- Home
- »
- Plastics, Polymers & Resins
- »
-
U.S. Healthcare Cold Chain Packaging Market Report, 2033GVR Report cover
![U.S. Healthcare Cold Chain Packaging Market Size, Share & Trends Report]()
U.S. Healthcare Cold Chain Packaging Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Labels & Sensors, Insulated Containers), By Packaging Format, By Temperature Requirement, By Application, And Segment Forecasts
- Report ID: GVR-4-68040-841-3
- Number of Report Pages: 85
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Bulk Chemicals
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
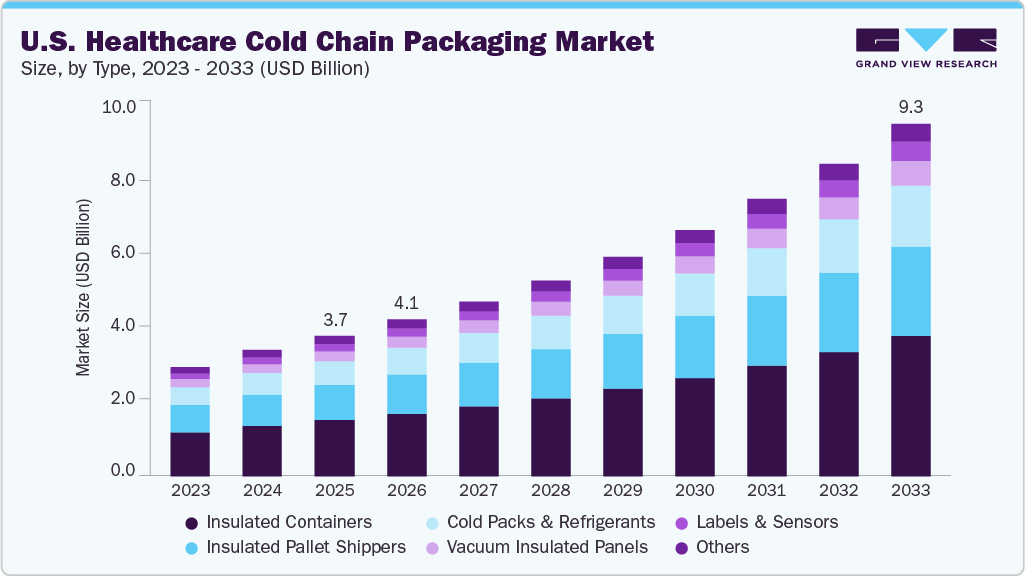
The U.S. healthcare cold chain packaging market size was estimated at USD 3.72 billion in 2025 and is projected to reach USD 9.32 billion by 2033, growing at a CAGR of 12.2% from 2026 to 2033. This is attributed to the increasing development and distribution of temperature-sensitive drugs, especially biologics, vaccines, cell and gene therapies, and advanced specialty medicines.
Key Market Trends & Insights
- By product, the insulated containers segment dominated the market with the highest revenue share of 40.1% in 2025
- The labels & sensors segment is forecasted to grow at the highest CAGR of 13.0% from 2026 to 2033.
- Based on packaging format, active cold chain packaging is forecasted to grow at the highest CAGR over the coming years.
- Based on temperature requirement, the Cryogenic (<-80°C) temperature segment is expected to grow at the highest CAGR of 12.9% from 2026 to 2033.
- Based on application, biopharmaceutical companies is forecasted to grow at the highest CAGR of 12.8% over the forecast period.
Market Size & Forecast
- 2025 Market Size: USD 3.72 Billion
- 2033 Projected Market Size: USD 9.32 Billion
- CAGR (2026 - 2033): 12.2%
These products often require precise thermal conditions throughout storage and transportation to retain potency and efficacy. For example, mRNA-based vaccines and many monoclonal antibodies lose effectiveness if exposed to even minor temperature variations, necessitating high-performance insulated packaging and ultracold solutions.
As pharmaceutical companies continue to shift focus from traditional small-molecule drugs toward biologics and personalized medicines, the reliance on robust cold chain packaging has surged. The demand for cold chain packaging in the U.S. healthcare market is expanding steadily, driven by structural shifts in pharmaceutical innovation, healthcare delivery models, regulatory oversight, and supply-chain risk management. Unlike earlier periods when cold chain requirements were limited to a narrow set of vaccines and biologics, temperature control has now become a foundational requirement across a broad and growing segment of healthcare products.
A primary factor driving the growth is the rapid increase in temperature-sensitive pharmaceuticals, particularly biologics, specialty drugs, vaccines, and advanced therapies such as cell and gene treatments. These products are inherently unstable outside narrow temperature ranges and can lose potency or become unusable if exposed to even brief temperature excursions. The U.S. pharmaceutical pipeline is increasingly dominated by biologics rather than traditional small-molecule drugs, which significantly raises the volume of products requiring refrigerated, frozen, or ultra-cold conditions. As a result, pharmaceutical manufacturers are investing heavily in validated cold chain packaging solutions that can ensure thermal stability across long and complex distribution routes.
Closely linked to this trend is the strong growth in biopharmaceutical research, clinical trials, and commercialization activity within the U.S. The country hosts a large share of global clinical trials, many of which involve investigational drugs that must be transported between manufacturing sites, trial centers, laboratories, and storage facilities under strict temperature controls. These shipments often span long distances and multiple handovers, increasing the risk of temperature deviation. Consequently, demand is rising for high-performance insulated packaging, phase-change materials, and qualified shipping systems that can maintain temperature integrity throughout the trial lifecycle.
Industry Characteristics
The U.S. healthcare cold chain packaging industry exhibits a moderate to high level of concentration, particularly at the premium and validated-solutions end of the market. A relatively small group of established players dominates large pharmaceutical contracts due to their validated product portfolios, regulatory compliance capabilities, and long-standing relationships with drug manufacturers and logistics providers. However, the market still accommodates a fragmented tier of regional and niche players serving localized healthcare providers, clinical trials, and specialty logistics needs. Entry barriers are higher in pharmaceutical-grade packaging compared to food or industrial cold chain applications, reinforcing concentration among experienced suppliers.
The degree of innovation in the industry is high and accelerating, driven by the increasing complexity of temperature-sensitive therapies. Innovation focuses on advanced insulation materials, phase-change materials (PCMs), reusable and hybrid packaging systems, and smart packaging technologies such as real-time temperature monitoring and data logging. Sustainability has also emerged as a key innovation theme, with companies developing recyclable, bio-based, and reusable cold chain solutions to meet ESG targets. Product differentiation increasingly depends on performance validation, duration control, and digital integration rather than basic insulation alone.

Regulatory influence on the industry is high and structural. Compliance with FDA guidelines, Good Manufacturing Practices (GMP), and Good Distribution Practices (GDP) significantly shapes product design, validation processes, and supplier selection. Cold chain packaging must demonstrate consistent thermal performance and traceability, which increases development costs and favors suppliers with established validation expertise. Regulatory scrutiny reduces the viability of low-cost or untested solutions, reinforcing quality-driven competition and long-term supplier relationships.
Product Insights
The insulated containers segment recorded the largest market revenue share of over 40.1% in 2025. Demand for insulated containers is the single largest driver in the U.S. market because they serve as the primary shipment vehicle for a wide range of temperature-sensitive healthcare products. Their versatility across Controlled Room Temperature (CRT), refrigerated (2°C-8°C), and frozen (-20°C to -40°C) applications makes them indispensable for pharmaceutical manufacturers, clinical research organizations (CROs), hospitals, specialty pharmacies, and logistic providers. Unlike pallet shippers that are used for bulk movements, insulated containers are optimized for parcel, last-mile delivery, and mid-mile transfer, accommodating the rising volume of direct-to-patient shipments driven by e-commerce, specialty drug distribution, and home healthcare services.
The demand for insulated pallet shippers is growing steadily due to their critical role in high-volume, long-haul, and bulk distribution of temperature-sensitive pharmaceuticals. Unlike containers intended for parcel and last-mile deliveries, pallet shippers are designed to protect large quantities of product, such as vaccines, biologics, and active pharmaceutical ingredients (APIs), through long domestic and international supply chains. They combine robust insulation with advanced refrigerants or active cooling elements to maintain precise temperature zones throughout extensive transit times, making them essential for centralized manufacturing and wide geographic distribution.
The demand for labels and sensors is expanding at the fastest rate of any segment in the U.S. market due to growing requirements for real-time visibility, compliance documentation, and risk management. This category includes temperature indicators, data loggers, RFID tags, Bluetooth-enabled trackers, and IoT-connected sensors that continuously monitor conditions throughout the shipment lifecycle. As regulatory scrutiny intensifies, particularly for biologics, vaccines, and high-value therapies, pharmaceutical companies, logistics providers, and healthcare end users increasingly mandate robust temperature monitoring to demonstrate chain-of-custody, maintain audit trails, and support quality assurance programs.
Packaging Format Insights
Based on packaging format, passive cold chain packaging led the U.S. market with a revenue share of 59.8% in 2025. Passive cold chain packaging continues to experience strong and sustained growth due to its cost efficiency, flexibility, and scalability across a wide range of healthcare distribution scenarios. Passive systems rely on insulation materials, such as EPS, PUR, or vacuum insulated panels (VIPs), combined with refrigerants like gel packs, phase change materials (PCMs), or dry ice to maintain target temperatures without external power. These solutions dominate parcel, last-mile, clinical trial, and direct-to-patient shipments, which account for the majority of healthcare shipment volumes in the U.S.
The demand for active cold chain packaging is increasing in the U.S. healthcare sector as pharmaceutical supply chains handle a rising volume of high-value, highly temperature-sensitive products over long distances and extended transit durations. Active systems, such as incorporating powered cooling or heating units, compressors, batteries, and precise temperature control mechanisms, are essential for maintaining narrow temperature tolerances during international shipments, long-haul domestic transport, and complex multi-modal logistics. As U.S. pharmaceutical manufacturers expand global sourcing and distribution networks, active packaging is increasingly used to reduce the risks associated with long transit times, customs delays, and exposure to extreme ambient conditions.
Temperature Requirement Insights
Based on temperature requirement, the refrigerated cold chain packaging segment dominated the segment with a major revenue share of 41.9% in 2025. The refrigerated 2°C-8°C segment represents a core growth driver for the U.S. market, largely due to the rising dominance of biologics, vaccines, and injectable therapies. These products exhibit narrow stability windows and are highly susceptible to degradation outside refrigerated conditions, making validated cold chain packaging essential. As biologics continue to account for a growing share of U.S. healthcare revenues, demand for insulated containers, gel packs, phase-change materials, and pallet-level refrigerated shippers has increased significantly.
This growth is further supported by the expanding distribution of vaccines, specialty drugs, and hospital-administered therapies, all of which require reliable temperature maintenance across multi-tier supply chains. Hospitals, pharmacies, clinical sites, and logistics providers depend on refrigerated packaging solutions that can maintain consistent temperatures across long-distance transportation and diverse climatic zones. Heightened regulatory scrutiny, combined with the high financial and clinical consequences of temperature excursions, has positioned refrigerated cold chain packaging as a critical risk-mitigation tool across the U.S. healthcare system.
Application Insights
The biopharmaceutical companies segment recorded the largest market share of over 37.7% in 2025 and is projected to grow at the fastest CAGR of 12.8% during the forecast period. The demand for cold chain packaging among biopharmaceutical companies is expanding rapidly due to the structural shift of the U.S. drug pipeline toward biologics, vaccines, and advanced therapies. Unlike small-molecule drugs, biologics such as monoclonal antibodies, recombinant proteins, and peptide-based injectables are highly sensitive to temperature excursions, requiring tightly controlled environments, mainly 2°C-8°C, and increasingly frozen or cryogenic conditions. As biologics account for a growing share of FDA approvals and revenue generation, manufacturers need to invest in reliable cold chain packaging to preserve product stability from manufacturing sites through distribution.
In addition, next-generation therapies such as cell and gene therapies, mRNA-based products, and personalized medicines have significantly elevated cold chain complexity. Many of these products require ultra-low or cryogenic temperatures, often for extended durations and across long transportation routes. This has intensified demand for high-performance insulated containers, dry ice solutions, vacuum insulated panels, and temperature-monitoring systems that can ensure compliance with stringent handling requirements.
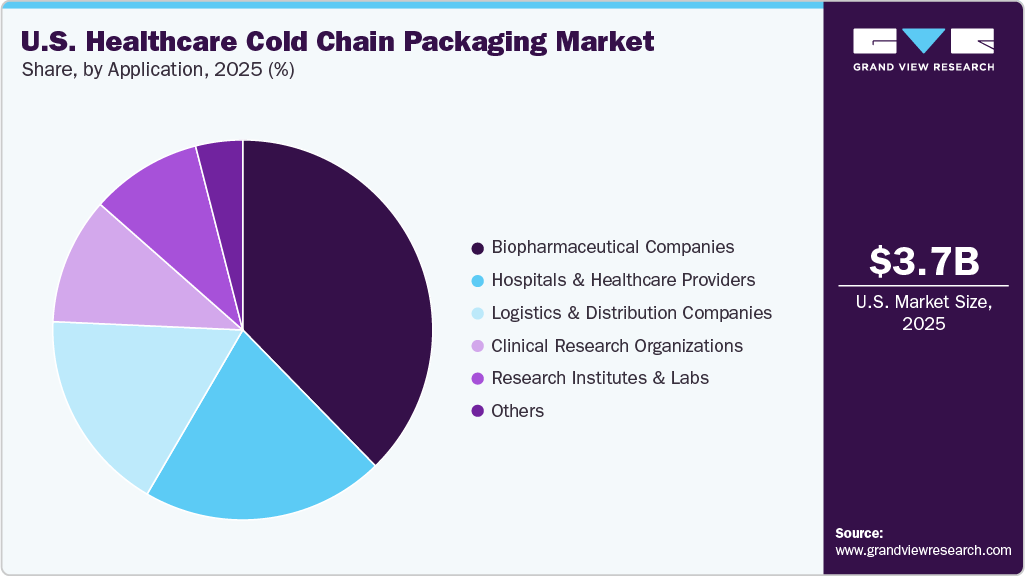
In the healthcare cold chain packaging industry, hospitals & healthcare providers represent a critical end use segment due to their high dependency on temperature-sensitive pharmaceuticals, biologics, and vaccines. Hospitals routinely receive and store a variety of medical products that require stringent temperature control, including insulin, monoclonal antibodies, oncology drugs, blood components, and mRNA-based vaccines. To maintain the efficacy and safety of these products, hospitals demand robust cold chain packaging solutions that ensure product stability during transit and in-hospital handling. This reliance makes hospitals not only large-volume consumers of cold chain packaging but also key stakeholders in pushing for innovations in temperature-controlled packaging technologies.
Key U.S. Healthcare Cold Chain Packaging Company Insights
The competitive environment of the U.S. market is intense, technology-driven, and quality-focused, shaped by regulatory compliance, product performance requirements, and long-term customer relationships. Competition extends beyond pricing to include validation expertise, thermal performance reliability, service capabilities, and the ability to support complex pharmaceutical distribution models.
Competition in the market is primarily non-price-based, particularly in pharmaceutical and biopharmaceutical applications. Suppliers compete on packaging performance, duration of temperature control, regulatory validation, and reliability under real-world distribution conditions. For high-value and temperature-sensitive therapies, pharmaceutical companies prioritize risk mitigation and compliance over cost savings, allowing established players with proven track records to maintain strong competitive positions.
-
In November 2025, Peli BioThermal LLC opened an expanded service center in Allentown, Pennsylvania, to increase throughput for reusable and on-demand single-use shippers. The upgrade expands capacity for refurbishment, testing, and Crēdo on-demand programs, shortening lead times for U.S. customers and strengthening local availability for pharmaceutical and biotech cold shipments.
-
In July 2025, Sonoco Products Company completed the sale of its ThermoSafe temperature-assured packaging business to private equity firm Arsenal Capital Partners. The divestiture refocuses Sonoco on core metal and fiber packaging, while ThermoSafe will operate under new ownership to accelerate investment in specialized pharma shippers and service capabilities relevant to U.S. life sciences customers.
Key U.S. Healthcare Cold Chain Packaging Companies:
- ThermoSafe Technologies Inc.
- Cryoport Systems, LLC
- Sealed Air Corporation
- Peli BioThermal LLC
- CSafe
- DGP Intelsius LLC
U.S. Healthcare Cold Chain Packaging Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 4.15 billion
Revenue forecast in 2033
USD 9.32 billion
Growth rate
CAGR of 12.2% from 2026 to 2033
Historical data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, competitive landscape, growth factors, and trends
Segments covered
Product, temperature requirement, packaging format, application
Key companies profiled
ThermoSafe Technologies Inc.; Cryoport Systems, LLC; Sealed Air Corporation; Peli BioThermal LLC; CSafe; DGP Intelsius LLC
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional, and segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Healthcare Cold Chain Packaging Market Report Segmentation
This report forecasts revenue growth at a country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. healthcare cold chain packaging market report based on product, temperature requirement, packaging format, and application:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Insulated Pallet Shippers
-
Insulated Containers
-
Vacuum Insulated Panels
-
Cold Packs & Refrigerants
-
Labels & Sensors
-
Others
-
-
Temperature Requirement Outlook (Revenue, USD Million, 2021 - 2033)
-
Controlled Room Temperature (CRT)
-
Refrigerated (2°C-8°C)
-
Frozen (-20°C to -40°C)
-
Cryogenic (<-80°C)
-
-
Packaging Format Outlook (Revenue, USD Million, 2021 - 2033)
-
Active
-
Passive
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Biopharmaceutical Companies
-
Clinical Research Organizations
-
Hospitals and Healthcare Providers
-
Research Institutes & Labs
-
Logistics and Distribution Companies
-
Others
-
Frequently Asked Questions About This Report
b. The U.S. healthcare cold chain packaging market is expected to grow at a compound annual growth rate of 12.2% from 2026 to 2033 to reach USD 9.32 billion by 2033.
b. The market is driven by increasing development and distribution of temperature-sensitive drugs, especially biologics, vaccines, cell and gene therapies, and advanced specialty medicines.
b. The U.S. healthcare cold chain packaging market size was estimated at USD 3.72 billion in 2025 and is expected to reach USD 4.15 billion in 2026
b. Based on temperature requirement, refrigerated cold chain packaging segment dominated the segment with a major revenue share of 41.9% in 2025. The refrigerated 2°C–8°C segment represents a core growth driver for the U.S. healthcare cold chain packaging market, largely due to the rising dominance of biologics, vaccines, and injectable therapies. These products exhibit narrow stability windows and are highly susceptible to degradation outside refrigerated conditions, making validated cold chain packaging essential.
b. Some of the key players operating in the U.S. healthcare cold chain packaging market include ThermoSafe Technologies Inc., Cryoport Systems, LLC, Sealed Air Corporation, Peli BioThermal LLC, CSafe, and DGP Intelsius LLC
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










