- Home
- »
- Healthcare IT
- »
-
AI In Regulatory Affairs Market Size, Industry Report, 2033GVR Report cover
![AI In Regulatory Affairs Market Size, Share & Trends Report]()
AI In Regulatory Affairs Market (2025 - 2033) Size, Share & Trends Analysis Report By Component (Software/Platforms, Services), By Deployment Mode (Cloud-based, On-premises), By Application (Regulatory Intelligence, Dossier Management, Document Management), By End-use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-769-4
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
AI In Regulatory Affairs Market Summary
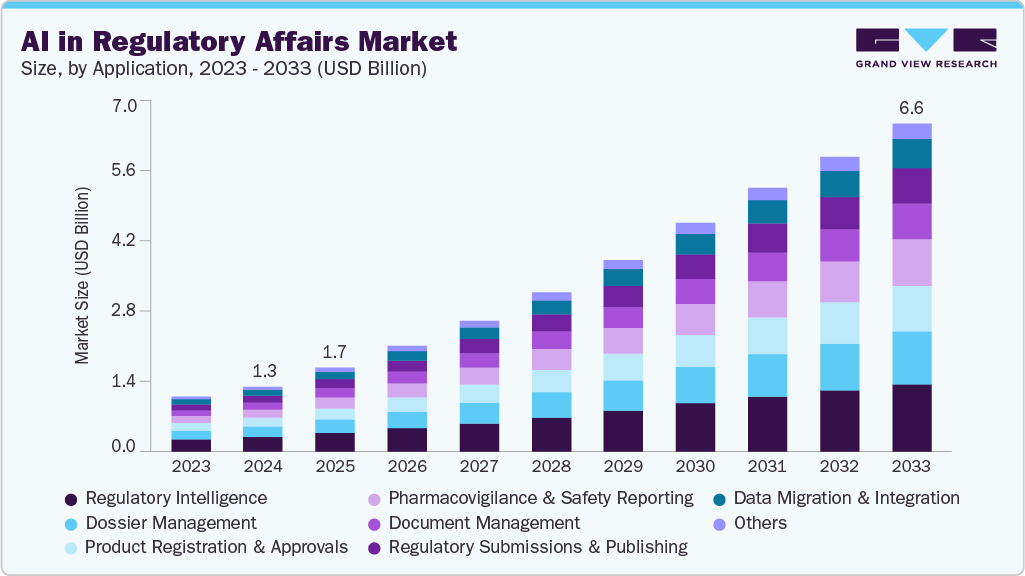
The global AI in regulatory affairs market size was estimated at USD 1.31 billion in 2024 and is projected to reach USD 6.65 billion by 2033, growing at a CAGR of 18.60% from 2025 to 2033. The growing complexity of regulatory submissions and rising cost pressures in drug development and regulatory operations are significant factors contributing to market growth.
Key Market Trends & Insights
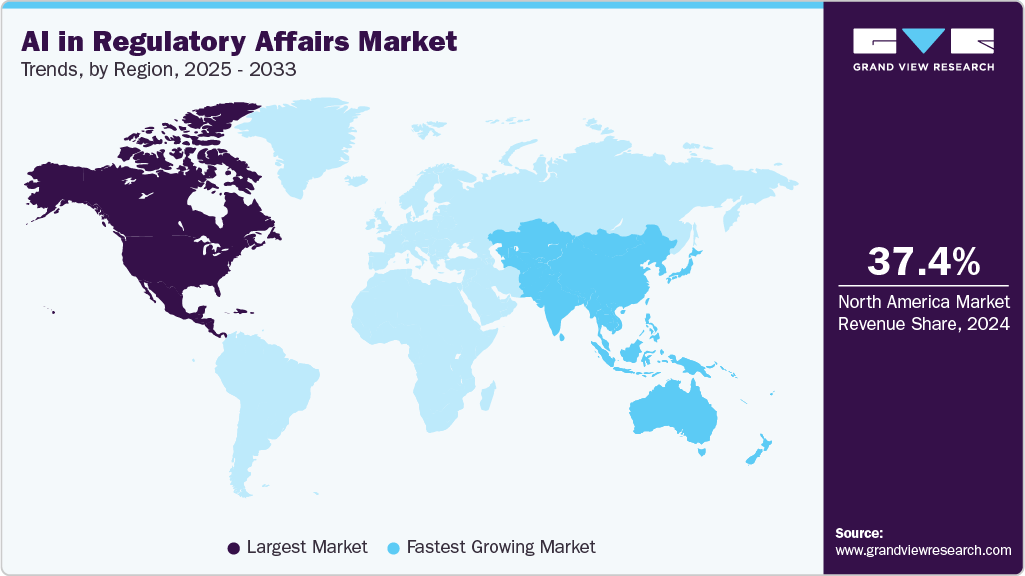
- The North America AI in regulatory affairs market held the largest revenue share in 2024.
- In the U.S., AI adoption in regulatory affairs benefits from advanced technological infrastructure and a robust life sciences ecosystem.
- By component, the software/platforms led the AI in regulatory affairs market with the largest market share of 68.12% in 2024.
- By deployment mode, the cloud-based segment accounted for the largest revenue share of over 58.01% in 2024.
- By application, the regulatory intelligence segment accounted for the largest revenue share of 22.00% in 2024.
Market Size & Forecast
- 2024 Market Size: USD 1.31 Billion
- 2033 Projected Market Size: USD 6.65 Billion
- CAGR (2025-2033): 18.60%
- North America: Largest market in 2024
- Asia Pacific: Fastest growing market
In addition, the rising cost pressures in drug development and regulatory operations, and technological advancements are some other factors fueling market growth further. The increasing complexity and volume of global regulatory requirements across the pharmaceutical, biotechnology, and medical device sectors drive the AI in regulatory affairs industry. AI platforms reduce time and labor costs by automating routine tasks such as data extraction, document validation, and submission formatting. Predictive analytics allow regulatory teams to forecast submission timelines and optimize resource allocation. Artificial intelligence (AI) also supports regulatory strategy planning, enabling faster approvals and market entry. For instance, in March 2025, ArisGlobal launched LifeSphere Unify, an R&D compliance platform harmonizing safety, medical affairs, regulatory, and quality data for efficiency and compliance.
Another key driver is the rising demand for real-time regulatory intelligence and decision support as the industry faces rapid innovation in drug development, including advanced therapies, biologics, and personalized medicine. AI-powered platforms gather actionable insights from large and diverse datasets, helping regulatory professionals anticipate trends and respond more efficiently. In addition, outsourcing regulatory affairs functions to AI-enabled service providers is growing, offering cost-effective and scalable compliance solutions. For instance, in September 2025, Parexel partnered with Weave Bio to accelerate regulatory submission processes and speed time to market for new therapies. Parexel is Weave’s CRO design partner, leveraging regulatory expertise to enhance Weave’s AI platform.

Furthermore, stringent regulatory frameworks, including those from the FDA, EMA, and other regional agencies, are compelling organizations to maintain continuous compliance with increasing scrutiny on data integrity, pharmacovigilance, and product lifecycle management. AI supports robust documentation, traceability, and audit readiness, mitigating risks of non-compliance and costly penalties. In addition, the growing integration of regulatory information management (RIM) systems with AI-driven automation further enables harmonized regulatory processes across multiple jurisdictions. Enhancements in natural language processing and generative AI also facilitate faster dossier preparation and regulatory writing, reducing cycle times and resource burdens.
Market Concentration & Characteristics
The chart below illustrates the relationship between industry concentration, characteristics, and participants. The x-axis represents the level of industry concentration, ranging from low to high. The y-axis represents various industry characteristics, including industry competition, level of partnerships & collaboration activities, degree of innovation, impact of regulations, and regional expansion. The market is slightly consolidated. However, several emerging players are entering the market, thereby contributing to increased fragmentation within the market. The degree of innovation is high. The level of merger & acquisition activities is moderate. Moreover, the impact of regulations and the regional expansion of industry is high.
The AI in regulatory affairs market is characterized by constant innovation, with a strong focus on launching new platforms to leverage administrative tasks. Prominent players are launching advanced solutions to sustain a competitive advantage. In addition, growing regulatory approvals propel market growth further. For instance, in July 2025, RegASK launched the industry's first agentic AI architecture for regulatory affairs, combining a vertical large language model (V-LLM) with specialized AI agents to deliver personalized insights and automate multi-step compliance workflows. Features include document retrieval, translation, summarization, and assessment generation, coordinated by a project manager agent and validated by an evaluator agent.
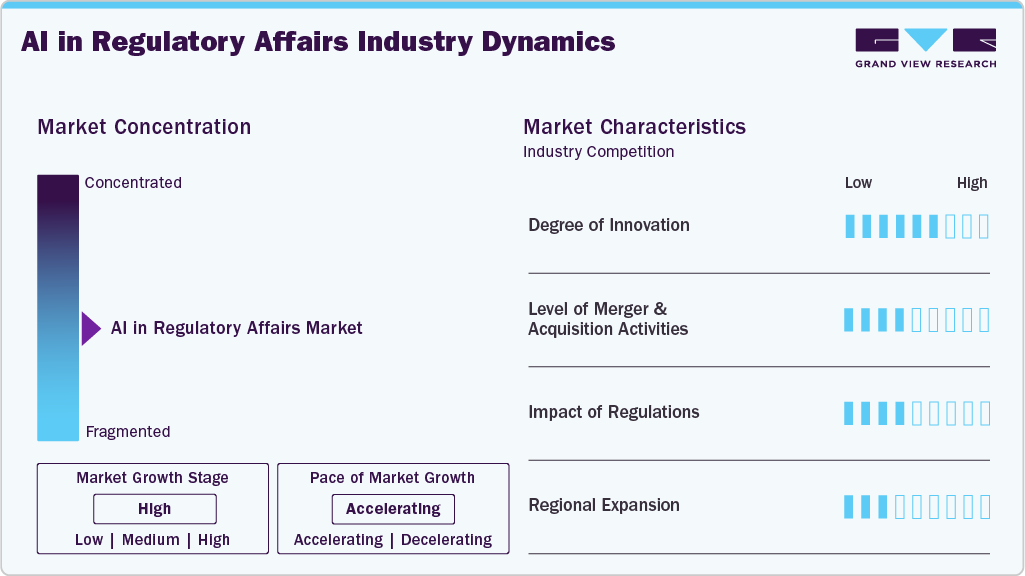
The AI in regulatory affairs industry is experiencing a moderate level of merger and acquisition activities undertaken by several key players. This is due to the desire to gain a competitive advantage in the industry, enhance technological capabilities, and consolidate in a rapidly growing market.
The market is influenced by global guidelines on data integrity, electronic submissions, and software as a medical device (SaMD). Agencies such as the U.S. FDA, EMA, PMDA, and Health Canada provide standards for AI-driven tools used in dossier management, pharmacovigilance, and regulatory intelligence. Compliance with Good Regulatory Practices (GxP) and 21 CFR Part 11 ensures data security, traceability, and audit readiness. AI platforms must undergo validation and maintain transparency in decision-making processes. Additionally, region-specific requirements govern cloud deployment, cross-border data transfer, and AI algorithm accountability.
Companies within the AI in regulatory affairs industry seek geographic expansion strategies to maintain their foothold in emerging markets and attract customers from these regions. Many service providers are expanding into emerging markets to support companies entering those regions and to serve local firms seeking global approvals.
Case Study Insights: Accelerating Regulatory Submission Preparation Using AI - A MedTech Leader's Success with RegDesk
A regulatory leader from an image-guided therapeutic business unit of a major medtech company faced significant challenges with slow submission preparation, which limited the team's capacity and risked missing aggressive business goals. The manual process, stretched across multiple global markets, combined with regulatory transitions such as MDR, compounded delays and cost inefficiencies. Seeking innovative solutions, the leader adopted RegDesk's AI-driven submission generation platform to streamline workflows globally.

This case illustrates the transformative impact of AI-powered regulatory platforms in optimizing global compliance workflows, reducing costs, and accelerating market access.
Component Insights
The software/platforms led the AI in regulatory affairs market with the largest market share of 68.12% in 2024. These platforms centralize regulatory information management, enabling seamless tracking, documentation, and submission lifecycle management. AI-enabled platforms integrate machine learning and natural language processing to streamline dossier preparation, automate eCTD submissions, and monitor real-time regulatory intelligence. The user-friendly interfaces and rapid implementation timelines drive adoption among pharmaceutical, biotechnology, and medical device firms. Thus, these platforms provide measurable ROI by optimizing resource allocation, improving submission quality, and expediting market access.
The services segment is expected to grow at the fastest CAGR during the forecast period. Pharmaceutical and biotechnology companies face growing challenges of regulatory compliance, submission management, and post-market surveillance, thereby increasing the demand for outsourcing to expert service providers employing AI-powered tools. Furthermore, vendors are enhancing their offerings with AI-powered regulatory intelligence, cloud migration services, and workflow optimization support. This shift toward outcome-based service models positions the segment for sustained high growth.
Deployment Mode Insights
The cloud-based segment accounted for the largest revenue share of over 58.01% in 2024 due to its scalability, flexibility, and cost-efficiency. Cloud platforms enable regulatory teams to access real-time AI-powered tools and regulatory intelligence from multiple geographic locations, supporting global compliance requirements. The flexibility to rapidly scale computational resources and storage without heavy upfront infrastructure investment attracts pharmaceutical and biotechnology companies. Cloud solutions also facilitate seamless integration with enterprise systems such as regulatory information management (RIM), allowing centralized policy enforcement and audit trail maintenance. With enhanced cybersecurity protocols and continuous platform updates, cloud deployments effectively address regulatory concerns around data privacy and system validation.
Moreover, the web-based segment of the AI in regulatory affairs industry is anticipated to experience steady growth over the forecast period. Web-based platforms allow regulatory teams to access AI-driven tools directly through browsers without extensive IT infrastructure, reducing setup time and upfront costs. This accessibility makes them attractive for small and mid-sized pharmaceutical, biotech, and medical device companies that may not require full-scale cloud solutions.
Application Insights
The regulatory intelligence segment accounted for the largest revenue share of 22.00% in 2024. AI-powered regulatory intelligence platforms automate the continuous surveillance and analysis of regulatory databases, health authority updates, and industry publications, providing timely and actionable insights to regulatory professionals. These platforms utilize natural language processing to extract and interpret critical information from vast volumes of unstructured regulatory documents, significantly reducing manual review time and minimizing human errors.
The pharmacovigilance & safety reporting segment is expected to grow at the fastest CAGR during the forecast period, due to increasing regulatory scrutiny and the rising volume of adverse drug reaction (ADR) data. Growing adoption of AI-driven pharmacovigilance platforms enhances real-time monitoring and predictive analytics, enabling proactive risk management and faster response to safety concerns. In addition, expanding clinical trials and accelerated drug approvals generate higher volumes of safety data, further fueling segment growth.
End-use Insights
The pharmaceutical companies segment held the largest market share of 39.84% in 2024. These companies lead in drug development, clinical trials, and product lifecycle management, necessitating robust AI-powered regulatory solutions to manage vast volumes of submissions and evolving regulations. The significant investment in research and development, including innovative biologics and personalized therapies, drives increased demand for AI tools that streamline regulatory intelligence, dossier management, and pharmacovigilance. In addition, pharmaceutical companies also invest heavily in digital transformation and AI adoption to improve operational efficiency, ensure data integrity, and minimize the risk of regulatory non-compliance. Their capacity to deploy scalable AI solutions across multiple regions further reinforces their dominance in the market.
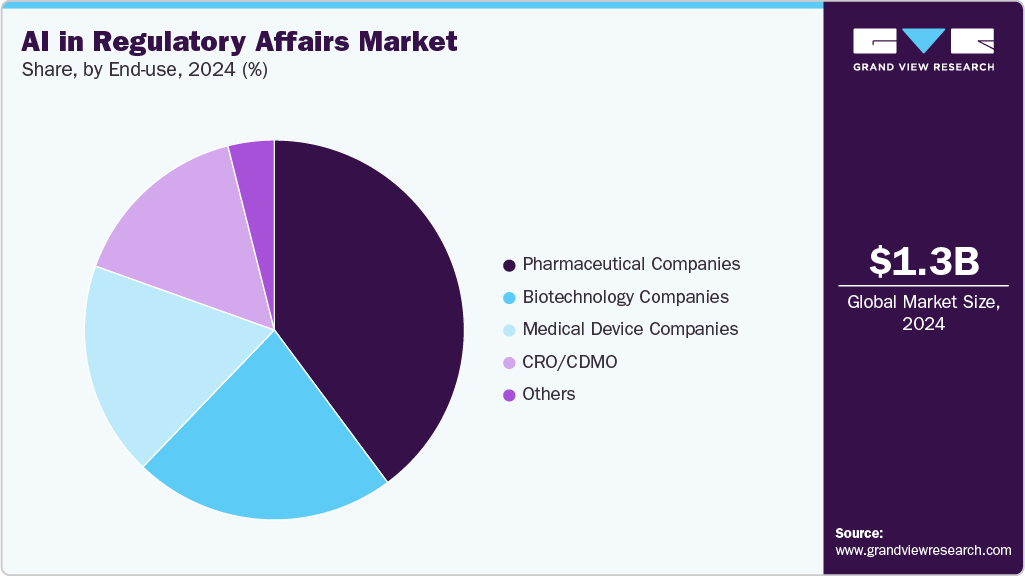
The CRO/CDMO segment is anticipated to grow at the fastest CAGR from 2025 to 2033. Contract Research Organizations (CROs) and Contract Development and Manufacturing Organizations (CDMOs) are increasingly integrating AI-enabled platforms into their operations for regulatory information management, submission automation, and pharmacovigilance services. This strategic adoption facilitates the development of scalable, multi-client solutions that enhance efficiency, accuracy, and compliance across different regions.
Regional Insights
The North America AI in regulatory affairs market held the largest revenue share of 37.45% in 2024. The complexity and volume of regulatory requirements from agencies such as the U.S. FDA and Health Canada propel market growth. Moreover, the rising number of clinical trials and personalized medicine development necessitate adaptable, scalable regulatory solutions. Integration of AI and data analytics improves real-time monitoring and compliance management. In addition, the demand for cost-effective outsourcing of regulatory functions further accelerates market growth.
U.S. AI In Regulatory Affairs Market Trends
In the U.S., AI adoption in regulatory affairs benefits from advanced technological infrastructure and a robust life sciences ecosystem. The FDA’s evolving guidance on AI/ML-enabled medical devices drives demand for intelligent regulatory submission and risk assessment tools. In addition, the growing focus on expedited approval pathways for biologics and gene therapies is further driving the adoption of AI. For instance, in September 2025, Elsevier launched PharmaPendium AI, a generative AI assistant for regulatory intelligence in drug development. It accelerates access to FDA and EMA regulatory documents, providing citation-backed answers to complex regulatory questions.
Europe AI In Regulatory Affairs Market Trends
The Europe AI in regulatory affairs industry is expected to witness significant growth during the forecast period. This growth is attributed to the stringent regulations, such as the EU MDR and IVDR, requiring sophisticated documentation and post-market surveillance. Growing pharmaceutical innovation and increasing cost pressures drive the outsourcing of regulatory functions supported by AI tools. In addition, the increasing complexity of cross-border submissions under the EU Clinical Trial Regulation (CTR) fosters demand for AI-enabled platforms.
The UK AI In regulatory affairs market is expected to grow over the forecast period. The UK market benefits from the Medicines and Healthcare Products Regulatory Agency (MHRA) initiatives, including pilot programs to integrate AI in regulatory evaluations. Post-Brexit, companies need AI-driven systems to manage dual EU and UK regulatory submissions. In addition, the country's growing life sciences innovation hubs foster partnerships between AI providers and pharma companies.
The AI in regulatory affairs market in Germany is supported by its strong pharmaceutical and MedTech sectors, which face complex regulatory frameworks under EMA and national BfArM oversight. In addition, the growth is driven by the EU AI Act, which creates a legal mandate for compliant AI deployment in 'high-risk' AI systems. Academic-industry collaborations in digital health accelerate the integration of AI into regulatory processes. Furthermore, the country's emphasis on innovation in precision medicine and digital therapeutics drives the adoption of AI-driven submissions.
Asia Pacific AI In Regulatory Affairs Market Trends
The Asia Pacific AI in regulatory affairs industry is expected to grow at the fastest CAGR in the coming years. This growth is attributed to the diverse regulatory frameworks across countries, which increase the need for AI-driven harmonization tools. In addition, the market is boosted by rapid growth in the pharmaceutical and biotechnology sectors, which increases demand for expedited approvals. Countries such as China, India, and Japan are scaling up clinical trial activity, driving AI adoption for efficiency. The region’s digital health boom and government-led healthcare modernization initiatives also accelerate the uptake of the AI-enabled platforms.
India’s AI in regulatory affairs market growth is driven by the country’s rapidly expanding pharmaceutical and biotechnology sectors, which demand efficient regulatory compliance to support accelerated drug approvals. Moreover, increasing government initiatives to digitize healthcare and streamline drug registration processes promote AI adoption in regulatory submissions and dossier management. For instance, in September 2025, India joined the HealthAI Global Regulatory Network (GRN), becoming a pioneer country in this global coalition of health regulators focused on the safe and effective use of AI in healthcare. This collaboration supports India’s national AI strategy, aiming to responsibly integrate AI across the health ecosystem and contribute to global AI governance efforts.
The AI in regulatory affairs market in Japan is expected to grow rapidly over the forecast period. Japan’s Pharmaceuticals and Medical Devices Agency (PMDA) is driving digital adoption, creating opportunities for AI in regulatory submissions and safety monitoring. Rising numbers of clinical trials in oncology and advanced therapies push demand for artificial intelligence (AI) to handle large datasets.
Latin America AI in Regulatory Affairs Market Trends
Latin America’s AI in regulatory affairs industry growth benefits from increasing clinical trial activities and investments in healthcare innovation. Governments in Brazil and Argentina are streamlining regulatory frameworks, encouraging AI adoption for compliance and submission management. In addition, the region’s expanding biotech and pharmaceutical sectors leverage AI for cost-efficient regulatory outsourcing. Furthermore, digital infrastructure improvements support AI-driven data analytics and regulatory intelligence applications. Growing emphasis on transparency and data integrity fosters further AI integration in life sciences regulatory processes.
Middle East and Africa AI in Regulatory Affairs Market Trends
The Middle East and Africa AI in regulatory affairs industry is expected to experience significant growth during the forecast period. This growth is attributed to the increasing clinical trials and rising investments in healthcare infrastructure. In addition, governments’ focus on regulatory harmonization and digital transformation encourages AI integration in regulatory affairs. Strategic development plans such as the Saudi Vision 2030 and UAE Vision 2031 emphasize innovation and the adoption of AI in life sciences regulatory processes.
Key AI In Regulatory Affairs Company Insights
Key players operating in the AI in regulatory affairs market are undertaking various initiatives to strengthen their market presence and increase the reach of their products and services. Strategies such as new product launches and partnerships play a key role in propelling market growth.
Key AI In Regulatory Affairs Companies:
The following are the leading companies in the AI in regulatory affairs market. These companies collectively hold the largest market share and dictate industry trends.
- Freyr
- Celegence
- Wipro
- Indegene
- Clarivate
- Zenovel
- RegDesk, Inc.
Recent Developments
- In August 2025, Clarivate launched an AI-powered Regulatory Assistant within the Cortellis Regulatory Intelligence suite, designed to help regulatory professionals easily navigate global requirements.
“Today’s regulatory environment is increasingly complex and fast-changing. The beta launch of the Regulatory Assistant is a key milestone in our mission to empower regulatory teams with intuitive, AI-driven tools that enhance productivity and support confident, compliant decision-making. By working closely with our Collaborative Partners, we are building a solution that reflects real-world workflows and priorities.”
-Yuval Kiselstein, VP, R&D, Life Sciences & Healthcare at Clarivate
-
In April 2025, EVERSANA launched EVERSANA ORCHESTRATE MLR, an AI-powered platform transforming the Medical, Legal, and Regulatory (MLR) review process. Integrated with Veeva PromoMats and developed with regulatory experts, it streamlines compliance, improves accuracy, and reduces administrative burden for life sciences companies.
-
In January 2025, AINGENS launched the Medical Affairs Content Generator (MACg), an AI-powered assistant designed to streamline scientific medical writing and regulatory submissions in life sciences.
AI In Regulatory Affairs Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 1.70 billion
Revenue forecast in 2033
USD 6.65 billion
Growth rate
CAGR of 18.60% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Component, deployment mode, application, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; Germany; UK; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; South Korea; Australia; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Freyr; Celegence; Wipro; Indegene; Clarivate; Zenovel; RegDesk, Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global AI In Regulatory Affairs Market Report Segmentation
This report forecasts revenue growth at the global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global AI in regulatory affairs market report based on component, deployment mode, application, end-use, and region:
-
Component Outlook (Revenue, USD Million, 2021 - 2033)
-
Software/Platforms
-
Services
-
-
Deployment Mode Outlook (Revenue, USD Million, 2021 - 2033)
-
Cloud-based
-
On-Premises
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Regulatory Intelligence
-
Data Migration & Integration
-
Dossier Management
-
Document Management
-
Product Registration & Approvals
-
Pharmacovigilance & Safety Reporting
-
Regulatory Submissions & Publishing
-
Others
-
-
End-use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical Companies
-
Biotechnology Companies
-
Medical Device Companies
-
CRO/CDMO
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
Japan
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
MEA
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










