- Home
- »
- Plastics, Polymers & Resins
- »
-
U.S. Medical Grade Polypropylene Market Size Report, 2033GVR Report cover
![U.S. Medical Grade Polypropylene Market Size, Share & Trends Report]()
U.S. Medical Grade Polypropylene Market (2025 - 2033) Size, Share & Trends Analysis Report By Type (Homopolymer Polypropylene, Random Copolymer Polypropylene, Impact Copolymer Polypropylene), By Application (Medical Devices), And Segment Forecasts
- Report ID: GVR-4-68040-684-9
- Number of Report Pages: 90
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Bulk Chemicals
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Medical Grade Polypropylene Market Summary
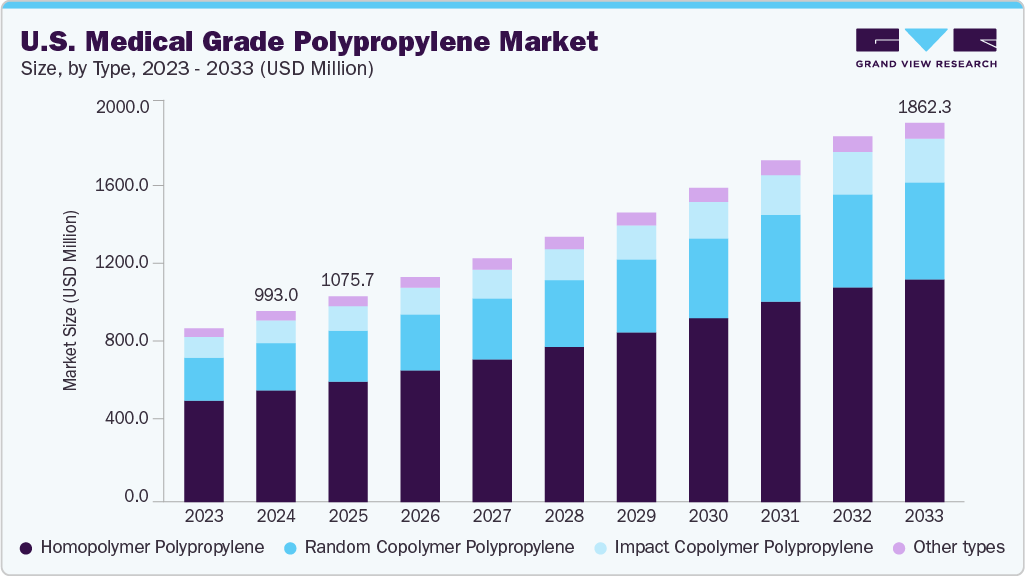
The U.S. medical grade polypropylene market size was estimated at USD 993.0 million in 2024 and is projected to reach USD 1,862.32 million by 2033, growing at a CAGR of 7.1% from 2025 to 2033. The U.S. market is anticipated to experience consistent growth, fueled by the growing need for reliable, durable, and affordable materials in the healthcare industry.
Key Market Trends & Insights
- Homopolymer polypropylene dominated the type segment in 2024 with a market share of 58.52%.
- By type, the random copolymer polypropylene segment is expected to grow at the fastest CAGR of 7.5% from 2025 to 2033 in terms of revenue.
- By applications, the drug delivery systems segment is expected to grow at the fastest CAGR of over 7% from 2025 to 2033 in terms of revenue.
Market Size & Forecast
- 2024 Market Size: USD 993.00 Million
- 2033 Projected Market Size: USD 1,862.32 Million
- CAGR (2025-2033): 7.1%
Increased investments in the manufacturing of advanced medical devices, along with a significant focus on domestic production and adherence to regulations, are contributing to the rising adoption of medical-grade polypropylene across the country.The market for medical-grade polypropylene in the U.S. is expected to grow steadily, driven by rising demand in key sectors such as medical devices, diagnostics, pharmaceutical packaging, and laboratory equipment. The material's lightweight nature, chemical resistance, and compatibility with various sterilization methods are essential for ensuring patient safety, maintaining product quality, and offering cost-effective healthcare solutions.
Furthermore, rising healthcare expenditure, ongoing improvements in medical facilities, and the expansion of pharmaceutical and diagnostic services across the U.S. are promoting greater use of medical-grade polypropylene. The market is also supported by the increasing need for high-performance, patient-safe, and regulation-compliant materials in clinical settings. As the U.S. healthcare industry continues to focus on cost-efficiency, sterility, and material traceability, the demand for medical-grade polypropylene is expected to grow steadily in the coming years.
Drivers, Opportunities & Restraints
The market for medical-grade polypropylene in the U.S. is expected to grow steadily, driven by rising demand in key areas such as medical devices, pharmaceutical packaging, diagnostics, and laboratory instruments. Its lightweight design, excellent chemical resistance, and ability to withstand various sterilization methods, including steam, ethylene oxide, and gamma irradiation, make polypropylene essential for secure, single-use medical products and effective healthcare delivery.
In the U.S. medical-grade polypropylene industry, new opportunities are emerging due to a growing focus on sustainability, compliance with regulations, and increased efficiency in healthcare systems. The adoption of polypropylene variants that are recyclable, biocompatible, and capable of withstanding standard sterilization processes is growing, as hospitals and manufacturers seek environmentally friendly and safe materials. Additionally, the rising demand for point-of-care diagnostics, the expansion of home-based healthcare services, and the increase in domestic production of single-use medical and pharmaceutical products are creating significant opportunities for polypropylene to provide safe, compliant, and cost-effective healthcare solutions.
The U.S. medical-grade polypropylene industry is expected to face challenges, primarily due to rising environmental concerns about plastic waste and the limited recyclability of some single-use healthcare products. Increased regulatory oversight regarding material biocompatibility, traceability, and sterilization compatibility is likely to raise development and compliance costs for manufacturers. Additionally, fluctuations in raw material prices and potential disruptions in both domestic and global supply chains may impact production stability. The market is also seeing increased competition from alternative materials like medical-grade polyolefins and specialty polymers, which could limit the use of polypropylene in certain advanced or high-performance medical applications.
Market Concentration & Characteristics
The market for medical-grade polypropylene in the U.S. is experiencing steady growth, fueled by increasing demand in key healthcare sectors such as medical devices, pharmaceutical packaging, diagnostics, and laboratory tools. Despite its fragmentation, major companies continue to exert significant influence by leveraging their extensive regulatory expertise, a wide range of product options, and strong supply chains both domestically and globally. Leading firms like LyondellBasell, Borealis AG, SABIC, ExxonMobil, TotalEnergies Corbion, and INEOS Olefins & Polymers play a crucial role in shaping market trends in the U.S. These companies pioneer innovations, working on high-purity medical-grade polypropylene formulations, sustainable and recyclable solutions, and high-performance materials that meet the strict standards set by U.S. regulatory agencies such as the FDA and USP. Their efforts align with the evolving priorities of the American healthcare system, which emphasizes patient safety, cost efficiency, and environmental sustainability.
The U.S. medical-grade polypropylene industry is transforming through strategic mergers and acquisitions, enabling companies to expand their product lines and enhance supply chain efficiency. These efforts are driving innovation toward environmentally friendly, biocompatible options in response to stricter FDA regulations and sustainability goals. Regulatory guidelines set by the FDA and USP influence material choices, encouraging the use of recyclable, non-toxic, and sterilization-compatible polymers. Rising compliance costs and evolving healthcare demands are prompting investments in cleaner processing methods and improved polypropylene formulations.
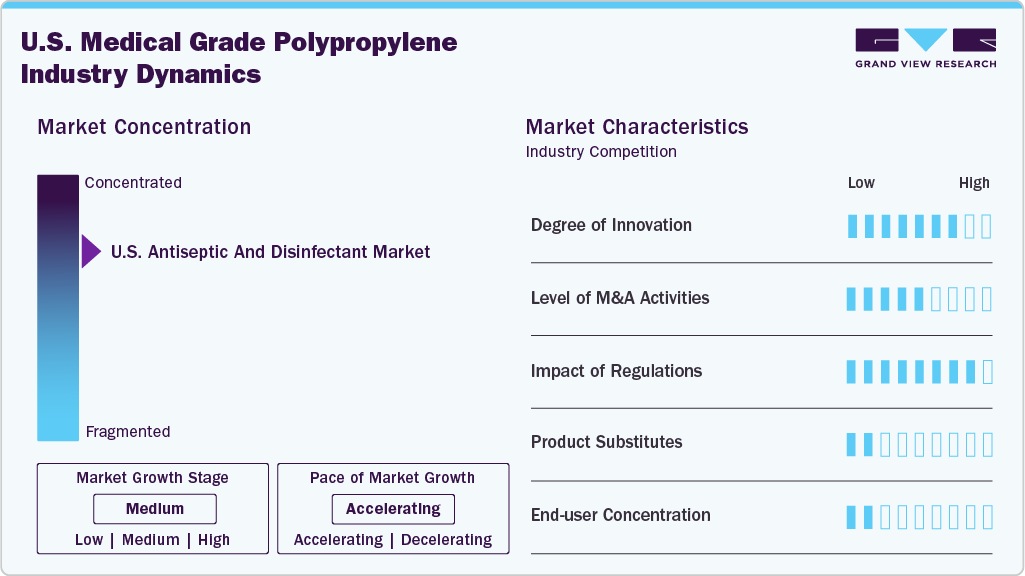
The U.S. market for medical-grade polypropylene experiences moderate competition from other materials such as medical-grade polyethylene, polycarbonate, and specialized polymers like TPE and TPU. Nonetheless, polypropylene maintains a robust presence in the market due to its lightweight nature, resistance to chemicals, compatibility with sterilization processes, and cost-effectiveness. In the U.S., there is a notable concentration among end users, particularly within the medical device, pharmaceutical, and diagnostics industries. Prominent OEMs, healthcare providers, and contract manufacturers significantly influence procurement strategies, impacting both product development and material choices. Consequently, the market is highly reactive to alterations in FDA regulations, cycles of innovation, and evolving requirements from end users.
Type Insights
In the U.S. market for medical-grade polypropylene, homopolymer polypropylene has become the leading type by market share, accounting for 58.52% of revenue in 2024. This dominance is due to its wide use in medical devices, syringes, containers, and caps, thanks to its high stiffness, chemical resistance, and suitability for sterilization. Continued strong demand is expected, supported by ongoing growth in hospital infrastructure, increased domestic production of single-use medical items, and strict hygiene and regulatory standards from the FDA. These factors are reinforcing the preference for homopolymer PP across the U.S. healthcare supply chain.
In the U.S. market for medical-grade polypropylene, random copolymer polypropylene is anticipated to experience the fastest CAGR during the projected period. This growth can be attributed to its rising use in medical packaging, IV components, diagnostic tools, and transparent lab equipment, where clarity and durability are essential. The increasing application of single-use medical items and point-of-care diagnostics, along with stricter FDA regulations concerning safety and performance, is predicted to enhance the demand for random copolymer polypropylene further. Demand will especially surge in regions with advanced healthcare systems and developing diagnostic technologies.
Application Insights
In the U.S. medical-grade polypropylene market, the medical devices segment led the application market, capturing 30.83% of the share in 2024. This dominance arises from the widespread use of polypropylene in syringes, IV components, diagnostic kits, surgical tools, and single-use medical instruments. Its favorable properties, such as chemical resistance, lightweight structure, biocompatibility, and compatibility with steam, gamma, and ethylene oxide sterilization, make it a preferred material for high-volume, disposable medical products. Ongoing investments in hospital infrastructure, the increasing number of surgical procedures, and the continued demand for cost-effective and safe materials are expected to sustain the segment's growth in the coming years.
In the U.S. market for medical-grade polypropylene, the segment focused on medical packaging is expected to see significant growth during the forecast period. This growth is fueled by the increasing demand for lightweight, cost-effective, and sterile packaging solutions in the pharmaceutical and healthcare industries. Medical-grade polypropylene is increasingly used in blister packs, vials, caps, pouches, and rigid containers due to its resistance to chemicals, durability, and compatibility with sterilization processes. The rising emphasis on patient safety, longer shelf life, and compliance with FDA packaging regulations is encouraging the use of high-performance polypropylene materials in medical packaging applications across the U.S.
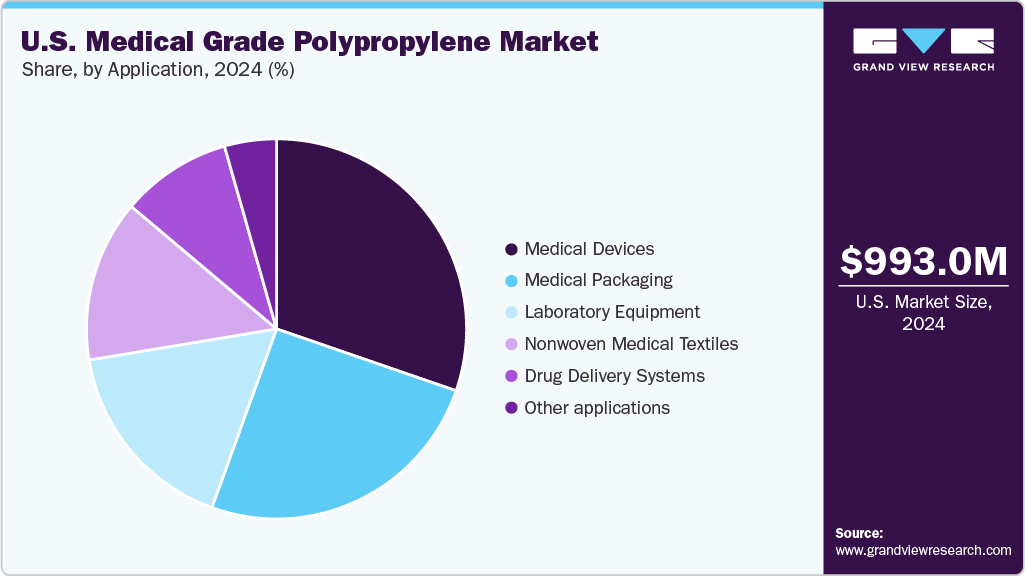
In the U.S. market for medical-grade polypropylene, the segment for drug delivery systems is projected to experience considerable growth, fueled by the increasing demand for safe, effective, and cost-efficient materials in the distribution of pharmaceuticals. Medical grade polypropylene is commonly utilized in inhalers, syringes, ampoules, cartridges, and auto-injectors because of its outstanding chemical resistance, dimensional stability, and compatibility with sterilization techniques.
Key U.S. Medical Grade Polypropylene Company Insights
The market for medical-grade polypropylene in the U.S. is currently experiencing moderate growth, driven by its increasing use in medical devices, pharmaceutical packaging, diagnostics, and laboratory instruments. While the market remains somewhat fragmented, leading companies hold significant influence over the competitive landscape due to their specialized expertise, regulatory compliance, and established distribution networks. Companies such as LyondellBasell, Borealis AG, SABIC, ExxonMobil, TotalEnergies Corbion, INEOS Olefins & Polymers, and Mitsui Chemicals are shaping the market. These firms are creating polypropylene formulations that are biocompatible, resistant to sterilization, and meet FDA standards, aligning with the changing regulations and sustainability demands of the U.S. healthcare system. Their investments in advanced materials and cleaner manufacturing processes are addressing the need for safe, efficient, and eco-friendly solutions for high-volume medical applications.
Key U.S. Medical Grade Polypropylene Companies:
- LyondellBasell
- Borealis AG
- SABIC
- ExxonMobil
- TotalEnergies Corbion
- INEOS Olefins & Polymers
- Mitsui Chemicals
- HMC Polymers
- Braskem
- RTP Company
Recent Developments
-
In July 2025, Johnson & Johnson announced impressive earnings for Q2 2025 and revised its predicted impact of U.S. tariffs on Chinese imports down to $200 million, a reduction from the previous estimate of $400 million. The company credited the favorable outlook to successful adjustments in its supply chain and a rise in demand for medical technologies, such as devices and drug delivery systems, which heavily utilize medical-grade polypropylene. Additionally, J&J adjusted its full-year sales growth forecast upwards to 3.2-3.7%, highlighting strong performance in the medical technology sector.
U.S. Medical Grade Polypropylene Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 1075.7 million
Revenue forecast in 2033
USD 1,862.32 million
Growth rate
CAGR of 7.1% from 2025 to 2033
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion, Volume in Kilotons, and CAGR from 2025 to 2033
Report coverage
Revenue forecast, competitive landscape, growth factors, and trends
Segments covered
Type, applications, region
Country Scope
U.S.
Key companies profiled
LyondellBasell; Borealis AG, SABIC, ExxonMobil, TotalEnergies Corbion, INEOS Olefins & Polymers, Mitsui Chemicals, HMC Polymers, Braskem, RTP Company
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Medical Grade Polypropylene Market Report Segmentation
This report forecasts revenue growth at the country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For the purpose of this study, Grand View Research has segmented the U.S. medical grade polypropylene market report on the basis of type and applications:
-
Type Outlook (Revenue, USD Million; Volume, Kilotons; 2021 - 2033)
-
Homopolymer Polypropylene
-
Random Copolymer Polypropylene
-
Impact Copolymer Polypropylene
-
Other types
-
-
Applications Outlook (Revenue, USD Million; Volume, Kilotons; 2021 - 2033)
-
Medical Devices
-
Medical Packaging
-
Laboratory Equipment
-
Nonwoven Medical Textiles
-
Drug Delivery Systems
-
Other applications
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










