancers, with scans reviewed by specialists at MNJ Cancer Institute and NIMS. Following the pilot, the program aims to expand to all medical colleges and district-level day-care cancer centers, highlighting India’s growing efforts to integrate technology and diagnostics for early cancer detection.
In addition, the Tata Memorial Centre uses Natera’s Signatera ctDNA-based test to detect minimal residual disease (MRD) in colorectal cancer, addressing a projected 1.7 million annual cancer cases by 2035. This initiative underscores the role of liquid biopsy and NGS-based technologies in enhancing early detection, treatment monitoring, and personalized cancer management. Such advancements and rising precision oncology initiatives are expected to accelerate the adoption of oncology-based molecular diagnostics in India through 2033.
China Oncology Based Molecular Diagnostics Market Trends
The oncology based molecular diagnostics market in China is expected to grow lucratively over the forecast period, driven by increasing cancer prevalence, a strong focus on early detection, and the expansion of precision oncology programs. China’s universal healthcare system, encompassing basic medical insurance, critical illness coverage, and urban-rural medical assistance, enhances access to advanced diagnostic services. Rising government initiatives, including national cancer screening programs and substantial funding for genomics research, are further propelling the adoption of molecular diagnostics across various cancer types.
Key Asia Pacific Oncology Based Molecular Diagnostics Company Insights
Key participants in the Asia Pacific oncology-based molecular diagnostics market focus on strategies such as expanding their test portfolios, collaborating with hospitals and cancer research centers, and investing in advanced technologies like next-generation sequencing (NGS) and liquid biopsy. These initiatives aim to improve diagnostic accuracy, enhance access to personalized oncology testing, and align with evolving clinical practices.
Key Asia Pacific Oncology Based Molecular Diagnostics Companies:
- Abbott
- Agilent Technologies, Inc
- bioMerieux SA
- Danaher
- F. Hoffmann-La Roche Ltd.
- Illumina
- Novartis AG
- QIAGEN
- Siemens Healthineers AG
- TBG Diagnostics Limited
- Thermo Fisher Scientific Inc
- Sysmex
Recent Developments
-
In May 2025, researchers at the Indian Institute of Technology, Indore (IIT-I), unveiled a Quantum AI-based sequencing technology. This innovation combines explainable AI with solid-state nanopore sequencing to enable rapid and accurate detection of genetic mutations, potentially surpassing traditional NGS in speed and cost-efficiency. Offering high-resolution genomic insights, this development is expected to advance the future of molecular diagnostic tools in India, supporting precision oncology and personalized treatment approaches.
-
In April 2025, Promega’s ProDx MSI Detection Kit received an innovative medical device registration certificate from China’s National Medical Products Administration (NMPA). This PCR-based assay detects microsatellite instability (MSI) in colorectal cancer tissue, facilitating the identification of patients who may require further genetic testing for Lynch syndrome. This milestone enhances the accessibility of advanced molecular diagnostics in China’s clinical laboratories, strengthening the adoption of precision oncology solutions in the region.
-
In September 2023, Agilent Technologies, Inc., signed a research collaboration agreement with the National Cancer Centre Singapore (NCCS) to advance translational cancer research focused on the genomic landscape of cancers prevalent in Asia. This initiative uses Agilent’s automated NGS platforms and multi-gene sequencing assays at the Cancer Discovery Hub in Singapore to enable precise molecular profiling and support the development of personalized cancer diagnostics.
Asia Pacific Oncology Based Molecular Diagnostics Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 920.8 million
Revenue forecast in 2033
USD 1,560.75 million
Growth rate
CAGR of 6.8% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, type, technology, country
Regional scope
Asia Pacific
Country scope
Japan; China; India; Australia; New Zealand
Key companies profiled
Abbott; Agilent Technologies Inc; bioMerieuz SA; Danaher; F. Hoffmann-La Roche Ltd.; Illumina Inc; Novartis AG; QIAGEN; Siemens Healthineers AG; TBG Diagnostics Limited; Thermo Fisher Scientific Inc; Sysmex
Customization scope
Free report customization (equivalent up to 8 analysts’ working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Asia Pacific Oncology Based Molecular Diagnostics Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the Asia Pacific oncology based molecular diagnostics Market based on product, technology, type, and country:
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Breast Cancer
-
Prostate Cancer
-
Colorectal Cancer
-
Cervical Cancer
-
Liver Cancer
-
Lung Cancer
-
Blood Cancer
-
Kidney Cancer
-
Others
-
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Instruments
-
Reagents
-
Others
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
PCR
-
In situ hybridization
-
INAAT
-
Chips and microarrays
-
Mass spectrometry
-
Sequencing
-
TMA
-
Others
-
-
Country Outlook (Revenue, USD Million, 2021 - 2033)
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
New Zealand
-
-
Frequently Asked Questions About This Report
b. The Asia Pacific oncology based molecular diagnostic market is estimated at USD 822.5 million in 2024 and is expected to reach USD 920.8 million in 2025.
b. The Asia Pacific oncology based molecular diagnostic market is expected to grow at a CAGR of 6.8% from 2025 to 2033 to reach USD 1.56 billion in 2033.
b. Breast cancer dominated the overall market in 2024 in terms of revenue share due to large test volumes and an extensive portfolio of commercialized products.
b. Some of the key players in the Asia Pacific oncology based molecular diagnostic market include Abbott Laboratories, Agilent Technologies, Inc, bioMerieux SA, Danaher Corporation, F. Hoffmann-La Roche Ltd., Illumina, Novartis AG, Qiagen NV, Siemens Healthineers AG, TBG Diagnostics Limited, Thermo Fisher Scientific Inc, Sysmex Corporation.
b. The rising prevalence of various types of cancer, such as breast, prostate, and colon, along with the increase in geriatric population, advancement in technology, rising necessity of point-of-care testing (POCT), and growth in biomarker identification systems, are predicted to be the major drivers for the market.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










