- Home
- »
- Plastics, Polymers & Resins
- »
-
Biocompatible 3D Printing Resin Market Size Report, 2033GVR Report cover
![Biocompatible 3D Printing Resin Market Size, Share & Trends Report]()
Biocompatible 3D Printing Resin Market (2025 - 2033) Size, Share & Trends Analysis Report By Product Type (Photo-curable Resins, Hydrogels & Bio-inks, Thermoplastic Biocompatible Filaments), By End-use (Dental, Implants & Prosthetics, Surgical Guides & Devices), By Region, And Segment Forecasts
- Report ID: GVR-4-68040-790-4
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Bulk Chemicals
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Biocompatible 3D Printing Resin Market Summary
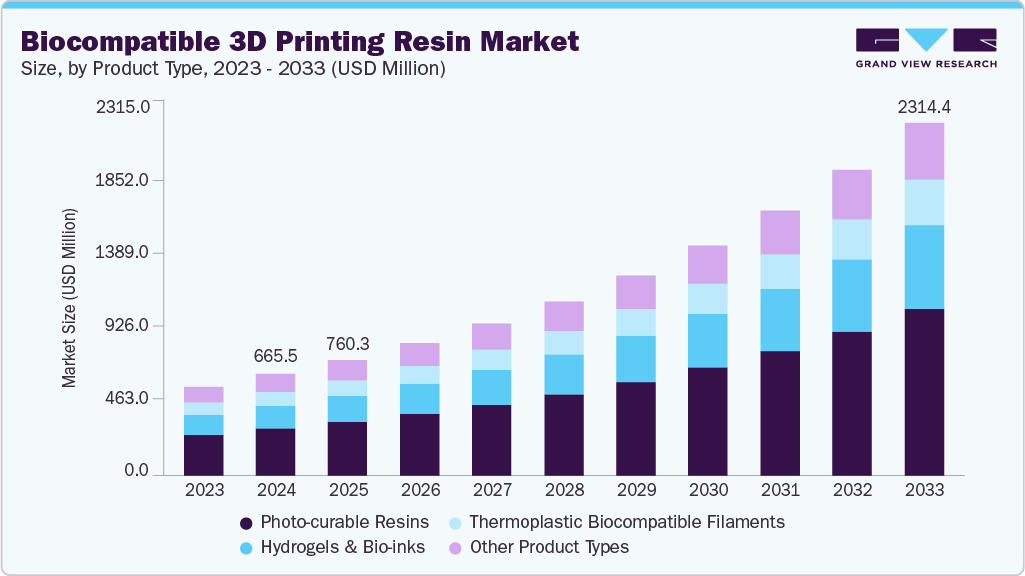
The global biocompatible 3D printing resin market size was estimated at USD 665.5 million in 2024 and is projected to reach USD 2,314.4 million by 2033, growing at a CAGR of 14.9% from 2025 to 2033. Growing adoption of digital dentistry and surgical planning software is driving demand for biocompatible resins that integrate seamlessly with 3D printing workflows.
Key Market Trends & Insights
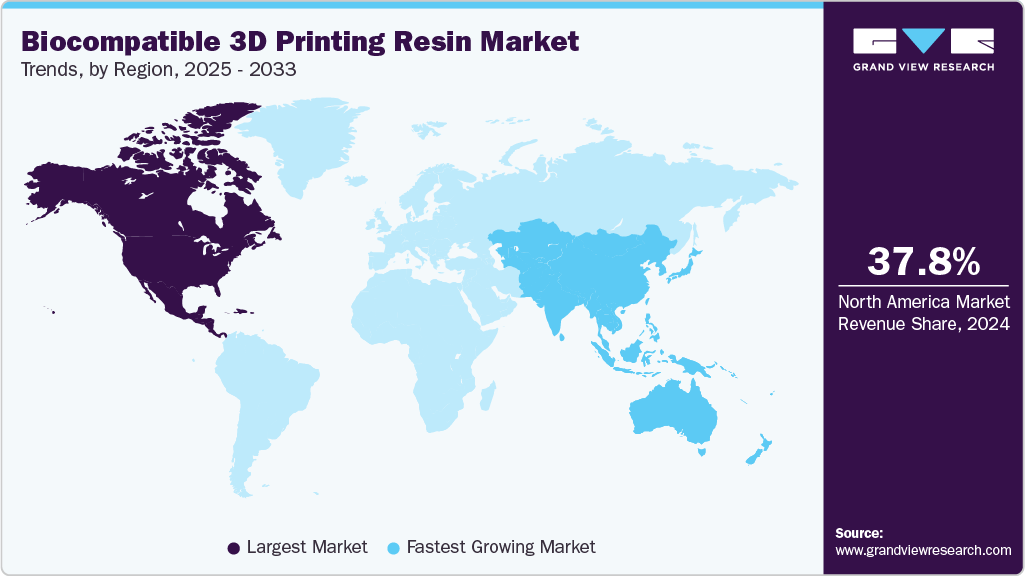
- North America dominated the biocompatible 3D printing resins market with the largest revenue share of 37.82% in 2024.
- The biocompatible 3D printing resins market in Canada is expected to grow at a substantial CAGR of 15.1% from 2025 to 2033.
- By product type, the hydrogels & bio-inks segment is expected to grow at a considerable CAGR of 15.7% from 2025 to 2033 in terms of revenue.
- By end use, the tissue engineering/bioprinting segment is expected to grow at a considerable CAGR of 15.5% from 2025 to 2033 in terms of revenue.
Market Size & Forecast
- 2024 Market Size: USD 665.5 Million
- 2033 Projected Market Size: USD 2,314.4 Million
- CAGR (2025-2033): 14.9%
- North America: Largest market in 2024
- Asia Pacific: Fastest growing market
Hospitals and clinics are increasingly investing in in-house printing capabilities to improve efficiency and reduce outsourcing costs. The market is maturing from generic biocompatible photopolymers to application-specific resin families tailored for dental restorations, surgical guides, hearing aids and implantable prototypes. Manufacturers increasingly offer graded performance portfolios that balance cytocompatibility, mechanical strength and sterilization resilience, enabling wider clinical adoption. Parallel improvements in resin chemistry and printer software have shortened qualification cycles for point of care production, shifting demand toward validated, end-use ready material systems. This specialization is accelerating commercial partnerships between resin suppliers, device OEMs and contract manufacturers.
Drivers, Opportunities & Restraints
Rising clinical demand for patient specific devices and the operational advantages of point of care manufacturing are driving uptake of biocompatible resins. 3D printing reduces lead times for custom dental prostheses, orthodontic appliances and surgical planning models while lowering inventory and logistics costs for hospitals and clinics. Regulatory bodies in key markets are clarifying pathways for 3D printed devices, encouraging hospitals and medtech firms to invest in qualified resin-printer workflows. As clinical workflows prioritize speed and customization, resin suppliers that can demonstrate consistent biocompatibility and process robustness capture significant share.
There is a clear growth opportunity in developing resins that support biological integration, including biodegradable load bearing formulations and cell-laden hydrogels for tissue engineering. Clinical translation of resorbable scaffolds and bioactive implants would open large markets in orthopedics and regenerative medicine where current solutions are limited. Suppliers that can couple proprietary chemistries with validated sterilization and long term safety data, and that partner with academic and clinical research centers, stand to establish high barriers to entry and premium pricing. Geographic expansion into Asia Pacific and Latin America also offers high incremental volume as hospital infrastructure upgrades.
Commercialization of biocompatible resins faces lengthy and expensive biocompatibility testing, sterilization validation and clinical qualification, which slows new product introductions and raises total cost of ownership for customers. Fragmented printer hardware and post processing requirements mean a material that is validated on one platform may not be transferable elsewhere, limiting scale.
Market Concentration & Characteristics
The market growth stage of the biocompatible 3D printing resins market is high, and the pace is accelerating. The market exhibits consolidation, with key players dominating the industry landscape. Major companies like 3Dresyns, Tetra Growth LLC, Liqcreate, BMF Materials, Stratasys, and others play a significant role in shaping the market dynamics. These leading players often drive innovation within the market, introducing new products, technologies, and end uses to meet evolving industry demands.

Innovation in biocompatible resins is accelerating along two tracks: functional chemistry that delivers graded mechanical performance and long-term biological safety, and process-coupled developments that reduce qualification time for clinical workflows. Companies are investing in photo-initiator systems and polymer backbones that tolerate sterilization and deliver predictable post-cure properties, while collaborative validation with printer OEMs is shortening time to hospital adoption. Breakthroughs in miniaturized curing hardware and formulation tuning are also expanding usable clinical applications from dental restorations to certified surgical guides.
Resins compete with a range of established and emerging materials depending on the clinical use case. Metals and ceramic powders remain preferred for load-bearing implants where fatigue life and osseointegration are critical, while thermoplastics and silicone-based materials offer lower-cost, regulatory-familiar routes for non-implantable devices and soft-tissue applications. Hydrogels and cell-compatible bioinks are displacing resins in tissue engineering and bioprinting because they better support cell viability and matrix remodeling, creating parallel value chains rather than direct one-to-one substitution in many applications. Strategic material selection now depends on trade-offs between regulatory complexity, mechanical requirements, and biological functionality.
Product Type Insights
Photo-curable resins dominated the biocompatible 3D printing resins market across the product segmentation in terms of revenue, accounting for a market share of 46.26% in 2024 and is forecasted to grow at 15.2% CAGR from 2025 to 2033. Manufacturers are accelerating development of fast-curing, printer-validated photopolymers that deliver repeatable part accuracy and post-cure performance required by clinical workflows.
This technical focus reduces cycle time for dental and surgical applications and enables higher throughput in centralized production labs, making photo-curable resins the preferred route for short-run, high-precision medical parts. As OEMs certify resin-printer combinations, buyers favor suppliers that can provide turnkey material-process packages, shifting purchasing decisions toward validated photopolymer portfolios.
The hydrogels & bio-inks segment is anticipated to grow at a substantial CAGR of 15.7% through the forecast period. Demand for hydrogels and extracellular matrix derived bio-inks is being driven by their superior ability to support cell viability and tissue maturation in regenerative medicine research.
Academic and commercial bioprinting programs are funding formulation work that mimics native tissue mechanics and biochemical cues, accelerating translation from bench to first-in-human studies. This research-led pull creates a premium for suppliers who can offer biologically functional, reproducible hydrogel platforms compatible with multiple bioprinter modalities
End Use Insights
Dental dominated the biocompatible 3D printing resins market across the end use segmentation in terms of revenue, accounting for a market share of 37.70% in 2024 and is forecasted to grow at 14.5% CAGR from 2025 to 2033. Dental clinics and labs are rapidly adopting biocompatible resins because in-house printing cuts lead time, lowers outsourcing cost and supports personalized prosthetics at scale. Commercial milestones such as large OEM contracts for direct-tooth workflows and regulatory clearances for denture and aligner technologies are catalyzing investment in validated resin-printer ecosystems. Consequently, resin vendors that demonstrate consistent clinical accuracy and regulatory support capture accelerated uptake across both labs and point-of-care settings.
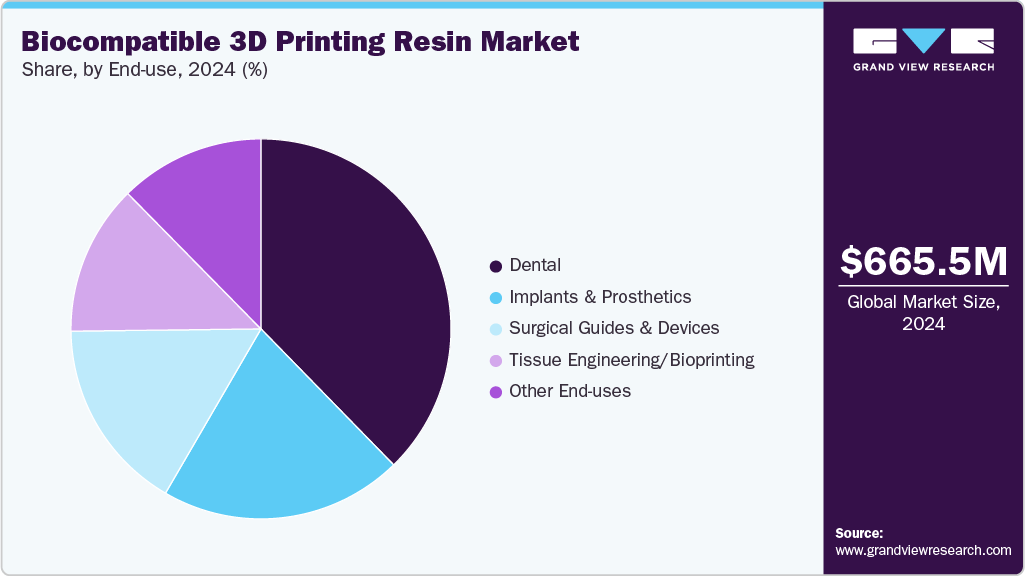
The tissue engineering / bioprinting segment is expected to expand at a substantial CAGR of 15.5% through the forecast period. The tissue engineering segment is pulling specialized resins and scaffold chemistries through increased translational funding and a wave of early-stage clinical trials for bioprinted implants. Investors and research consortia prioritize materials that enable cell infiltration, controlled resorption and regulatory tractability, creating a commercial advantage for suppliers collaborating with hospitals and academic centers. As preclinical success compels larger validation programs, demand shifts toward material developers who can supply scaled, quality controlled batches for clinical studies.
Regional Insights
North America held the largest share of 37.82% in terms of revenue of the biocompatible 3D printing resins market in 2024 and is expected to grow at the fastest CAGR of 14.1% over the forecast period. Large hospital systems and specialized surgical centers in North America are scaling in-house 3D printing to shorten surgical planning cycles and produce patient specific devices on demand, driving steady demand for validated biocompatible resins that fit clinical workflows.
Adoption is being supported by growing investments in hospital additive manufacturing labs and by supplier efforts to deliver printer-validated material-process packages that reduce qualification burden for health systems. This operational shift from outsourcing to on-premise production raises predictable recurring demand for certified resin consumables and post processing solutions.
U.S. Biocompatible 3D Printing Resin Market Trends
In the U.S., evolving FDA guidance for additive manufacturing and clearer expectations around device and manufacturing controls are lowering entry friction for clinically oriented resin suppliers. Coupled with a sizeable and well-funded healthcare market that prioritizes point of care manufacturing, these regulatory and commercial conditions accelerate adoption of resins that can demonstrate process traceability and repeatable biocompatibility. Vendors that translate regulatory intelligence into ready-to-use validated workflows gain preferential access to hospital procurement budgets.
Europe Biocompatible 3D Printing Resin Market Trends
Europe’s Medical Device Regulation and related biomaterials scrutiny force suppliers to invest in extended biocompatibility testing and clinical evidence, which increases time to market but creates a premium for materials with comprehensive dossiers. Buyers in Europe reward vendors offering certified material-printer combinations and full technical documentation that simplifies notified body reviews. As a result, firms that build robust regulatory and quality systems convert compliance burden into a commercial moat and stronger long-term contracts with clinics and dental labs.
Asia Pacific Biocompatible 3D Printing Resin Market Trends
Asia Pacific is experiencing one of the fastest growth rates for bioprinting and dental additive manufacturing driven by public and private investment, expanding medical device manufacturing capacity and rising demand for affordable personalized care. China, Japan and South Korea are scaling bioprinting research and local production of photopolymers while dental chains across the region adopt intraoral scanning and in-house printing at scale. This combination of demand pull and local manufacturing capacity creates a high-volume growth corridor for suppliers who can localize production and meet regional regulatory requirements.
Key Biocompatible 3D Printing Resin Company Insights
The Biocompatible 3D Printing Resins Market is highly competitive, with several key players dominating the landscape. Major companies include 3Dresyns, Tetra Growth LLC, Liqcreate, BMF Materials, and Stratasys. The biocompatible 3D printing resins market is characterized by a competitive landscape with several key players driving innovation and market growth. Major companies in this sector are investing heavily in research and development to enhance the performance, cost-effectiveness, and sustainability of their types.
Key Biocompatible 3D Printing Resin Companies:
The following are the leading companies in the biocompatible 3D printing resins market. These companies collectively hold the largest market share and dictate industry trends.
- 3Dresyns
- Tetra Growth LLC
- Liqcreate
- BMF Materials
- Stratasys
Recent Developments
-
In November 2024, Henkel unveiled new Loctite 3D material additions and showcased a medical-grade resin at Formnext. The company positioned Loctite MED3394 and other portfolio extensions to address growing demand for certified medical and industrial photopolymers.
-
In January 2024, Formlabs announced a resin pumping system and two new materials, including Premium Teeth Resin. The release targeted dental workflows by improving material handling and introducing a higher-performance, biocompatible teeth resin for in-house dental production.
Biocompatible 3D Printing Resin Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 760.3 million
Revenue forecast in 2033
USD 2,314.4 million
Growth rate
CAGR of 14.9% from 2025 to 2033
Historical data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million, volume in kilotons, and CAGR from 2025 to 2033
Report coverage
Revenue & volume forecast, competitive landscape, growth factors, and trends
Segment covered
Product type, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country Scope
U.S.; Canada; Mexico; Germany; France; UK; Spain; Italy; China; Japan; India; South Korea; Brazil; Argentina; Saudi Arabia; South Africa; UAE
Key companies profiled
3Dresyns; Tetra Growth LLC; Liqcreate; BMF Materials; Stratasys
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Biocompatible 3D Printing Resin Market Report Segmentation
This report forecasts revenue growth at the global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For the purpose of this study, Grand View Research has segmented the biocompatible 3D printing resin market report on the basis of product type, end use, and region:

-
Product Type Outlook (Revenue, USD Million; Volume, Kilotons; 2021 - 2033)
-
Photo-curable Resins
-
Hydrogels & Bio-inks
-
Thermoplastic Biocompatible Filaments
-
Other Product Types
-
-
End Use Outlook (Revenue, USD Million; Volume, Kilotons; 2021 - 2033)
-
Dental
-
Implants & Prosthetics
-
Surgical Guides & Devices
-
Tissue engineering / Bioprinting
-
Other End Uses
-
-
Regional Outlook (Revenue, USD Million; Volume, Kilotons; 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
-
Asia Pacific
-
China
-
India
-
Japan
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
Saudi Arabia
-
South Africa
- UAE
-
-
Frequently Asked Questions About This Report
b. The global biocompatible 3D printing resins market size was estimated at USD 665.5 million in 2024 and is expected to reach USD 760.3 million in 2025.
b. Growing adoption of digital dentistry and surgical planning software is driving demand for biocompatible resins that integrate seamlessly with 3D printing workflows. Hospitals and clinics are increasingly investing in in-house printing capabilities to improve efficiency and reduce outsourcing costs.
b. The global biocompatible 3D printing resins market is expected to grow at a compound annual growth rate of 14.9% from 2025 to 2033 to reach USD 2,314.4 million by 2033.
b. Photo-curable resins dominated the biocompatible 3D printing resins market across the product segmentation in terms of revenue, accounting for a market share of 46.26% in 2024 and is forecasted to grow at 15.2% CAGR from 2025 to 2033.
b. Some key players operating in the biocompatible 3D printing resins market include 3Dresyns, Tetra Growth LLC, Liqcreate, BMF Materials, and Stratasys
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










