- Home
- »
- Healthcare IT
- »
-
Middle East Clinical Trials Market Size, Industry Report 2033GVR Report cover
![Middle East Clinical Trials Market Size, Share & Trends Report]()
Middle East Clinical Trials Market (2025 - 2033 ) Size, Share & Trends Analysis Report By Phase (Phase I, Phase II, Phase III, Phase IV), By Study Design, By Indication, By Indication By Study Design, By Service, By Sponsor, By Country, And Segment Forecasts
- Report ID: GVR-4-68040-729-0
- Number of Report Pages: 180
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Middle East Clinical Trials Market Summary
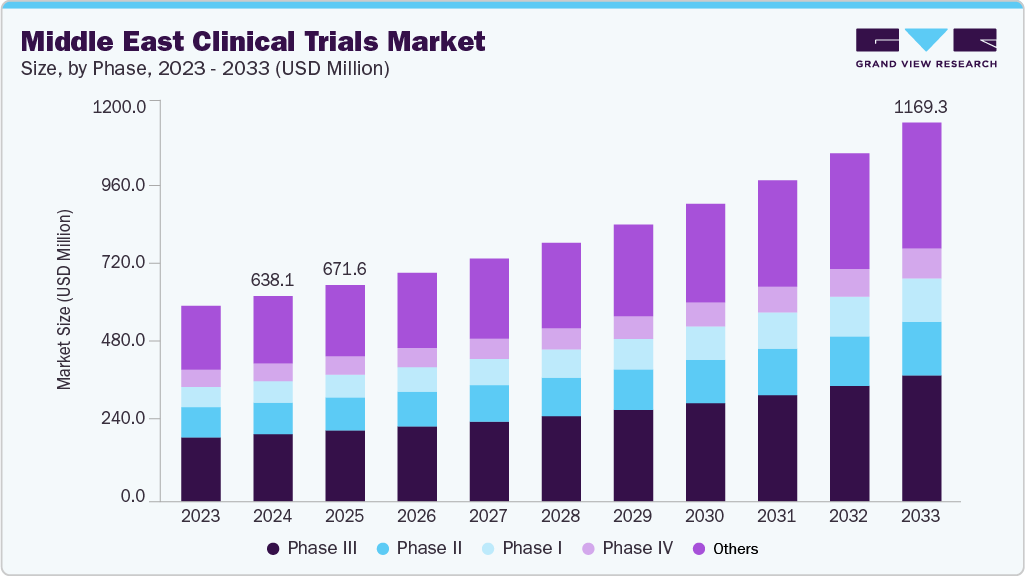
The Middle East clinical trials market size was estimated at USD 638.11 million in 2024 and is projected to reach USD 1,169.34 million by 2033, growing at a CAGR of 7.18% from 2025 to 2033. The Middle East faces a rising prevalence of chronic diseases such as diabetes, cardiovascular disorders, obesity, and cancer, creating strong demand for new therapies.
Key Market Trends & Insights
- Saudi Arabia clinical trials market held the largest share of 26.38% % of the market in 2024.
- By phase, the phase III segment held the largest market share of 48.70% in 2024.
- By study design, the interventional trials segment held the highest market share in 2024.
- By indication, the oncology segment held the largest revenue share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 638.11 Million
- 2033 Projected Market Size: USD 1,169.34 Million
- CAGR (2025-2033): 7.18%
Countries like Saudi Arabia, UAE, and Qatar report some of the world’s highest diabetes rates, making them prime locations for trials targeting metabolic and lifestyle-related conditions. According to the Telecommunications and Digital Government Regulatory Authority 2024, as of 2021, diabetes affected 11.8% of the UAE population. Reducing its prevalence remains a key performance indicator under the world-class healthcare pillar of the UAE National Agenda 2021. This growing disease burden drives pharmaceutical companies to establish local clinical studies to test the efficacy of novel drugs on region-specific populations. The urgent need for better therapeutic options also ensures government and healthcare sector support, fueling market expansion in the region.Middle Eastern governments are prioritizing research and innovation to strengthen healthcare systems and diversify economies. Saudi Arabia’s Vision 2030, UAE’s Centennial 2071, and Qatar’s National Health Strategy all emphasize clinical research, digital health, and partnerships with global pharma firms. Regulatory reforms are streamlining trial approvals, while infrastructure investments expand research centers and trial sites. Setting up clinical trials in the Middle East offers sponsors higher patient recruitment and stronger investigator engagement compared to traditional hubs. Conducting trials in regions with unmet clinical needs enhances access, optimizes efficiency, and supports innovation, especially when combined with AI and real-world data integration for regulatory submissions.
The adoption of reliance models and harmonized regulatory frameworks across the Gulf Cooperation Council (GCC) and Arab Regulators Network is a strong growth driver. Saudi Arabia’s SFDA and Qatar’s Ministry of Public Health are advancing regulatory reforms to align with global standards through reliance practices, digitalization, and collaboration. Efforts include streamlined approvals, pharmacovigilance, and AI integration. By referencing approvals from trusted agencies like the US FDA, EMA, or regional peers, Middle Eastern regulators accelerate the review and approval of clinical trials and new therapies. This harmonization reduces duplication, shortens approval timelines, and enhances confidence among international sponsors. Combined with digital regulatory platforms and cross-border collaborations, these practices strengthen efficiency and ensure high safety and quality standards, making the Middle East an increasingly attractive destination for global clinical research.
Public-private partnerships further attract foreign investment. For instance, in December 2024, Uncommon Cures partnered with UAE-based Carexso and PDC FZ LLC to accelerate orphan drug clinical trials in the Middle East. Combining expertise in rare disease drug development with regional infrastructure, this collaboration aimed to streamline regulatory pathways, expand access, and deliver innovative therapies to underserved rare disease patients. This supportive environment encourages both multinational and regional companies to conduct clinical trials, positioning the Middle East as an emerging hub for pharmaceutical innovation and life sciences research.
The Middle East region has seen rapid development in healthcare and research infrastructure, including modern hospitals, advanced laboratories, and specialized research institutes. Leading Contract Research Organizations (CROs) and local clinical research units are expanding operations in countries such as Saudi Arabia, the UAE, and Egypt, bringing global expertise and technology. The availability of electronic health records, biobanks, and genomic research initiatives further enhances trial efficiency. This robust infrastructure supports complex trials, from Phase I to Phase IV, while ensuring compliance with international standards. As a result, more pharmaceutical companies are choosing the Middle East for high-quality, large-scale clinical studies

R&D Pipeline: Some of the oncology drugs undergoing clinical trials in Saudi Arabia and UAE
NCT Number
Study Title
Study Status
Conditions
Interventions
Sponsor
NCT04529122
A Worldwide Cancer Registry Enrolling Participants Profiled With a Next-Generation Sequencing Test
Recruiting
Solid Tumors
NA
Hoffmann-La Roche
NCT06926868
A Study of Izalontamab Brengitecan Versus Chemotherapy in Participants With Previously Untreated, Locally Advanced, Recurrent Inoperable, or Metastatic Triple-negative Breast Cancer Ineligible for Anti-PD(L)1 Drugs (IZABRIGHT-Breast01)
Recruiting
Breast Neoplasms
DRUG: Iza-bren
DRUG: Nab-paclitaxel
DRUG: Paclitaxel
DRUG: Capecitabine
DRUG: Carboplatin
DRUG: Gemcitabine
Bristol-Myers Squibb
NCT05443126
A Study of EP0031 in Patients With Advanced RET-altered Malignancies
Recruiting
Advanced Solid Tumor
DRUG: EP0031
Ellipses Pharma
NCT05952557
An Adjuvant Endocrine-based Therapy Study of Camizestrant (AZD9833) in ER+/HER2- Early Breast Cancer (CAMBRIA-2)
Recruiting
Breast Cancer, Early Breast Cancer
DRUG: Camizestrant
DRUG: Tamoxifen
DRUG: Anastrozole
DRUG: Letrozole
DRUG: Exemestane
DRUG: Abemaciclib
AstraZeneca
NCT06013527
Physical Rehabilitation Program for Cardiorespiratory Health and Quality of Life Among Breast Cancer Survivors in UAE.
Recruiting
Breast Cancer
OTHER: Supervised Training
OTHER: Individually tailored home program
Gulf Medical University
Market Concentration & Characteristics
Market growth stage is moderate, and the pace of the market growth is accelerating. The Middle East clinical trials market is characterized by evolving technologies and therapeutics, regulatory considerations, growing need for advanced therapeutics, increasing demand for personalized medicine, and increasing outsourcing of development processes to leverage cost advantages and specialized capabilities.
The market is increasingly embracing innovation through decentralized trial models, digital platforms, and AI-driven analytics. Countries like Saudi Arabia and the UAE are piloting advanced technologies, while regional CROs are adopting wearable-based monitoring and e-consent solutions. In December 2024, Ellipses Pharma launched the UAE’s first large-scale Phase 1/2 oncology trial at the Cleveland Clinic and Tawam Hospital, evaluating its RET inhibitor EP0031/A400 for RET-altered lung and thyroid cancers. Innovation remains in early-to-mid adoption, but strong government support and global collaborations accelerate transformation.

Stringent quality protocols and regulatory norms by several nations to ensure patient safety and data integrity, which highly impact operational capabilities in the Middle East clinical trials market. Market participants with robust compliance measures and a track record of fulfilling regulatory standards gain credibility and preference from biopharmaceutical sponsors. Compliance with these regulations demands substantial resources, leading to barriers for smaller or newer firms.
Mergers and acquisition activities in this market are increasing and witness similar growth during the analysis timeframe. Several companies are acquiring development-stage companies to enhance the company’s service portfolio to cater large patient pool. In May 2024, Department of Health Abu Dhabi partnered with Roche Pharmaceuticals Middle East to advance real-world data research on spinal muscular atrophy and Duchenne Muscular Dystrophy. Moreover, these firms are integrating advanced facilities and forming strategic alliances to achieve synergies in capabilities and resources, enhancing their competitiveness.
The market comprises a large number of bio-pharmaceutical, medical device manufacturers and CROS specialized in drug development leading to a fragmented market scenario.
Pharmaceutical, biotech, medical device firms, and academic research centers are major consumers of clinical trial services. Their preferences, bargaining power, and concentration can significantly influence the market. Concentration among these end users might favor specific service providers or trial conductors.
Phase Insights
Phase III was the largest segment with a revenue share of 48.70% in 2024, due to its critical role in assessing drug safety and efficacy in large patient populations. Rising prevalence of chronic diseases, government-backed healthcare investments, and expanding patient recruitment capabilities in the UAE, Saudi Arabia, and Qatar are fueling demand for late-stage trials. Additionally, global pharmaceutical companies increasingly choose the region for Phase III studies, leveraging advanced infrastructure, strong regulatory frameworks, and strategic partnerships with leading hospitals and CROs.
Phase I is emerging as the fastest-growing segment in the Middle East clinical trials market, driven by the region’s focus on early-stage research and innovation. Governments in the UAE, Saudi Arabia, and Qatar are investing heavily in biotech hubs, precision medicine, and genomic studies, encouraging sponsors to initiate first-in-human trials locally. Rising collaborations with global pharmaceutical companies and CROs, coupled with improved regulatory efficiency and advanced clinical infrastructure, are fostering strong growth. This trend positions the region as a competitive hub for pioneering therapies.
Study Design Insights
Interventional trials represented the largest segment, accounting for 79.20% in 2024, supported by strong government initiatives, academic partnerships, and investments from global pharmaceutical companies. Countries such as Saudi Arabia, the UAE, and Qatar are prioritizing interventional studies to evaluate innovative drugs, biologics, and advanced therapies across oncology, rare diseases, and immunology. Robust regulatory frameworks, expanded research centers, and collaborations with CROs have accelerated the growth of this segment. With rising patient enrollment and adoption of precision medicine, interventional trials dominate the regional clinical trial landscape.
Observational trials are emerging as the fastest-growing segment in the Middle East clinical trials market, driven by rising demand for real-world evidence (RWE), regulatory emphasis on post-marketing surveillance, and the need to monitor long-term safety and effectiveness of therapies. Governments and healthcare authorities in the region are increasingly supporting data-driven research to improve treatment outcomes and inform policymaking. Collaborations between hospitals, CROs, and pharmaceutical companies are fueling this growth, with advancements in electronic health records (EHRs), AI-driven analytics, and biostatistics enhancing data accuracy and reliability in observational studies.
Indication Insights
Oncology represented both the largest and the fastest-growing segment in the Middle East clinical trials market, fueled by the region’s rising cancer burden, which includes high incidences of breast, lung, and colorectal cancers. Governments are prioritizing oncology research through investments in advanced cancer centers, national cancer control programs, and collaborations with global pharmaceutical firms. Precision medicine, immuno-oncology, and targeted therapies are key areas of focus, supported by regional adoption of innovative technologies such as AI-driven diagnostics and genomic profiling.
Major players like Roche, Novartis, AstraZeneca, and regional CROs are actively conducting oncology trials, positioning the Middle East as a hub for cancer research. Furthermore, partnerships between local health authorities and international firms are accelerating patient recruitment and trial execution. This strong clinical pipeline, along with increasing patient awareness and demand for novel treatments, ensures oncology’s dominance and rapid growth in the Middle East clinical trials market.
Indication By Study Design Insights
Oncology interventional trials held the largest share in the Middle East clinical trials market in 2024, driven by the region’s rising cancer prevalence and urgent demand for advanced therapies. Governments and healthcare authorities are actively supporting oncology research through specialized cancer centers, funding initiatives, and collaborations with leading global pharmaceutical companies.
Interventional trials in oncology focus on innovative areas such as immunotherapies, targeted therapies, and gene therapies, attracting strong industry and academic partnerships. There are approximately 100 clinical trials ongoing for oncology interventional trials.
Service Insights
Laboratory services represented the largest share in the Middle East clinical trials market, owing to their critical role in supporting drug development, biomarker analysis, genetic testing, and advanced diagnostics. The region’s rising clinical research activity, coupled with investments in modern laboratory infrastructure, has accelerated demand for high-quality testing and validation services. Leading global and regional laboratories collaborate with pharmaceutical companies and CROs to ensure accuracy, compliance, and faster turnaround. With advancements in molecular diagnostics and personalized medicine, laboratory services remain central to driving efficiency and credibility in Middle East clinical trials.
Patient recruitment is the fastest growing segment over the forecast period, driven by expanding trial diversity, growing disease burden, and improved digital engagement platforms. The region’s rising population, increased awareness, and supportive regulatory frameworks have enhanced enrollment efficiency. Partnerships between hospitals, CROs, and global pharma companies are streamlining recruitment through AI-driven tools, electronic health records, and real-world data integration. These innovations not only accelerate trial timelines but also improve patient diversity and retention. As demand for advanced therapies rises, patient recruitment continues to be a critical growth driver in the region’s clinical trials landscape.
Sponsor Insights
Pharmaceutical and biopharmaceutical companies represented the largest and fastest-growing segment in the Middle East clinical trials market, driven by increasing investments in drug development and expanding research infrastructure across the region. These companies are actively conducting trials to evaluate innovative therapies in areas such as oncology, cardiovascular diseases, and rare genetic disorders. The region’s growing patient pool, supportive regulatory frameworks, and rising healthcare expenditure have made it an attractive destination for clinical research.
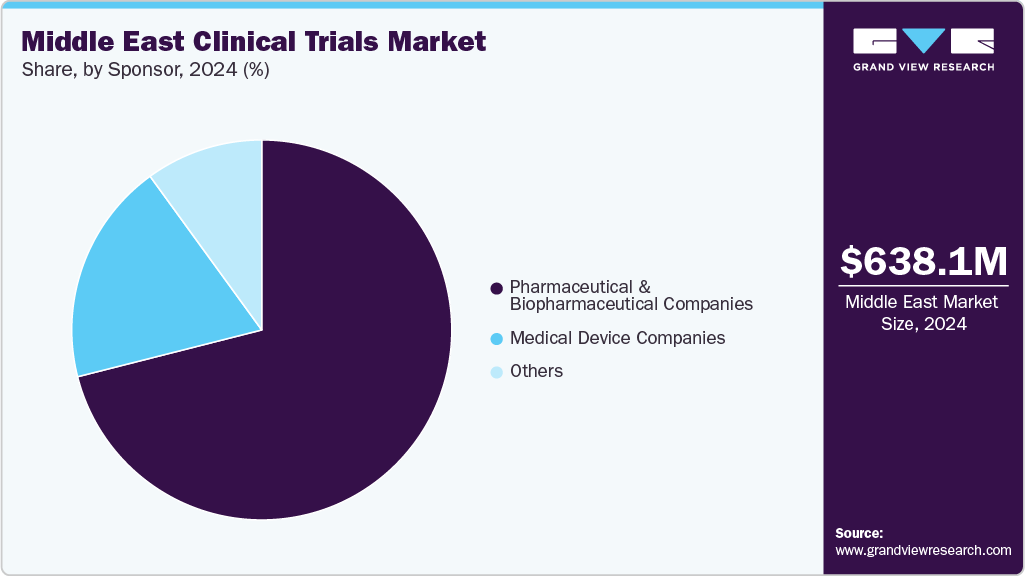
Multinational pharmaceutical firms are increasingly collaborating with local hospitals, contract research organizations (CROs), and academic institutions to accelerate trial timelines and ensure high-quality data generation. Biopharmaceutical innovations, including biologics, cell and gene therapies, and precision medicines, are fueling demand for region-specific clinical studies. This trend is further supported by government initiatives promoting clinical research and public-private partnerships, which collectively enhance the capabilities of the Middle East to host complex, large-scale trials, positioning pharmaceutical and biopharmaceutical companies at the forefront of market growth.
Country Insights
Middle East clinical trials market is rapidly growing, driven by rising chronic disease prevalence, supportive government initiatives, and regulatory alignment with global standards. Some of the players such as Roche, Novartis, and regional CROs like Carexso and PDC are strengthening their presence. Advancements in digital platforms, AI-driven data management, and cross-border regulatory harmonization are further accelerating market growth and patient access. In May 2024, Cellenkos Inc. and King Faisal Specialist Hospital & Research Centre collaborated to launch Saudi-U.S. clinical trials on Treg therapies for graft vs. host disease and aplastic anemia, advancing cell therapy infrastructure.
Saudi Arabia Clinical Trials Market Trends
Saudi Arabia’s clinical trials market held the largest revenue share of 26.38% in 2024, driven by Vision 2030, rising demand for innovative therapies, and strong government support. Saudi Food and Drug Authority (SFDA) is transforming Saudi Arabia’s clinical trials market through regulatory innovation, digitalization, and global collaborations. By advancing gene and cell therapies, medical device approvals and localized research. By the end of 2024, the SFDA approved around 149 medical device clinical trials, spanning implantable technologies and AI-driven digital tools. Major innovations include the Dose Check app for diabetes, INOVA for diabetic retinopathy, and Cardio-iSelfie for remote vital signs monitoring.
UAE Clinical Trials Market Trends
The UAE clinical trials marketis the fastest growing country over the forecast period, driven by strong government support, regulatory reforms, and world-class healthcare infrastructure. Key players like Cleveland Clinic Abu Dhabi, Burjeel Medical City, and CROs like IROS enhance trial execution. Strategic partnerships, digital health adoption, and investments in oncology, dermatology, and rare disease research are positioning the UAE as a regional hub for innovation. In May 2025, Medicus Pharma Ltd received UAE DOH approval to begin a Phase 2 study treating basal cell carcinoma non-invasively, enrolling 36 patients across four major hospitals, coordinated by IROS, an M42 company.
Kuwait Clinical Trials Market Trends
Kuwait’s clinical trials market is gradually expanding, driven by rising demand for advanced treatments, a strong burden of chronic diseases, and government healthcare investments. Key players include the Kuwait Cancer Control Center, Dasman Diabetes Institute, and global pharma firms partnering with local institutions. Advancements in digital health platforms, regulatory alignment, and regional collaborations are enhancing trial efficiency, patient recruitment, and research capacity.
Oman Clinical Trials Market Trends
Oman’s clinical trials market is gradually expanding, supported by government healthcare investments, rising prevalence of chronic diseases, and regulatory modernization. The Ministry of Health, Sultan Qaboos University Hospital, and international pharma firms drive activity. Advancements include digital health integration, improved research infrastructure, and collaborations with GCC partners, positioning Oman as an emerging hub for clinical research in the Middle East.
Qatar Clinical Trials Market Trends
Qatar’s clinical trials market is advancing, driven by investments under Qatar National Vision 2030, increasing chronic disease burden, and the government's focus on precision medicine. Key players include Hamad Medical Corporation, Sidra Medicine, and partnerships with global pharma companies. Advancements in genomics, digital health platforms, and biobank integration strengthen Qatar’s research ecosystem, positioning it as a competitive hub for regional clinical trials.
Key Middle East Clinical Trials Company Insights
Key players operating in the Middle East clinical trials companies market are undertaking various initiatives to strengthen their presence and increase the reach of their products and services. Strategies such as expansion activities and partnerships are key in propelling the market growth.
Key Middle East Clinical Trials Companies:
- Sanofi
- Novartis AG
- Pfizer
- GSK plc
- Novo Nordisk A/S
- Hikma Pharmaceuticals PLC
- Julphar
- SPIMACO
- Tabuk Pharmaceuticals
- F. Hoffmann-La Roche Ltd
- Johnson & Johnson
Recent Developments
-
In May 2025, Medicus Pharma received UAE DOH approval to begin a Phase 2 clinical trial for non-invasive BCC treatment across four hospitals, with IROS, a UAE-based CRO, coordinating the study.
-
In May 2025, KFSHRC and Cellenkos signed an MOU to launch Saudi-U.S. clinical trial collaborations, starting with Treg therapies for GVHD and aplastic anemia, while expanding into ALS, cardiovascular, and cell therapy infrastructure.
-
In May 2025, ImmunityBio, MISA, KFSHRC, and KAIMRC signed an MOU to launch the FDA-approved Cancer BioShield platform in Saudi Arabia, pioneering immune-restorative therapies and advancing NK and T-cell protection in cancer care.
-
In December 2025, Uncommon Cures partnered with Carexso and PDC in the UAE to accelerate orphan drug trials, streamline regulatory processes, and expand treatment options for rare disease patients across the Middle East.
Middle East Clinical Trials Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 671.57 million
Revenue forecast in 2033
USD 1,169.34 million
Growth rate
CAGR of 7.18% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Phase, study design, indication, indication by study design, service, sponsor, country
Regional scope
Middle East
Country scope
Saudi Arabia; UAE; Kuwait; Oman; Qatar
Key companies profiled
Sanofi; Novartis AG; Pfizer; GSK plc; Novo Nordisk A/S; Hikma Pharmaceuticals PLC; Julphar; SPIMACO; Tabuk Pharmaceuticals; F. Hoffmann-La Roche Ltd; Johnson & Johnson
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Middle East Clinical Trials Market Report Segmentation
This report forecasts revenue growth at regional and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the Middle East clinical trials market report based on phase, study design, indication, indication by study design, service, sponsor, and country:
-
Phase Outlook (Revenue, USD Million, 2021 - 2033)
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Study Design Outlook (Revenue, USD Million, 2021 - 2033)
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Autoimmune/Inflammation
-
Rheumatoid Arthritis
-
Multiple Sclerosis
-
Osteoarthritis
-
Irritable Bowel Syndrome (IBS)
-
Others
-
-
Pain Management
-
Chronic Pain
-
Acute Pain
-
-
Oncology
-
Blood Cancer
-
Solid Tumors
-
Others
-
-
CNS Conditions
-
Epilepsy
-
Parkinson's Disease (PD)
-
Huntington's Disease
-
Stroke
-
Traumatic Brain Injury (TBI)
-
Amyotrophic Lateral Sclerosis (ALS)
-
Muscle Regeneration
-
Others
-
-
Diabetes
-
Obesity
-
Cardiovascular Diseases
-
Others
-
-
Indication by Study Design Outlook (Revenue, USD Million, 2021 - 2033)
-
Autoimmune/Inflammation
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Pain Management
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Oncology
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
CNS Conditions
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Diabetes
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Obesity
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Cardiovascular Diseases
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Others
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Protocol Designing
-
Site Identification
-
Patient Recruitment
-
Laboratory Services
-
Analytical Testing Services
-
Clinical Trial Data Management Services
-
Others
-
-
Sponsor Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical & Biopharmaceutical Companies
-
Medical Device Companies
-
Others
-
-
Country Outlook (Revenue, USD Million, 2021 - 2033)
-
Middle East
-
Saudi Arabia
-
UAE
-
Kuwait
-
Oman
-
Qatar
-
-
Frequently Asked Questions About This Report
b. The Middle East clinical trials market size was estimated at USD 638.11 million in 2024 and is expected to reach USD 671.57 million in 2025.
b. The Middle East clinical trials market is expected to grow at a compound annual growth rate of 7.18% from 2025 to 2033 to reach USD 1,169.34 billion by 2033.
b. Phase III segment dominated the Middle East clinical trials market with a share of 48.70% in 2024. This is attributable to its critical role in assessing drug safety and efficacy in large patient populations.
b. Some key players operating in the Middle East clinical trials market include Sanofi, Novartis AG, Pfizer, GSK plc, Novo Nordisk A/S, Hikma Pharmaceuticals PLC, Julphar, SPIMACO, Tabuk Pharmaceuticals, F. Hoffmann-La Roche Ltd, Johnson & Johnson
b. Key factors that are driving the market growth include increasing government initiatives to strengthen healthcare research infrastructure, growing investments from multinational pharmaceutical and medical device companies, and rising prevalence of chronic diseases such as diabetes, cardiovascular disorders, and cancer that create strong demand for innovative therapies.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










