- Home
- »
- Plastics, Polymers & Resins
- »
-
Medical Plastics Market Size & Share, Industry Report, 2033GVR Report cover
![Medical Plastics Market Size, Share & Trends Report]()
Medical Plastics Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Polyethylene, Polypropylene, Polycarbonate, Liquid Crystal Polymer, Polyphenylsulfone, Polyethersulfone), By Application, By Region, And Segment Forecasts
- Report ID: GVR-1-68038-823-7
- Number of Report Pages: 170
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Bulk Chemicals
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Medical Plastics Market Summary
The global medical plastics market size was valued at USD 61.35 billion in 2025 and is projected to reach USD 99.25 billion by 2033, growing at a CAGR of 6.0% from 2026 to 2033. The growing geriatric population in North America, Europe, and Asian countries like Japan, and the increasing demand for home healthcare are the major factors driving the growth of Medical Plastics in the forecast period.
Key Market Trends & Insights
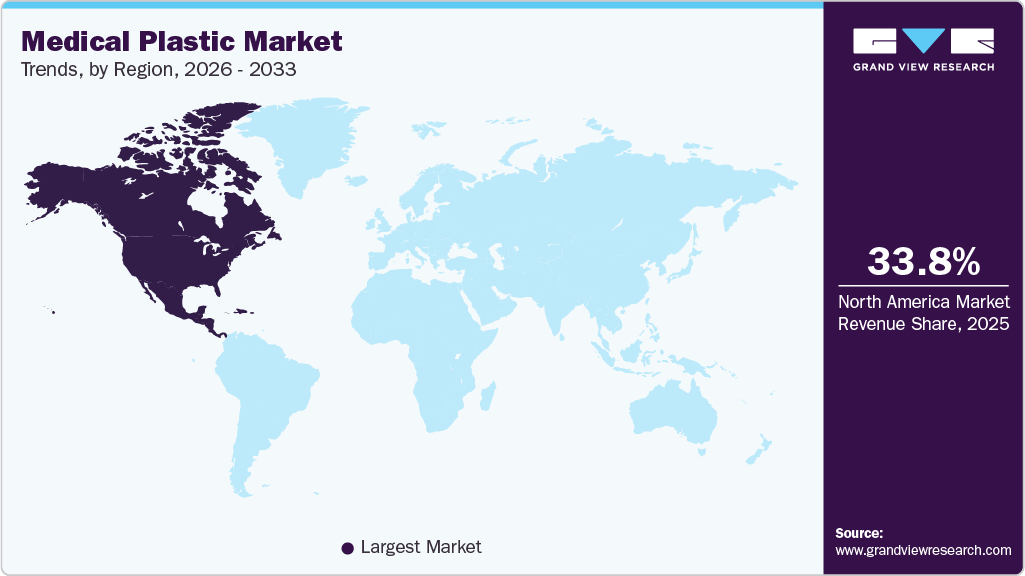
- North America medical plastics market led with the largest revenue share of 33.8% in 2025.
- The medical plastics market in China is expected to grow at a substantial CAGR of 10.8% from 2026 to 2033.
- By product, the PPSU segment led the market in 2025.
- By application, the medical component segment led the market in 2025.
Market Size & Forecast
- 2025 Market Size: USD 61.35 Billion
- 2033 Projected Market Size: USD 99.25 Billion
- CAGR (2026-2033): 6.0%
- North America: Largest market in 2025
The market for medical plastics is a fundamentally robust, innovation-led segment within the broader healthcare materials domain, bolstered by rising healthcare spending, broader access to medical services, and consistent growth in the use of medical devices. Demand is driven by demographic factors such as an aging population, the rising incidence of chronic illnesses, and the increasing number of procedures performed in hospitals and outpatient settings. The regulatory focus on patient safety, infection prevention, and device efficacy has further promoted the use of high-purity medical polymers in both disposable and reusable applications.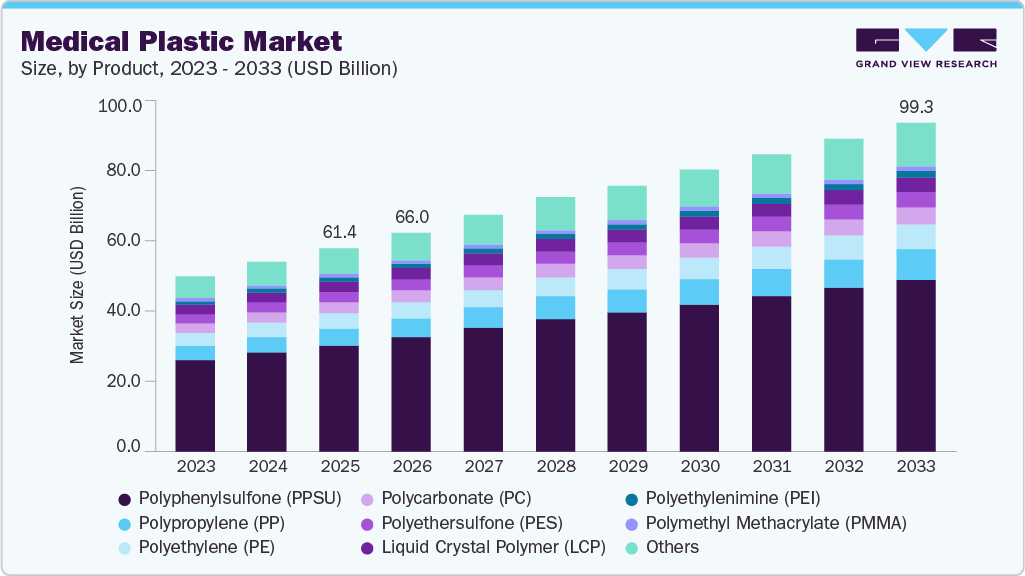
The market is primarily led by commodity and engineering plastics designed for medical applications, such as polyvinyl chloride (PVC), polypropylene (PP), polyethylene (PE), polystyrene (PS), polycarbonate (PC), and specialized polymers including polyamides, ABS, PEEK, and silicone elastomers. Engineering and high-performance plastics are increasingly being chosen for advanced medical devices, implants, and minimally invasive instruments, as original equipment manufacturers (OEMs) focus on materials that offer enhanced mechanical performance, confirmed biocompatibility, and long-lasting durability under frequent, intense sterilization processes.
Drivers, Opportunities & Restraints
The expansion of the industry is mainly fueled by increasing healthcare spending, higher numbers of procedures, and the greater adoption of disposable medical devices aimed at enhancing infection control and ensuring patient safety. Factors related to demographics, including an aging population and a rising incidence of chronic diseases, are maintaining the demand for medical devices, diagnostic consumables, and packaging solutions.
Expansion of the medical polymer market is constrained by strict regulatory standards on material authorization, traceability, and biocompatibility, which lengthen development timelines and increase compliance costs for manufacturers. Increased focus on plastics waste and sustainability in the healthcare sector, especially regarding single-use items, has also posed a barrier, leading hospitals and regulatory bodies to consider alternatives or implement disposal and recycling requirements. Furthermore, fluctuations in polymer feedstock prices and supply chain disruptions can affect profit margins and procurement consistency.
The market offers opportunities to apply engineering and high-performance polymers in innovative medical fields, including minimally invasive devices, implants, and drug-delivery systems. Progress in bio-based, recyclable, and low-carbon medical-grade plastics is creating new growth opportunities as healthcare systems strive to meet sustainability objectives while upholding safety and performance standards.
Market Concentration & Characteristics
The medical plastics sector is marked by significant regulatory scrutiny, a strong focus on maintaining quality and consistency, and enduring supply partnerships between material providers and medical device original equipment manufacturers (OEMs). Demand in the medical device plastics market sector is primarily non-cyclical and defensive, driven by critical healthcare needs and consistent procedural volumes, which help stabilize it amid broader economic fluctuations.
The industry exhibits a moderate to high level of innovation, driven by ongoing advancements in biocompatibility, resistance to sterilization, miniaturization, and sustainability. Suppliers of polymers are increasingly focusing their investments on specialty grades and formulations tailored for specific applications, while original equipment manufacturers (OEMs) in the medical device sector are prioritizing design optimization and integration of functions. Mergers and acquisitions are occurring in a selective, strategic manner to broaden medical-grade product lines, ensure compliance with regulatory manufacturing standards, and enhance geographic reach, especially in rapidly growing healthcare markets.
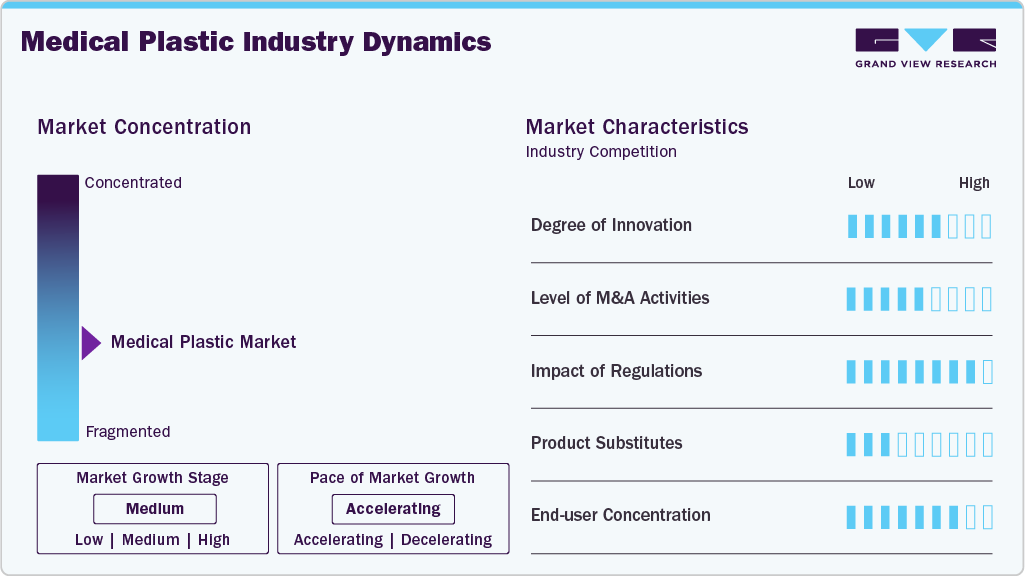
Regulatory frameworks significantly influence market dynamics, as strict requirements for approval, testing, and documentation oversee medical-grade plastics and end-use devices. Although these regulations increase compliance costs and delay time-to-market, they also create high barriers to entry and safeguard existing suppliers. In numerous applications, the potential for product substitution is limited due to concerns regarding performance, safety, and cost; nonetheless, metals, glass, and new bio-based materials present a selective risk of substitution in implantable and environmentally sensitive applications.
The concentration of end users in the industry is moderate, with demand spread across a wide range of medical device manufacturers, packaging converters, and healthcare product suppliers. However, purchasing power is becoming increasingly concentrated among large, multinational medical device original equipment manufacturers (OEMs) and healthcare systems, which are exerting pressure on pricing, quality assurance, and supply reliability. Smaller OEMs and contract manufacturers meet additional demand but usually operate within the qualification standards established by leading end users.
Product Insights
Polyphenylsulfone (PPSU) dominated the global market in the product segment, accounting for 52.05% of overall revenue in 2025. Polyphenylsulfone exhibits good heat and chemical resistance, making it ideal for replacing metals in medical applications. These plastics possess high flexural strength, impact resistance, and durability, and are ideal for producing single- and multi-use surgical instruments. Its high-temperature resistance makes it suitable for medical devices that require repeated steam sterilization. These plastics are increasingly being used in surgical robots and biopharmaceutical processing.
Polypropylene (PP) is used for manufacturing protective packaging and medical equipment due to its high toughness and durability. Furthermore, the strong chemical bonds make it suitable for manufacturing medical components, including disposable syringes, connectors, finger-joint prostheses, non-absorbable sutures, reusable plastics containers, pharmacy prescription bottles, clear bags, beakers, test tubes, and others.
Application Insights
The medical components segment dominated the global market, accounting for the largest revenue share of 41.72% in 2025. The growing application scope of PP in the manufacturing of diagnostic devices, trays, pans, containers, syringes, implant trials, and medical cover sheets is expected to drive the demand. Polypropylene is expected to witness substantial growth in medical components applications owing to its low weight, high bacterial and chemical resistance, and low cost. It has a wide scope of application in medical disposable manufacturing. Furthermore, it can be easily molded to precise dimensions as needed and thus has a high demand in the production of syringes.
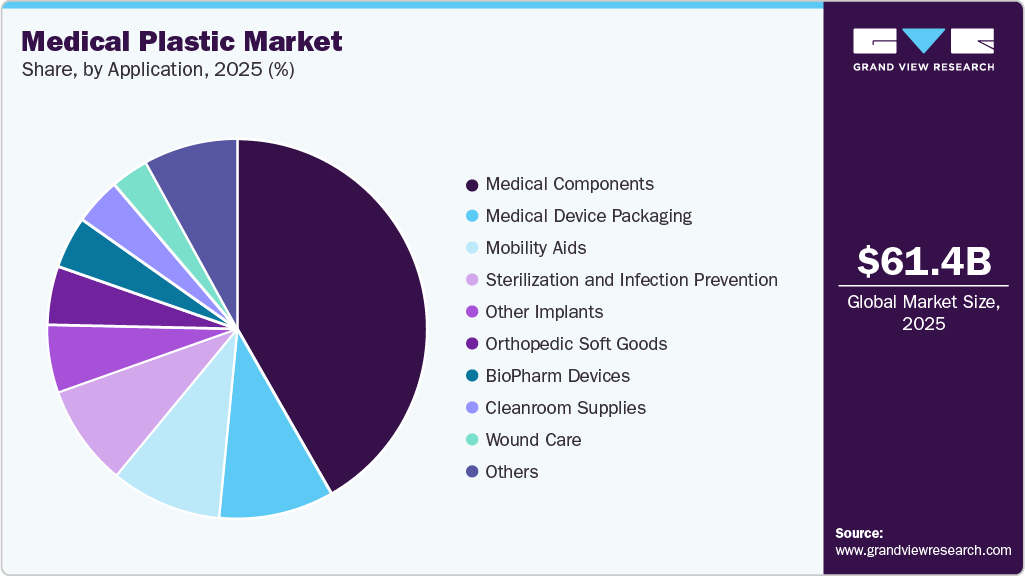
Mobility aids are the second-largest segment in which medical plastics are used. The use of plastics in the development of mobility aids is expected to grow significantly as the global geriatric population increases. Technological advancements and rising healthcare expenditure are expected to further drive the market. This is expected to increase the global consumption of plastics such as acrylonitrile butadiene styrene (ABS), PEEK, and polycarbonate (PC). The use of plastics in mobility aids such as wheelchairs, drinking aids, and bath seats is expected not only to contribute to the surge in product demand but also to the use of plastics as substitutes for metals in various medical applications, owing to their lightweight. In addition, the superior properties of medical plastics, such as strength and fatigue resistance, are expected to drive market growth.
Regional Insights
North America medical plastics market dominated the regional segment, accounting for 33.8% of overall revenue in 2025. The demand for medical plastics in North America is driven by the growing elderly population in the US and the increasing demand for home healthcare services. Rising product demand in pharmaceutical packaging applications and the rapid growth of the pharmaceutical industries in Mexico and Canada are expected to further drive the regional market.
U.S. Medical Plastics Market Trends
The medical plastics market in the U.S. is the largest and most developed in the world, bolstered by substantial healthcare expenditures, a robust domestic medical device production base, and ongoing advancements in cutting-edge medical technologies. The demand is fueled by high volumes of procedures, extensive use of single-use devices, and rigorous regulatory requirements that promote the use of high-quality, medical-grade polymers.
Europe Medical Plastics Market Trends
The medical plastics market in Europe is marked by stringent regulatory controls, a robust ecosystem for manufacturing medical devices, and consistent demand driven by advanced healthcare systems. Expansion is fueled by an aging population, rising healthcare utilization, and a growing focus on high-performance medical plastics, sustainable medical-grade polymers that comply with EU regulations and environmental standards.
Asia Pacific Medical Plastics Market Trends
The medical plastics market in the Asia Pacific is experiencing growth that exceeds the average, driven by the development of healthcare infrastructure, rising healthcare spending, and increased local production of medical devices. An increase in patient numbers, enhancements to regulatory systems, and the trend of global original equipment manufacturers moving to regional production centers are driving up demand for both commodity and engineering-grade medical plastics.
China is the largest and fastest-growing market in the Asia Pacific, bolstered by the rapid expansion of local medical device manufacturing and substantial government funding for healthcare infrastructure. Efforts towards localization and a rising adoption of advanced medical devices are fueling ongoing demand for medical-grade plastics.
Key Medical Plastics Company Insights
Key companies are adopting a range of organic and inorganic growth strategies, such as new product development, mergers & acquisitions, and joint ventures, to maintain and expand their market share.
Key Medical Plastics Companies:
The following key companies have been profiled for this study on the medical plastics market.
- Röchling SE & Co. KG
- Nolato AB
- Saint-Gobain Performance Plastics
- SABIC
- Orthoplastics Ltd
- Eastman Chemical Company
- Celanese Corporation
- Dow
- Tekni-Plex
- Solvay S.A.
- HMC Polymers
- ARAN BIOMEDICAL TEORANTA
- Trelleborg AB
- Avantor Inc
- Trinseo
- Evonik Industries AG
Recent Developments
-
In November 2025, SK Capital Partners completed the purchase of LISI Group's medical division and created a new independent entity called Precera Medical, enhancing its position in specialty medical materials and components for healthcare uses. This strategic acquisition is designed to utilize LISI's knowledge and SK Capital's investment strategy to promote growth in Medical Plastics, device components, and precision-manufactured products for hospitals and original equipment manufacturers (OEMs).
-
In July 2025, Biomerics, a contract manufacturer with vertical integration, announced the purchase of Dependable Plastics and the establishment of its new Interventional Medical Plastics Division (IMP). This acquisition enhances Biomerics' expertise in sophisticated plastics production, including thermoforming, liquid-cast resin molding, and additive manufacturing, for the robotic and interventional device sectors.
Medical Plastics Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 66.03 billion
Revenue forecast in 2033
USD 99.25 billion
Growth rate
CAGR of 6.0% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Volume in kilotons; revenue in USD million/billion, and CAGR (%) from 2026 to 2033
Report coverage
Volume forecast, revenue forecast, company profiles, competitive landscape, growth factors, and trends
Segments covered
Product, application, region
Region scope
North America; Europe; Asia Pacific; Central & South America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; Germany; UK; Italy; France; China; India; Japan; Brazil; Saudi Arabia
Key companies profiled
Röchling SE & Co. KG; Nolato AB; Saint-Gobain Performance Plastics; SABIC; Orthoplastics Ltd; Eastman Chemical Company; Celanese Corporation; Dow; Tekni-Plex; Solvay S.A.; HMC Polymers; ARAN BIOMEDICAL TEORANTA; Trelleborg AB; Avantor Inc; Trinseo; Evonik Industries AG
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional, and segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Medical Plastics Market Report Segmentation
This report forecasts volume & revenue growth at the global, regional, and country levels and provides an analysis of the latest industry trends in each sub-segment from 2026 to 2033. For this study, Grand View Research has segmented the global medical plastics market report based on product, application, and region:

-
Product Outlook (Volume, Kilotons; Revenue, USD Million, 2021 - 2033)
-
Polyethylene (PE)
-
Polypropylene (PP)
-
Polycarbonate (PC)
-
Liquid Crystal Polymer (LCP)
-
Polyphenylsulfone (PPSU)
-
Polyethersulfone (PES)
-
Polyethylenimine (PEI)
-
Polymethyl Methacrylate (PMMA)
-
Others
-
-
Application Outlook (Volume, Kilotons; Revenue, USD Million, 2021 - 2033)
-
Medical Device Packaging
-
Medical Components
-
Orthopedic Implant Packaging
-
Orthopedic Soft Goods
-
Wound Care
-
Cleanroom Supplies
-
BioPharm Devices
-
Mobility Aids
-
Sterilization and Infection Prevention
-
Tooth Implants
-
Denture Base Material
-
Other Implants
-
Others
-
-
Region Outlook (Volume, Kilotons; Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
Italy
-
France
-
-
Asia Pacific
-
China
-
India
-
Japan
-
-
Central & South America
-
Brazil
-
-
Middle East & Africa
-
Saudi Arabia
-
-
Frequently Asked Questions About This Report
Some key players operating in the medical plastics market include Solvay S.A. Celanese Corporation, Eastman Chemical Company, Dow, SABIC and Evonik Industries AG
Key factors that are driving the medical plastics market growth include rising healthcare expenditure, ascending demand for better healthcare facilities, improving medical infrastructure, and increasing demand for surgical instruments and tools such as gloves, clamps, forceps, syringes, surgical trays, and others.
North American region led the global medical plastics market in 2023 accounting for the largest revenue share of more than 33.0%
The global medical plastics market size was estimated at USD 61.35 billion in 2025 and is expected to reach USD 66.03 billion in 2026.
The global medical plastics market is expected to grow at a compound annual growth rate of 6.0% from 2026 to 2033 to reach USD 99.25 billion by 2030.
The medical components segment dominated the medical plastics market with a share of 41.72% in 2025. This is attributed to the rising product demand in product demand in various applications such as diagnostic devices, trays, pans, containers, syringes, implant trials, and medical cover sheets.
About the Author(s)
Plastics, Polymers & Resins Research Team
Bulk Chemicals · Plastics, Polymers & ResinsThis report was authored by the plastics, polymers & resins Research Team at Grand View Research - comprising two research analysts, one senior research analyst, and one industry expert - with specialized expertise in the plastics, polymers & resins segment of the bulk chemicals industry. All findings are based on proprietary bulk chemicals databases, executive interviews, and regulatory analysis, subject to internal peer review prior to publication.
Last Updated:
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









