- Home
- »
- Medical Devices
- »
-
Closed-Loop Spinal Cord Stimulation Device Market, 2033GVR Report cover
![Closed-Loop Spinal Cord Stimulation Device Market Size, Share & Trends Report]()
Closed-Loop Spinal Cord Stimulation Device Market (2026 - 2033) Size, Share & Trends Analysis Report By Lead Type (Percutaneous Leads, Paddle/Surgical Leads), By Technology, By Application (Failed Back Syndrome, Degenerative Disk Disease), By End-use, By Country, And Segment Forecasts
- Report ID: GVR-4-68040-905-2
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Closed-Loop Spinal Cord Stimulation Device Market Summary
The closed-loop spinal cord stimulation device market size was estimated at USD 310.0 million in 2025 and is projected to reach USD 889.29 million by 2033, growing at a CAGR of 14.06% from 2026 to 2033. The industry is growing as healthcare systems adopt advanced neuromodulation technologies to manage chronic pain.
Key Market Trends & Insights
- U.S. dominated the closed-loop spinal cord stimulation devices market in 2025.
- By lead type, the percutaneous leads segment led the market with the largest revenue share in 2025.
- By technology, the AI-adaptive closed-loop spinal cord stimulation segments led the market with the largest revenue share in 2025.
- By end-use, the hospitals segment led the market with the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 310.0 Million
- 2033 Projected Market Size: USD 889.29 Million
- CAGR (2026-2033): 14.06%
The shift toward feedback-driven stimulation systems that can automatically adjust therapy in real time is enhancing treatment precision and consistency, making these solutions more effective than traditional approaches. Rising prevalence of conditions such as chronic back pain, neuropathic pain, and post-surgical pain syndromes is driving demand, alongside a broader preference for minimally invasive and non-pharmacological treatment options aimed at reducing opioid dependence. Continuous technological advancements, including intelligent stimulation algorithms, improved implantable device designs, and longer-lasting batteries, are further strengthening clinical outcomes and patient satisfaction.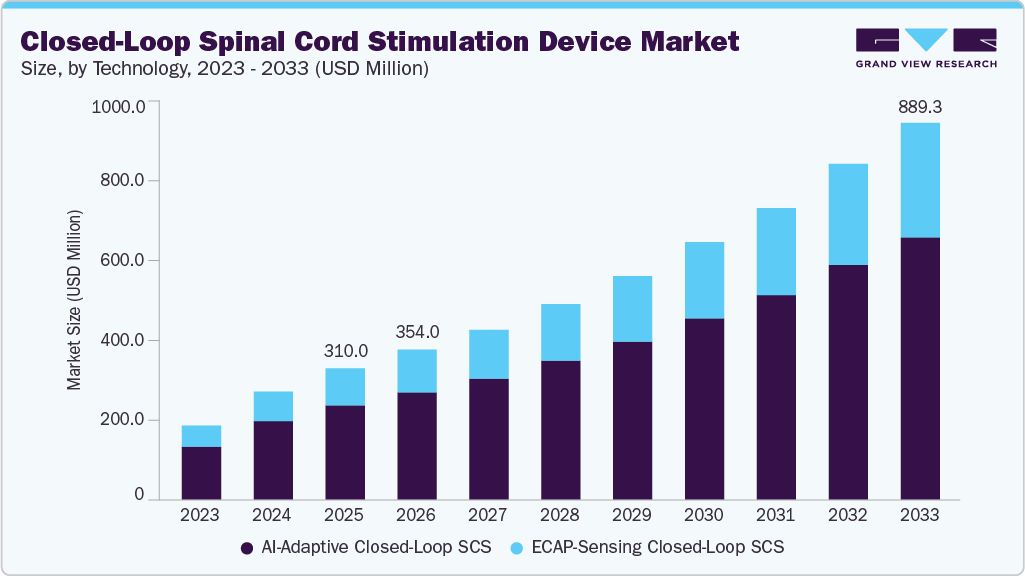
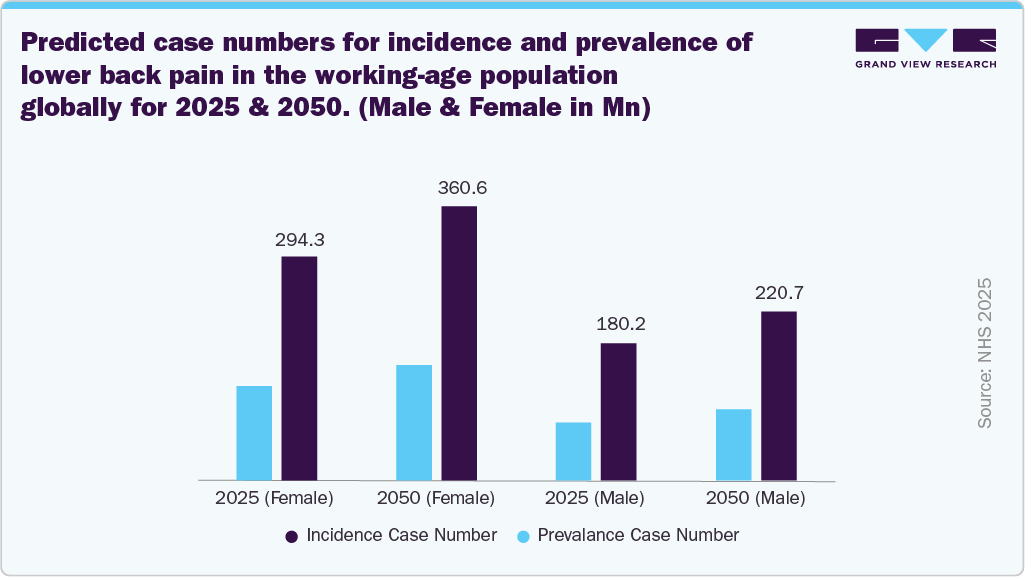
The increasing prevalence of chronic pain conditions, particularly lower back pain, is driving significant growth in the industry, as a larger patient population requires effective, long-term pain management solutions. Lower back pain remains one of the most common and disabling conditions globally, associated with aging, sedentary lifestyles, obesity, and degenerative spinal disorders, leading to persistent discomfort even after conventional treatments or surgical interventions. This creates a strong demand for advanced therapies that can provide consistent relief. Closed-loop systems meet this need by continuously monitoring neural responses and automatically adjusting stimulation in real time, enabling more precise and personalized therapy. As the burden of chronic pain continues to rise and the focus shifts toward non-pharmacological, long-term treatment options that reduce dependence on opioids, the adoption of these advanced neuromodulation technologies is rising, supporting market growth.
“The societal and economic impact of low back pain is enormous. We see loss of productivity from missed days at work and a reliance on medication to alleviate pain,”- Dr. Jaimie Steinmetz, managing research scientist at IHME.
Predicted case numbers and ASRs for incidence, prevalence, and DALYs of lower back pain in the working-age population globally from 2025 to 2050
Year
Incidence Case Number in Mn (95%Crl)
Incidence ASR (95%Crl)
Prevalence Case Number in Mn (95%Crl)
Prevalence ASR (95%Crl)
DALYs Case Number in Mn (95%Crl)
DALYs ASR (95%Crl)
2025
201.82
3655.38
474.54
8576.45
53.73
971.31
2026
204.15
3652.05
480.01
8567.09
54.28
969.15
2027
206.24
3649.28
484.98
8559.74
54.78
967.24
2028
207.94
3647.30
488.99
8554.28
55.17
965.54
2029
209.58
3646.39
492.84
8550.74
55.54
964.06
2030
211.36
3646.57
497.00
8549.24
55.95
962.77
2031
213.23
3647.62
501.40
8550.32
56.37
961.79
2032
215.14
3649.00
505.94
8552.62
56.82
960.96
2033
216.91
3650.70
510.12
8555.52
57.22
960.21
2034
218.51
3652.83
513.87
8558.82
57.58
959.51
2035
220.08
3655.51
517.52
8562.86
57.93
958.87
2036
Upgrade the report license to gain access to the complete analysis.
2037
2038
2039
2040
2041
2042
2043
2044
2045
2046
2047
2048
2049
2050
Source: NIH 2025
Note: ASR, age-standardized rate; DALYs, disability-adjusted life-years; CI, credible interval.
Failed Back Surgery Syndrome (FBSS) is a significant growth driver of the market, as it leads to a high incidence of persistent or recurrent pain following spinal surgery. A considerable proportion of patients undergoing procedures such as laminectomy or spinal fusion continue to experience chronic pain, creating a strong demand for effective long-term pain management solutions. Closed-loop systems are particularly well-suited for FBSS patients as they provide real-time, adaptive stimulation that responds to changes in spinal cord signals, ensuring consistent pain relief despite variations in body position or activity levels. This level of precision addresses one of the key limitations of traditional therapies, where inconsistent stimulation leads to suboptimal outcomes.
-
A 2025 study by the NIH indicates that failed back surgery syndrome remains a widely recognized but relatively underexplored condition, affecting approximately 10% to 40% of patients undergoing spinal surgeries. It is a major contributor to ongoing chronic pain and functional disability, placing a considerable burden on both patients and the broader healthcare system.
Advancements in closed-loop technology are driving the growth of the market by significantly improving the precision, consistency, and effectiveness of pain management therapies. Unlike conventional systems, modern closed-loop devices continuously monitor neural responses and automatically adjust stimulation levels in real time, ensuring that patients receive optimal therapy regardless of changes in posture or activity. This has led to more reliable pain relief, reduced instances of under- or over-stimulation, and higher patient satisfaction. In addition, innovations such as advanced sensing capabilities, adaptive algorithms, miniaturized implantable components, and longer-lasting rechargeable batteries enhancing device performance and convenience. These improvements not only improve clinical outcomes but also increase physicians' confidence in recommending these therapies. Thus, the growing body of positive clinical evidence and technological differentiation are accelerating adoption across healthcare settings, thereby expanding the market.
- In July 2025, Saluda Medical announced the full commercial launch of its EVA sensing technology in the U.S. for use with the Evoke SmartLoop System. The technology received U.S. Food and Drug Administration approval in December 2024 and enabled real-time monitoring of spinal cord activity to support personalized therapy based on neural response signals. As an extension of the Evoke platform, EVA analyzed spinal cord responses with high precision, allowing for more accurate and consistent therapy delivery. The Evoke SmartLoop System further optimized treatment by continuously sensing and adjusting stimulation levels based on each patient’s neural response biomarker, the evoked compound action potential (ECAP), ensuring therapy remained aligned with prescribed settings.
Market Concentration & Characteristics
The industry is characterized by some established medical device companies, largely due to the high technological complexity and stringent regulatory requirements that create significant barriers to entry. Companies in this space compete through proprietary platforms that integrate real-time neural feedback, advanced sensing capabilities, and strong clinical evidence demonstrating improved patient outcomes. The industry is driven by continuous innovation, particularly in adaptive stimulation technologies, device miniaturization, and enhanced precision in therapy delivery. Strategic initiatives such as regulatory approvals, partnerships, and geographic expansion play a key role in strengthening market positioning. In addition, the market exhibits high research and development intensity, long product development cycles, and a strong emphasis on clinical validation to differentiate from conventional systems. With growing demand for personalized pain management solutions, the competitive landscape continues to evolve as companies focus on technological advancements and improved therapy effectiveness.
The market is characterized by continuous and advanced innovation, driven by the integration of real-time sensing technologies, data analytics, and adaptive stimulation mechanisms. These systems are designed to monitor neural responses and automatically adjust stimulation parameters, enabling more precise, personalized therapy than conventional approaches. Innovation efforts focus on improving feedback algorithms, enhancing signal accuracy, and advancing biomarker-based dosing methods, such as evoking compound action potentials. Companies are also working on device miniaturization, longer battery performance, wireless programmability, and more intuitive interfaces to enhance both patient experience and clinical usability. In addition, the incorporation of digital health capabilities, including remote monitoring and data-driven therapy optimization, is further shaping the evolution of treatment, supported by ongoing investments in research and clinical validation.
Regulations play a critical role in the market, as manufacturers must comply with stringent standards to ensure device safety, performance, and clinical effectiveness. In the U.S., these systems are regulated by the U.S. Food and Drug Administration under the medical device framework, requiring Premarket Approval (PMA) due to their classification as high-risk implantable devices, along with extensive clinical evidence supporting real-time sensing and adaptive stimulation features. In Europe, manufacturers must comply with the Medical Device Regulation (MDR), which mandates rigorous clinical evaluation, risk assessment, and CE marking before commercialization. Globally, compliance with quality and safety standards, such as ISO 13485 for quality management and ISO 14971 for risk management, is essential, as is compliance with standards for implantable electronic devices and software validation. In addition, manufacturers are required to follow Good Manufacturing Practices (GMP) and conduct comprehensive clinical trials to demonstrate long-term safety and therapeutic effectiveness.
-
In April 2024, Medtronic announced the U.S. FDA approval of its Inceptiv closed-loop spinal cord stimulation devices, marking a significant advancement in chronic pain management technology. The system features real-time closed-loop sensing that monitors spinal cord signals and automatically adjusts stimulation levels to maintain consistent therapy throughout daily activities. Unlike traditional fixed-output systems, it continuously measures evoked compound action potentials and adapts stimulation multiple times per second to prevent under- or over-stimulation. The device also offers full-body MRI compatibility (including 3T access), a compact implantable design for improved patient comfort, and support for multiple waveform options, including proprietary therapies.
The level of mergers and acquisitions in the market is driven by a strong focus on acquiring advanced closed-loop technologies and innovation capabilities within the neuromodulation space. Market participants are engaging in transactions to integrate real-time neural sensing, adaptive stimulation algorithms, and next-generation implantable device platforms into their portfolios. These activities are primarily focused on strengthening technological expertise, accelerating product development, and enhancing the performance and precision of closed-loop systems. The trend reflects a shift toward innovation-led consolidation, where the objective is to improve clinical outcomes and expand treatment applications rather than pursue large-scale market consolidation. Thus, mergers and acquisitions play an important role in advancing technological capabilities and supporting the growth of the market.
-
In April 2025, Globus Medical announced the completion of its acquisition of Nevro Corp., a global provider of solutions for chronic pain management. This strategic acquisition aimed to strengthen Globus Medical’s position in the neuromodulation and musculoskeletal markets by incorporating Nevro’s high-frequency spinal cord stimulation technology into its portfolio. The transaction expanded Globus Medical’s presence in the chronic pain treatment segment and provided access to a significant market opportunity valued at approximately USD 2.5 billion. By integrating Nevro’s differentiated technologies, including its spinal cord stimulation platform, the company aimed to enhance its product offerings and potentially expand its treatment approaches in the neuromodulation space.
The regional expansion of the market is driven by increasing adoption of advanced neuromodulation technologies across both developed and emerging healthcare systems. The growing demand for highly precise, adaptive pain management solutions is driving wider clinical acceptance of closed-loop systems that automatically adjust stimulation based on real-time neural feedback. This shift is supported by the rising prevalence of chronic pain conditions, increasing preference for minimally invasive and non-drug therapies, and the need to improve long-term patient outcomes.
Healthcare systems worldwide are gradually integrating these technologies as awareness among physicians and patients improves, alongside advancements in clinical training and procedural expertise. Expansion is further supported by continuous improvements in healthcare infrastructure, greater investment in advanced medical devices, and increasing focus on personalized treatment approaches. As innovation in device design and software algorithms continues, global adoption is expected to strengthen, with broader utilization across hospitals and specialty pain management centers.
-
In September 2025, A strategic collaboration was announced between a leading healthcare provider and a global medical technology company to establish a specialized clinical skill and training laboratory focused on advanced minimally invasive and neuromodulation-related procedures.
Lead Type Insights
The percutaneous leads segment dominated the market in 2025, due to its minimally invasive implantation approach and widespread clinical adoption. These leads are inserted through a needle into the epidural space, eliminating the need for open surgery and enabling procedures to be performed in outpatient or ambulatory settings. This results in lower procedural costs, reduced hospital stays, and faster patient recovery, making them highly preferred by both physicians and patients. In addition, percutaneous leads are commonly used during trial stimulation phases before permanent implantation, further contributing to their high procedural volume and market share.
The paddle or surgical leads segment is experiencing significant growth, driven by its superior stability and enhanced long-term therapeutic outcomes. These leads are implanted via a surgical procedure, allowing for precise placement and reduced risk of lead migration compared to percutaneous leads. Their ability to deliver more targeted and consistent stimulation makes them preferred in complex or refractory chronic pain cases. Advancements in surgical techniques and the growing emphasis on durable pain relief solutions are further driving the adoption of paddle leads, positioning them as a significant growth segment in the market.
Technology Insights
AI-adaptive closed-loop spinal cord stimulation dominated the market in 2025 due to its ability to deliver highly personalized, dynamic pain management. These systems leverage advanced algorithms and data-driven insights to continuously learn from patient-specific neural responses and automatically adjust stimulation parameters in real time. This results in improved therapy consistency, reduced need for manual reprogramming, and enhanced patient outcomes. The growing focus on precision medicine, along with increasing integration of digital health platforms and smart implantable devices, has accelerated the adoption of AI-driven closed-loop SCS devices, positioning them as the leading technology in the market.
ECAP-sensing closed-loop spinal cord stimulation is the fastest-growing technology segment, driven by its strong clinical validation and objective measurement of neural activation. By directly monitoring evoked compound action potentials (ECAPs), these systems can accurately assess the spinal cord’s response to stimulation and make immediate adjustments to maintain optimal therapeutic levels. This objective, physiology-based approach reduces variability in treatment outcomes and enhances reliability compared to traditional systems. Increasing clinical adoption, expanding regulatory approvals, and growing evidence supporting its efficacy in chronic pain management are key factors driving the rapid market growth of ECAP-sensing closed-loop SCS.
Application Insights
The Complex Regional Pain Syndrome (CRPS) segment dominated the market in 2025, due to the severe, chronic, and treatment-resistant nature of the condition. CRPS patients experience persistent neuropathic pain that responds poorly to conventional therapies, making spinal cord stimulation a preferred treatment option. Closed-loop systems that deliver real-time, adaptive stimulation based on neural feedback have shown enhanced efficacy in managing fluctuating pain levels associated with CRPS. The high clinical reliance on neuromodulation for CRPS, along with strong physician preference and established treatment guidelines, has contributed to its market share.
Degenerative Disc Disease (DDD) is the fastest-growing application segment, driven by the rising global burden of age-related spinal disorders and chronic low back pain. As the aging population increases, the prevalence of DDD continues to rise, creating a larger patient pool requiring long-term pain management solutions. Closed-loop SCS systems are being adopted for DDD due to their ability to provide consistent and personalized pain relief, particularly in patients who have not responded to conservative treatments or surgery. Advancements in minimally invasive implantation techniques and growing awareness among patients and clinicians are further accelerating the adoption of closed-loop SCS in this segment.
End-use Insights
Hospitals dominated the market in 2025, due to their access to advanced surgical infrastructure, multidisciplinary expertise, and comprehensive patient care capabilities. Hospitals serve as primary centers for complex implantation procedures, particularly for patients requiring intensive monitoring and management of comorbid conditions. The availability of skilled neurosurgeons, pain specialists, and advanced imaging systems supports higher procedural volumes and better clinical outcomes. In addition, hospitals are key sites for clinical research and early adoption of advanced closed-loop technologies, furthering their market share.
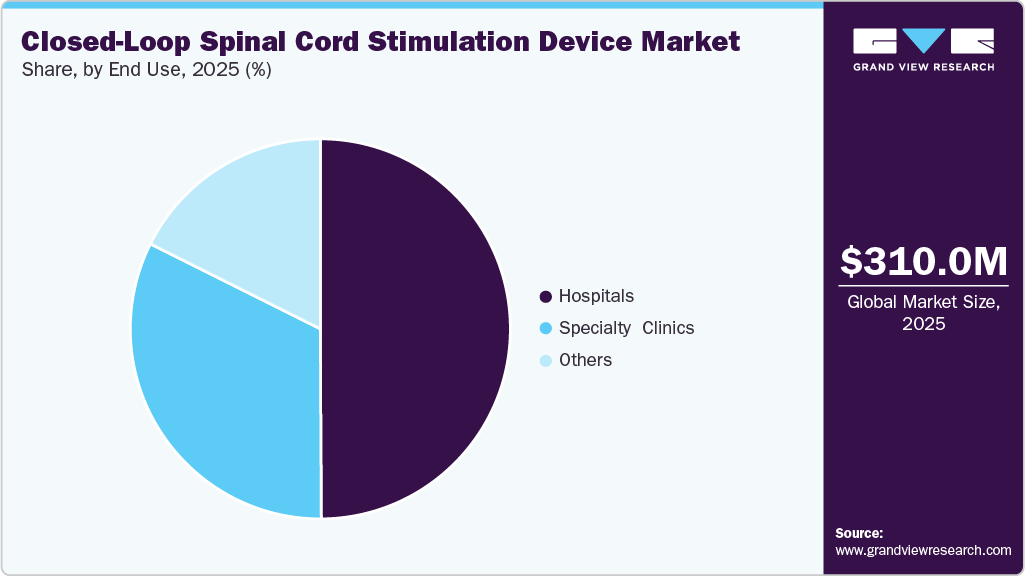
The specialty clinics segment is the fastest-growing segment, driven by the increasing shift toward outpatient care and targeted pain management services. These clinics provide focused expertise, enabling efficient patient selection, streamlined procedures, and personalized therapy adjustments. Their ability to offer minimally invasive treatments, reduced procedure costs, and faster recovery times has made them attractive to patients. Moreover, continuous follow-up care and device optimization services provided by specialty clinics enhance treatment effectiveness, contributing to their rapid growth in the evolving neuromodulation landscape.
Country Insights
U.S. Closed-Loop Spinal Cord Stimulation Device Market Trends
The closed-loop spinal cord stimulation device market in the U.S. is driven by the rising number of low back pain cases in the working-age population, as this condition represents one of the most common causes of chronic disability and work-related impairment. A large proportion of working adults experience persistent or recurrent pain that does not respond adequately to conventional treatments such as medication, physiotherapy, or injections, creating a growing need for advanced long-term pain management solutions. Closed-loop systems are gaining importance in this setting because they provide real-time adaptive stimulation that adjusts automatically to patient movement and neural feedback, offering more stable and personalized pain relief. This improved precision helps maintain functional ability, reduce opioid dependence, and enhance quality of life for individuals who need to remain active in the workforce. Thus, the burden of low back pain in the working-age population is expanding the eligible patient pool and accelerating the adoption of advanced closed-loop neuromodulation therapies in the U.S.
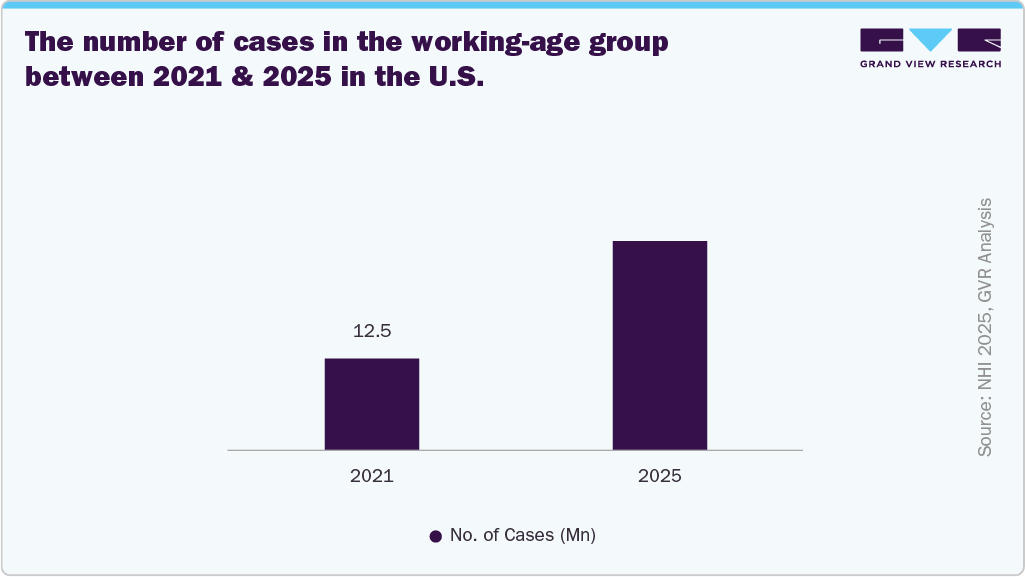
The rising number of clinical trials in the U.S. is significantly driving the growth of the closed-loop spinal cord stimulation devices market by strengthening clinical evidence, regulatory confidence, and physician acceptance of advanced neuromodulation technologies. Studies are evaluating real-time adaptive stimulation systems that adjust based on neural feedback, demonstrating improved pain relief, greater therapy consistency, and reduced overstimulation compared to conventional approaches. These trials are also expanding research across multiple chronic pain indications, broadening the eligible patient population and supporting wider clinical use. As more positive outcomes are generated, clinical trials help accelerate regulatory approvals and increase trust among healthcare providers, encouraging earlier adoption in routine practice. In addition, ongoing research into next-generation algorithms and personalized stimulation strategies further enhances device performance and treatment precision.
Clinical Trials of Closed-Loop Spinal Cord Stimulation Systems
NCT Number
Study Title
Completion Date
NCT05177354
Medtronic Closed-Loop Spinal Cord Stimulation System
7/28/2025
NCT06421350
Closed Loop Spinal Cord Stimulation for Complex Regional Pain Syndrome
2026-06
NCT04969042
Closed-loop Functional Spinal Cord Stimulation in Patients with Spinal Cord Injury
8/1/2027
NCT06616649
Closed Loop Spinal Cord Stimulation for Chemotherapy-Induced Peripheral Neuropathy, a Pilot Study.
7/1/2027
NCT07413731
Neurophysiological Effects of Medication Tapering During Treatment With Spinal Cord Stimulation
4/3/2027
NCT04662905
Novel Treatment Delivery of ECAP-controlled Closed-loop SCS for Chronic Pain
1/1/2027
NCT04319887
ECAP-controlled, Closed-loop Stimulation to Treat Chronic Trunk and/or Limb Pain (ECAP)
12/28/2023
NCT06377969
Prospective Evaluation of Effectiveness and Safety of Closed-Loop Spinal Cord Stimulation in Treating Patients with Chronic Pelvic Pain
5/31/2028
NCT06499220
Electrically Evoked Compound Action Potentials Human Observation Medtronic System Study (ECHO-MDT)
10/16/2023
NCT05740527
Ambulatory Closed-loop Stimulation for Bladder Control
10/1/2024
NCT07304401
Evaluation of the Effectiveness of Closed-loop Spinal Cord Stimulation (CL-SCS) Therapy for Treatment of Chemotherapy-Induced Peripheral Neuropathy
12/1/2028
NCT02924129
Safety and Efficacy Study of the Evoke™ SCS System with Feedback vs. Conventional Stimulation
9/9/2022
NCT06101199
Combined Therapeutic Air Mixture and Electrical Stimulation to Improve Breathing and Hand Function in Spinal Cord Injury
6/30/2025
NCT05940636
A Novel Combined Neuromodulation Therapy to Enhance Balance and Neuroplasticity
1/30/2026
Source: Clinical Trials. Gov
UK Closed-Loop Spinal Cord Stimulation Device Market Trends
UK closed-loop spinal cord stimulation devices market is growing due to increased clinical focus on advanced neuromodulation therapies for chronic pain conditions, particularly complex regional pain syndrome, which is attracting greater research attention as a difficult-to-treat neuropathic pain disorder. Rising investment from academic institutions and healthcare organizations in CRPS-focused studies is supporting the evaluation of real-time adaptive stimulation technologies, as clinical trials assess how closed-loop systems can improve pain control and functional recovery at both early and chronic stages of CRPS. These research efforts are strengthening clinical evidence and encouraging wider adoption of advanced neuromodulation approaches in specialist pain centers. In addition, growing collaboration between research bodies and healthcare providers is improving physician awareness and technical expertise in implantable neurostimulation therapies. The combination of rising R&D investment in CRPS management, supportive clinical infrastructure, and expanding use of minimally invasive pain therapies is driving the adoption of closed-loop spinal cord stimulation technologies across the U.K. market.
According to the UK Parliament in 2025, the National Institute for Health and Care Research has allocated approximately USD 242,000 over the last five financial years for research on Complex Regional Pain Syndrome (CRPS). This research has focused on strengthening evidence-based approaches to CRPS management to improve clinical outcomes across the entire care pathway.
Germany Closed-Loop Spinal Cord Stimulation Device Market Trends
Germany closed-loop spinal cord stimulation devices market is significantly driven by the rising prevalence of low back pain. Low back pain remains one of the most common chronic pain conditions and a leading cause of disability, particularly among the aging population and individuals with degenerative spinal disorders. Thus, a growing number of patients are not achieving adequate relief from conventional therapies such as pharmacological treatment and physiotherapy, increasing the demand for advanced neuromodulation solutions. This has accelerated the adoption of spinal cord stimulation technologies, especially closed-loop systems that provide real-time, adaptive pain control and improved clinical outcomes. According to data published by the National Library of Medicine in 2025, approximately 3,248,641 cases of lower back pain were reported among the working-age population in 2021.
Japan Closed-Loop Spinal Cord Stimulation Device Market Trends
The closed-loop spinal cord stimulation devices market in Japan is driven by a rapidly aging population, as older adults are more prone to chronic pain conditions such as degenerative spine disorders, neuropathic pain, and failed back surgery syndrome. With a large proportion of the population aged 65 years and above, there is an increasing clinical need for long-term, effective, and minimally invasive pain management solutions. Closed-loop spinal cord stimulation devices, which provide adaptive and real-time modulation of neural signals, are gaining preference over conventional therapies due to improved pain relief and reduced side effects. This demographic shift is therefore expanding the patient pool eligible for neuromodulation therapies, supporting sustained growth in Japan's closed-loop spinal cord stimulation devices market. In 2025, according to the World Economic Forum, Japan's Ministry of Internal Affairs and Communications reported that, as of 2024, the country had a record 36.25 million people aged 65 and over, accounting for 29.3% of the total population. This share is projected to rise to 34.8% in 2040 and 36.3% in 2045. Japan's proportion of elderly residents is already far higher than second-ranked Martinique (25.3%), making it the world's most aged society.
Key Closed-Loop Spinal Cord Stimulation Device Company Insights
The closed-loop spinal cord stimulation devices market is highly competitive, with key players holding significant market share. Major companies are pursuing a range of organic and inorganic strategies, including new product development, collaborations, acquisitions, mergers, and regional expansion, to address unmet customer needs.
Key Closed-Loop Spinal Cord Stimulation Device Companies:
The following key companies have been profiled for this study on the closed-loop spinal cord stimulation devices market.
- Medtronic plc
- Saluda Medical Pty Ltd.
Recent Developments
-
January 2026, Saluda Medical announced that its next-generation EVA Sensing Technology received CE certification for commercialization in Europe, with formal recognition of the approval in Australia. This regulatory milestone follows prior FDA approval granted in December 2024. EVA Sensing Technology builds on the company’s Evoke System, a closed-loop spinal cord stimulation platform designed to treat chronic neurological pain conditions. The system uses real-time sensing of evoked compound action potentials (ECAPs) to continuously measure spinal cord responses and automatically adjust stimulation parameters. This enables highly personalized neuromodulation therapy tailored to each patient’s neural activity.
-
April 2024, Medtronic announced that the U.S. Food and Drug Administration (FDA) approved its Inceptiv closed-loop rechargeable spinal cord stimulation devices for the treatment of chronic pain. The Inceptiv system represents a major advancement in spinal cord stimulation technology as it is the first Medtronic device to use closed-loop sensing. It continuously detects biological signals from the spinal cord and automatically adjusts stimulation in real time to maintain consistent, physician-prescribed therapy during patients’ daily activities.
Closed-Loop Spinal Cord Stimulation Device Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 354.02 million
Revenue forecast in 2033
USD 889.29 million
Growth rate
CAGR of 14.06% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Lead type, technology, application, end-use, county
Country scope
U.S.; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; Australia
Key companies profiled
Saluda Medical Pty Ltd.; Medtronic plc
Customization scope
Free report customization (equivalent to up to 8 analysts working days) with purchase. Addition or alteration to country, country & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Closed-Loop Spinal Cord Stimulation Device Market Report Segmentation
This report forecasts country-level revenue growth and analyzes the latest industry trends and opportunities across sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the closed-loop spinal cord stimulation devices market report based on lead type, technology, application, end-use, and country:
-
Lead Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Percutaneous Leads
-
Paddle/Surgical Leads
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
ECAP-Sensing Closed-Loop SCS
-
AI-Adaptive Closed-Loop SCS
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Failed Back Syndrome
-
Complex Regional Pain Syndrome
-
Degenerative Disk Disease
-
Unsuccessful Disk Surgery
-
Others
-
-
End-use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Specialty Clinics
-
Others
-
-
Country Outlook (Revenue, USD Million, 2021 - 2033)
-
U.S.
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
Japan
-
Australia
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










