- Home
- »
- Healthcare IT
- »
-
Randomization & Trial Supply Management Market Report 2033GVR Report cover
![Randomization And Trial Supply Management Market Size, Share & Trends Report]()
Randomization And Trial Supply Management Market (2026 - 2033) Size, Share & Trends Analysis Report By Delivery Mode (Cloud & Web-based, On-premise), By End-use (Hospitals, CROs, Academic Institutes, Pharma & Biotech Organizations), By Region, And Segment Forecasts
- Report ID: GVR-4-68040-901-6
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Randomization & Trial Supply Management Market Summary
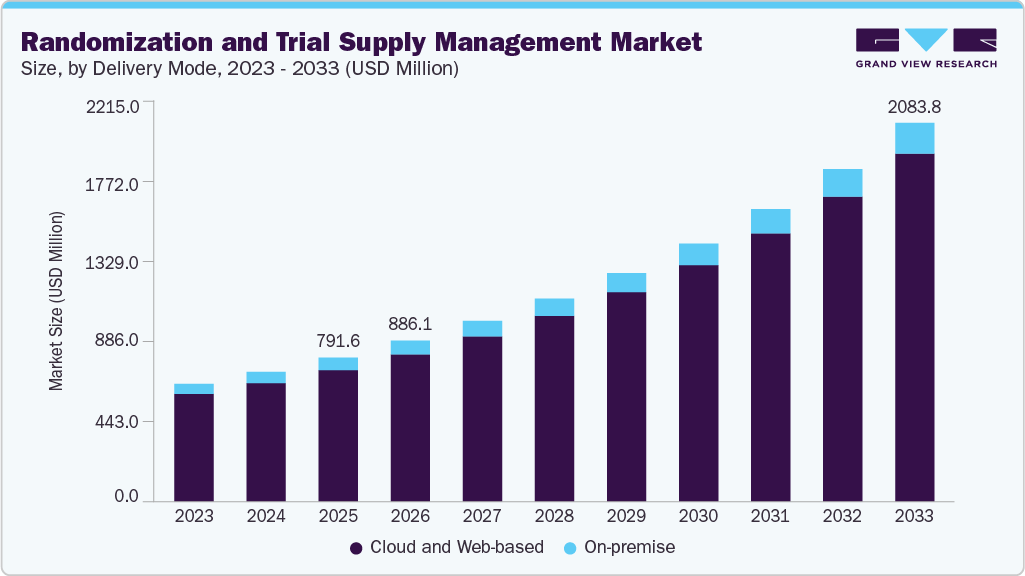
The global randomization and trial supply management (RTSM) market size was estimated at USD 791.6 million in 2025 and is projected to reach USD 2083.8 million by 2033, growing at a CAGR of 13.0% from 2026 to 2033. The growth is driven by the increasing complexity of clinical trials, particularly in areas such as oncology, rare diseases, and advanced therapies.
Key Market Trends & Insights
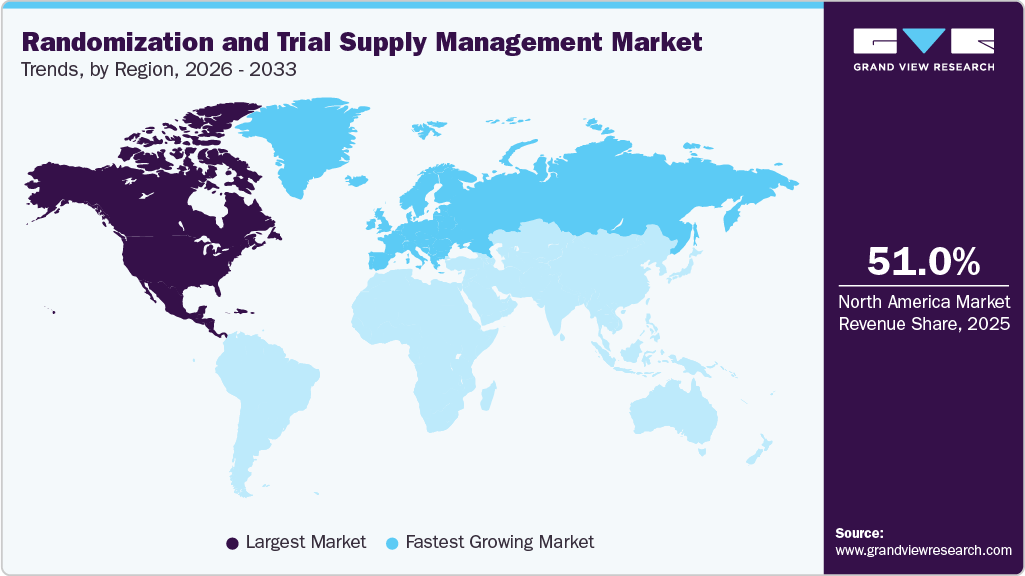
- North America dominated the market with a revenue share of 51.0% in 2025.
- The Europe market is anticipated to grow at the fastest CAGR of 14.7% from 2026 to 2033.
- By delivery mode, the cloud and web-based segment led the market with the largest revenue share of 91.4% in 2025.
- By end use, the CROs held the largest market share of 37.3% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 791.6 Million
- 2033 Projected Market Size: USD 2,083.8 Million
- CAGR (2026-2033): 13.0%
- North America: Largest market in 2025
- Europe: Fastest growing market
Modern clinical studies involve adaptive designs, multiple treatment arms, and global multi-site execution, which significantly increases the need for efficient patient randomization and drug supply coordination. randomization and trial supply management (RTSM) platforms address these challenges by enabling real-time management and automation. For instance, in 2025, innovations, such as IQVIA’s advanced IRT solutions for early-phase trials, offer flexible configurations and real-time cohort adjustments to handle complex study protocols.Moreover, the rising adoption of decentralized and hybrid clinical trials (DCTs) is accelerating the demand for RTSM solutions. As clinical trials are shifting toward remote participation, direct-to-patient drug delivery, and virtual monitoring, there is a growing need for systems that can manage distributed inventories and ensure seamless coordination across geographies. RTSM platforms play a critical role in enabling these capabilities, supporting patient-centric trial models and improving accessibility, which in turn drives their widespread adoption.
The growing emphasis on regulatory compliance and data integrity is further reinforced by evolving global guidelines and increasing inspection scrutiny. For instance, updated ICH E6(R3) guidelines now position RTSM/IRT systems as quality-critical infrastructure, requiring robust audit trails, real-time traceability, and risk-based oversight across the clinical trial lifecycle. Regulators increasingly expect systems to clearly document “who, when, and why,” ensuring full transparency and inspection readiness. In addition, in July 2025, IQVIA highlighted a rise in regulatory inspections involving RTSM systems since 2021, continuing into 2025, with a greater focus on data integrity, audit trails, and system validation. This trend is pushing sponsors and CROs to adopt advanced RTSM platforms that provide automated tracking, secure data management, and compliance-ready reporting, thereby accelerating market growth.
Furthermore, the ongoing digital transformation of clinical trial operations, including the integration of AI and cloud-based technologies, is significantly enhancing the capabilities of RTSM platforms. Advanced systems now incorporate predictive analytics for supply optimization and risk management, improving trial efficiency and reducing costs. For instance, in June 2025, Oracle introduced interoperability enhancements in its RTSM solution, enabling drug pooling and automated supply management across multiple trials and vendors, thereby improving efficiency and accelerating clinical trial timelines.
Market Concentration & Characteristics
The global RTSM market is characterized by a high degree of innovation, with continuous advancements in AI, cloud-based platforms, and predictive analytics to improve trial efficiency and supply optimization. Vendors are increasingly integrating RTSM with EDC, CTMS, and eClinical ecosystems to enable real-time data synchronization. Recent industry insights highlight the growing use of AI-powered supply forecasting and cloud-native RTSM platforms, enhancing scalability and decision-making. Strong venture capital and private equity investments are accelerating innovation. New entrants introduce differentiated capabilities to compete with established players. For instance, in December 2025, Suvoda was recognized as a leader in Everest Group’s RTSM PEAK Matrix, reflecting its strong innovation and market adoption. Its IRT platform was highlighted for low-code configurability, modular architecture, and AI-enabled automation, along with the introduction of the “Sofia” AI assistant to enhance real-time decision-making and trial efficiency.
The market exhibits a moderate level of M&A activity, with companies focusing on capability-driven acquisitions in areas such as AI and analytics rather than large-scale consolidation. There is also a clear shift toward partnerships and platform integrations to build end-to-end clinical ecosystems. For instance, in January 2025, Atreo partnered with N-SIDE to integrate RTSM with advanced supply forecasting solutions. This collaboration enables end-to-end visibility, improved supply chain efficiency, and data-driven decision-making, helping accelerate clinical trials and reduce operational costs.
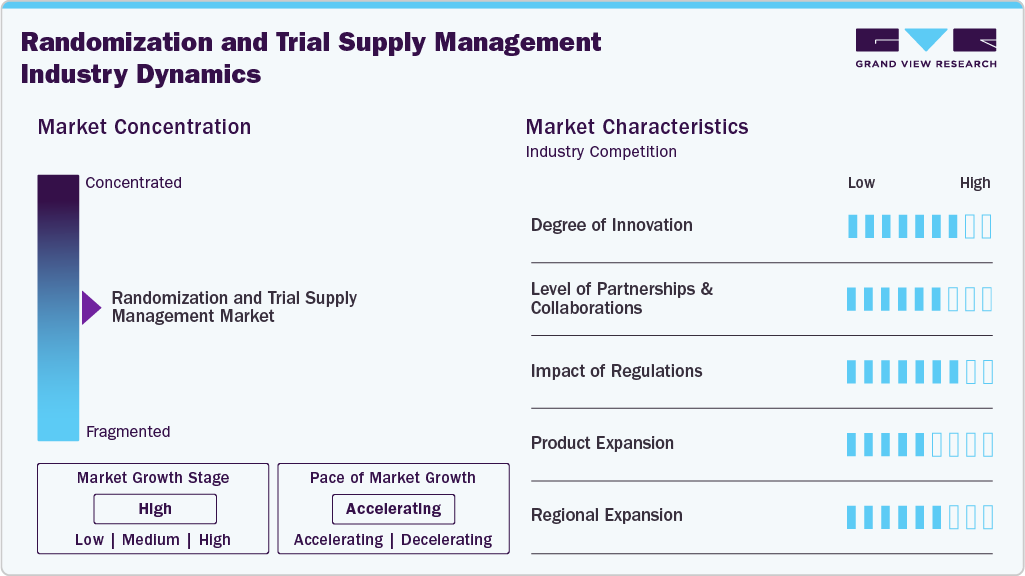
Regulatory frameworks such as FDA guidelines, EMA requirements, and ICH-GCP standards have a significant impact on the market. These regulations mandate data integrity, audit trails, and drug accountability, making RTSM systems essential for compliance. Increasing regulatory scrutiny and data privacy requirements (e.g., GDPR) are driving demand for validated, secure, and compliant RTSM platforms, leading to continuous investments in system validation and cybersecurity.
Product expansion in the RTSM market is characterized by the shift from standalone systems to integrated eClinical platforms. Vendors are increasingly enhancing their offerings with AI-driven analytics, real-time visibility, and interoperability to support complex clinical trials and improve operational efficiency. The growing focus on intelligent, scalable, and fully integrated RTSM solutions that enable better decision-making, optimize supply management, and support end-to-end clinical trial execution.
Delivery Mode Insights
Based on delivery mode, the market is segmented into cloud and web-based and on-premise. The cloud-based segment dominated the market with a revenue market share of 91.4% in 2025 and is anticipated to exhibit the fastest growth over the forecast period. This growth is driven by the scalability, flexibility, and ease of deployment across global clinical trials. These platforms enable real-time data access, centralized monitoring, and seamless collaboration among sponsors, CROs, and trial sites. The increasing adoption of decentralized clinical trials (DCTs) further drives the demand for cloud-based systems, as they facilitate remote access and distributed trial management. In addition, cloud deployment reduces IT infrastructure costs and implementation time, making it highly attractive for both large pharmaceutical companies and mid-sized biotech firms. Continuous advancements in AI integration and data analytics are further strengthening the dominance of this segment. For instance, in September 2023, Signant Health launched its integrated Signant Biotech platform, offering a unified cloud-based ecosystem combining EDC, eCOA, eConsent, RTSM, and telemedicine solutions, enabling faster and more efficient clinical trial execution across global studies in biopharmaceutical organizations.
Based on delivery mode, the on-premise segment is expected to grow significantly over the forecast period. This growth is driven by organizations with strict data security, regulatory, and customization requirements. Large pharmaceutical companies often prefer on-premise deployment to maintain greater control over sensitive clinical trial data and system configurations. These systems are also favored in regions or studies with stringent data residency and compliance regulations. Involving higher upfront costs and longer implementation timelines, on-premise solutions offer enhanced data ownership and tailored functionality. Growth in this segment is supported by organizations managing highly complex or confidential trials where full control over infrastructure is a priority.
End-use Insights
The CROs segment held the largest market share of 37.3% in 2025. This is due to the extensive involvement across the clinical trial value chain, including study design, patient recruitment, and trial execution. These organizations manage multiple trials simultaneously for various sponsors, creating a strong need for scalable and centralized RTSM solutions. Their ability to offer integrated services (R&D, data management, and logistics) further drives the adoption of advanced RTSM platforms. Additionally, increasing outsourcing of clinical trials by pharmaceutical and biotech companies has significantly strengthened the position of CROs. Their expertise in handling complex, multi-regional trials makes RTSM a critical tool in their operations.
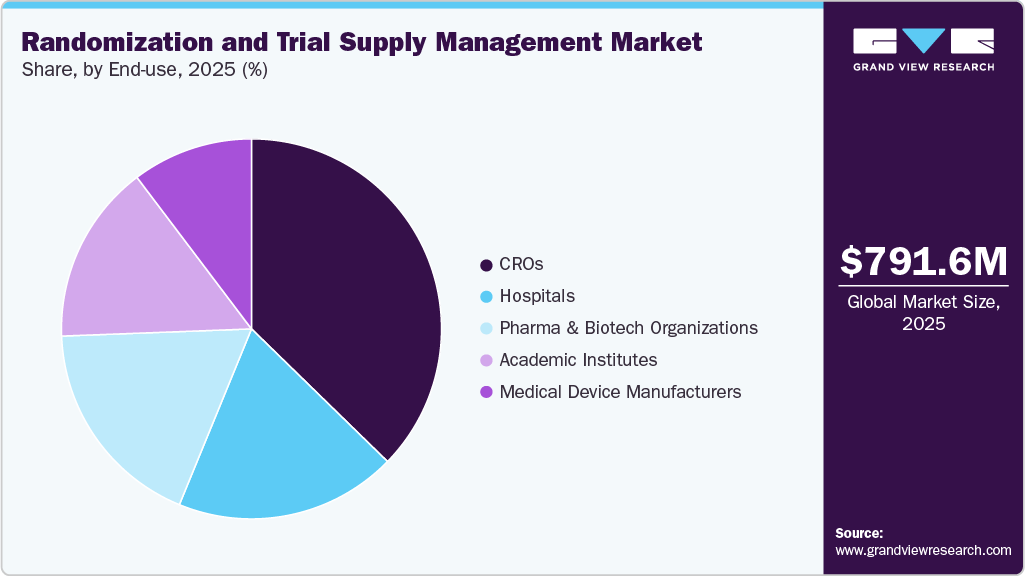
The pharmaceutical & biotechnology organizations segment is expected to grow significantly over the forecast period from 2026 to 2033. This growth is driven by rising investments in drug development and clinical research activities. These organizations are increasingly adopting RTSM platforms to gain greater control, transparency, and efficiency in trial operations. The shift toward in-house trial management and digital transformation strategies is further accelerating adoption. Moreover, the growing focus on personalized medicine, biologics, and advanced therapies requires sophisticated randomization and supply management capabilities. As a result, pharma and biotech firms are rapidly investing in integrated, cloud-based RTSM systems to enhance trial outcomes and reduce operational complexities.
Regional Insights
North America randomization and trial supply management market dominated the global market with a revenue share of 51.0% in 2025. This is driven by a well-established clinical research ecosystem and high adoption of advanced eClinical technologies. The presence of major pharmaceutical companies, CROs, and leading RTSM providers such as IQVIA and Oracle, along with significant investments in R&D, particularly in oncology, biologics, and precision medicine, supports market growth. In addition, stringent regulatory requirements from agencies such as the U.S. Food and Drug Administration drive the adoption of compliant and robust RTSM systems. The growing trend of decentralized clinical trials and digital transformation further accelerates market expansion.
U.S. Randomization and Trial Supply Management Market Trends
The randomization and trial supply management (RTSM) industry in the U.S. held the largest share in 2025. This is due to the high volume of clinical trials and advanced healthcare infrastructure. The presence of leading companies such as IQVIA, Veeva Systems Inc, and Medidata further strengthens the adoption of advanced RTSM platforms. The U.S. is the world’s largest clinical research hub, with over 165,409 registered clinical studies as of 2026, reflecting its strong R&D ecosystem and continuous demand for efficient trial management solutions. In addition, strict regulatory guidelines by the U.S. FDA ensure high standards of data integrity, transparency, and compliance, which further accelerate RTSM adoption.
Europe Randomization and Trial Supply Management Market Trends
Europe randomization and trial supply management (RTSM) industry is anticipated to witness the fastest CAGR of 14.7% over the forecast period from 2026 to 2033. This growth is driven by a strong clinical research ecosystem and well-established pharmaceutical industry. Countries such as Germany, the UK and France are key contributors due to high clinical trial activity and strong R&D investments. Strict regulatory frameworks under the European Medicines Agency (EMA) drive the need for compliant, transparent, and audit-ready RTSM systems. Increasing adoption of decentralized and cross-border clinical trials is further boosting demand for advanced RTSM platforms. In addition, growing collaboration between CROs and pharmaceutical companies is accelerating digital transformation in clinical trial management across Europe.
The randomization and trial supply management industry in Germany dominated the European market in 2025. This is due to the strong pharmaceutical manufacturing base and advanced clinical research infrastructure. The country’s high clinical trial activity, especially in oncology and biologics, drives demand for efficient RTSM solutions. Regulatory oversight by the Federal Institute for Drugs and Medical Devices (BfArM) and alignment with EMA guidelines ensure strict compliance and data integrity in trial processes. Increasing adoption of digital and cloud-based clinical trial technologies is further accelerating RTSM market growth. In addition, strong collaboration between CROs, academic institutions, and life sciences companies is accelerating digital transformation in clinical trial operations across Germany.
The randomization and trial supply management industry in the UK is experiencing significant growth, driven by a strong clinical research ecosystem and a high concentration of pharmaceutical and biotech companies. Supportive regulatory oversight by the Medicines and Healthcare Products Regulatory Agency (MHRA) ensures strict compliance and data integrity in clinical trials in the country. Increasing adoption of decentralized and hybrid clinical trial models is further driving demand for advanced RTSM solutions.
Asia Pacific Randomization and Trial Supply Management Market Trends
The Asia Pacific randomization and trial supply management (RTSM) industry is expected to witness significant growth over the forecast period, driven by increasing clinical trial activities and rising pharmaceutical R&D investments. Countries such as China, India, Japan, and South Korea are major contributors due to large patient pools, lower trial costs, and expanding CRO presence. Growing adoption of decentralized and hybrid clinical trials is further increasing the demand for advanced RTSM solutions in the region. Regulatory reforms and improving clinical trial infrastructure are also supporting market expansion. In addition, increasing participation of global pharmaceutical companies in Asia Pacific trials is accelerating the adoption of digital RTSM platforms. The region is emerging as a key hub for cost-efficient and large-scale clinical research operations. For instance, in August 2025, Takeda Pharmaceutical Company announced plans to conduct clinical trials in India to accelerate the development and launch of innovative therapies, including cancer drugs. The company highlighted India as a strategic growth market and aims to strengthen its R&D integration with global pipelines through local partnerships, reinforcing the country’s growing role in global drug development.
The randomization and trial supply management (RTSM) industry in China is expected to grow at a significant CAGR over the forecast period. This is driven by continuous regulatory modernization and digital transformation of clinical research infrastructure, a rapidly expanding clinical trial ecosystem, and growing investments in pharmaceutical R&D. The country is emerging as one of the fastest-growing markets in the Asia Pacific because of large patient pools, cost-efficient trial execution, and rising adoption of digital clinical trial technologies. The increasing use of cloud-based RTSM platforms and centralized trial management systems is accelerating market adoption. A strong focus on innovation in oncology, biologics, and precision medicine is driving demand for advanced RTSM solutions.
Latin America Randomization and Trial Supply Management Market Trends
The Latin America randomization and trial supply management (RTSM) industry is expected to witness significant growth over the forecast period, driven by the increasing number of clinical trials and rising outsourcing activities by global pharmaceutical companies. Countries such as Brazil, and Argentina are emerging as key clinical research hubs due to lower trial costs, diverse patient populations, and improving healthcare infrastructure. The region is also benefiting from growing adoption of cloud-based and web-based RTSM solutions, which enhance trial efficiency and regulatory compliance. In addition, increasing participation of CROs and multinational pharma companies is strengthening demand for advanced clinical trial technologies.
Middle East & Africa Randomization and Trial Supply Management Market Trends
The MEA randomization and trial supply management (RTSM) industry is expected to witness significant growth over the forecast period, supported by increasing clinical trial activities and growing interest from global pharmaceutical companies. The countries in the region such as Saudi Arabia, the UAE, and South Africa are emerging as key hubs due to improving healthcare infrastructure and rising investment in life sciences research. The region is also witnessing increased adoption of cloud-based and web-based RTSM solutions, which help overcome infrastructure limitations and improve trial efficiency. The collaborations between CROs and international pharma companies are strengthening clinical research capabilities in the region.
Key Randomization And Trial Supply Management Company Insights
Some key players in the randomization and trial supply management industry include IQVIA, Oracle, Veeva Systems Inc, and Medidata. IQVIA offers integrated RTSM/IRT capabilities within its broader clinical research ecosystem, supporting complex global trials. Oracle and Veeva Systems provide cloud-based RTSM platforms with strong interoperability, scalability, and integration across clinical trial workflows, while Medidata delivers advanced RTSM through its Rave Clinical Cloud, enabling seamless randomization and supply management. These companies are actively focusing on strategies such as platform integration, cloud expansion, AI-driven innovation, and partnerships to strengthen their market presence and enhance end-to-end clinical trial management capabilities.
Key Randomization & Trial Supply Management Companies:
The following key companies have been profiled for this study on the randomization and trial supply management (RTSM) market.
- IQVIA
- Oracle
- Medidata (Acquired by Dassault Systèmes)
- Veeva Systems Inc.
- Parexel
- Suvoda LLC
- 4G Clinical
- Signant Health
- Calyx
- Almac Group
- Clinion
- Mednet
- ICON plc
Recent Developments
- In April 2025, Atreo launched its v2.0 RTSM platform, featuring enhanced configurability, improved UI/UX, and advanced cross-protocol reporting capabilities. The upgrade aims to streamline complex clinical trial designs, reduce custom coding, and accelerate drug development through more agile and scalable RTSM solutions.
"The launch of v2.0 marks a major milestone for Atreo, our client partners, and site users, reflecting our dedication to continuous innovation driving a simplified, high-quality experience for our global users, We're proud to provide a platform that not only addresses the unique challenges faced by large pharma but also accelerates the journey of bringing vital therapies to patients worldwide for sponsors of all sizes. Having witnessed firsthand the downstream impact of stalled R&D efforts at companies in our field, Atreo's leadership team remains committed to delivering future releases that continually improve client and site experiences. Our unwavering commitment also enables Atreo to overcome the scalability challenges and limitations of outdated RTSM technology, delivering curated solutions without cumbersome SDLC models, costly delays, and quality issues common of yesterday's options."
- Ryan Harrison, Atreo CEO.
-
In November 2024, Perceptive eClinical launched ClinPhone 5, an advanced Randomization and Trial Supply Management (RTSM) platform developed with CRScube. The cloud-native, multi-tenant platform accelerates clinical trial setup and delivery, offering agility, adaptability, scalability, advanced drug supply forecasting, and medication pooling. It enhances efficiency and reliability for sponsors and CROs globally.
-
In March 2024, Signant Health enhanced its clinical supply management with updates to SmartSignals GxP Inventory and RTSM solutions, introducing the Essential+ module for streamlined multi-trial inventory control and a free picking feature in RTSM. These innovations improve inventory visibility, reduce waste and errors, and enable just-in-time packaging and labeling, boosting efficiency, compliance, and overall trial success.
"In a clinical supply landscape characterized by fragmented, disparate systems with hidden risks and costs at every stage, our GxP Inventory Essential+ and RTSM solutions provide sponsors with the ability to reduce risk, improve visibility and traceability, and simplify workflows”.
- Oli Cunningham, vice president clinical supply chain solutions.
Randomization And Trial Supply Management Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 886.15 million
Revenue forecast in 2033
USD 2,083.8 million
Growth rate
CAGR of 13.0% from 2026 to 2033
Historical data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company share, competitive landscape, growth factors and trends
Segments covered
Delivery mode, end-use, region
Regional Scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country Scope
U.S.; Canada; Mexico; Germany; UK; France; Italy; Spain; Norway; Denmark; Sweden; Japan; China; India; Australia; Thailand; South Korea; New Zealand; Taiwan; Hong Kong; Singapore; Vietnam; Brazil; Argentina; Chile; South Africa; Saudi Arabia; UAE; Egypt; Qatar; Kuwait
Key companies profiled
IQVIA; Oracle; Medidata (Acquired by Dassault Systèmes); Veeva Systems Inc.; Parexel; Suvoda LLC; 4G Clinical; Signant Health; Calyx; Almac Group; Clinion; Mednet; ICON plc
Customization scope
Free report customization (equivalent up to 8 analysts’ working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Randomization And Trial Supply Management Market Report Segmentation
This report forecasts revenue growth at global, regional & country levels and provides an analysis of the industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global randomization and trial supply management (RTSM) market report based on delivery mode, end-use, and region:
-
Delivery Mode Outlook (Revenue, USD Million, 2021 - 2033)
-
On-premise
-
Cloud and web-based
-
-
End-use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
CROs
-
Academic Institutes
-
Pharma & Biotech Organizations
-
Medical Device Manufacturers
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Netherlands
-
Norway
-
Denmark
-
Sweden
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
New Zealand
-
Taiwan
-
Hong Kong
-
Singapore
-
Vietnam
-
-
Latin America
-
Brazil
-
Argentina
-
Chile
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Egypt
-
Qatar
-
Kuwait
-
-
About the authors:
Author: GVR Healthcare IT Research Team | Last Updated:
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









