- Home
- »
- Medical Devices
- »
-
Inhaled Drugs Contract Manufacturing Market Report, 2033GVR Report cover
![Inhaled Drugs Contract Manufacturing Market Size, Share & Trends Report]()
Inhaled Drugs Contract Manufacturing Market (2025 - 2033) Size, Share & Trends Analysis Report By Service (Clinical Manufacturing, Commercial Manufacturing, Packaging & Labeling), By Product (Active Pharmaceutical Ingredient, Finished Drug Service), By End-use, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-778-7
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
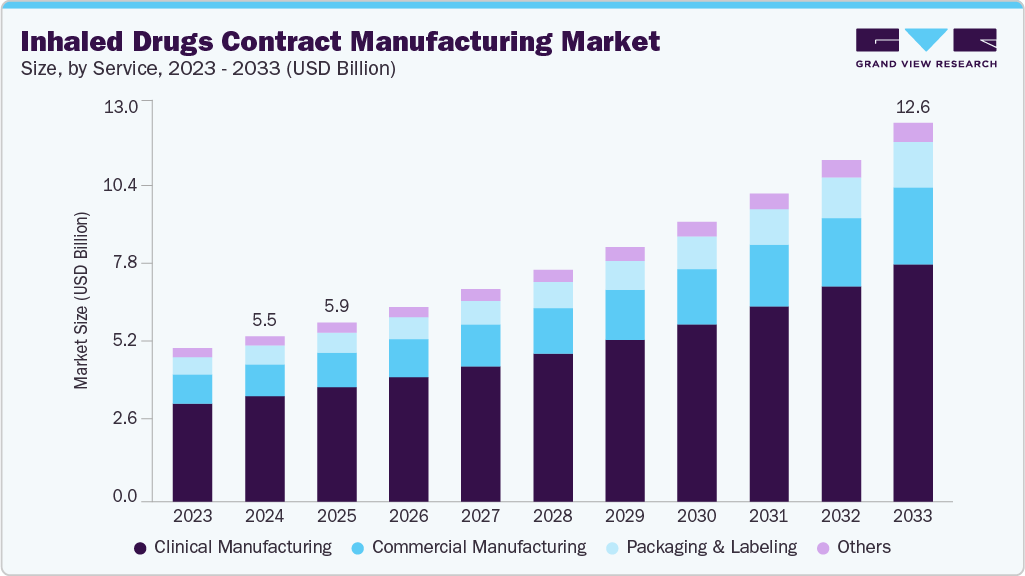
Inhaled Drugs Contract Manufacturing Market Summary
The global inhaled drugs contract manufacturing market size was estimated at USD 5.47 billion in 2024 and is projected to reach USD 12.56 billion by 2033, growing at a CAGR of 9.82% from 2025 to 2033. The market growth is driven due to the rising prevalence of respiratory disorders such as asthma and chronic obstructive pulmonary disease (COPD), which is fueling the demand for effective and targeted therapeutic options.
Key Market Trends & Insights
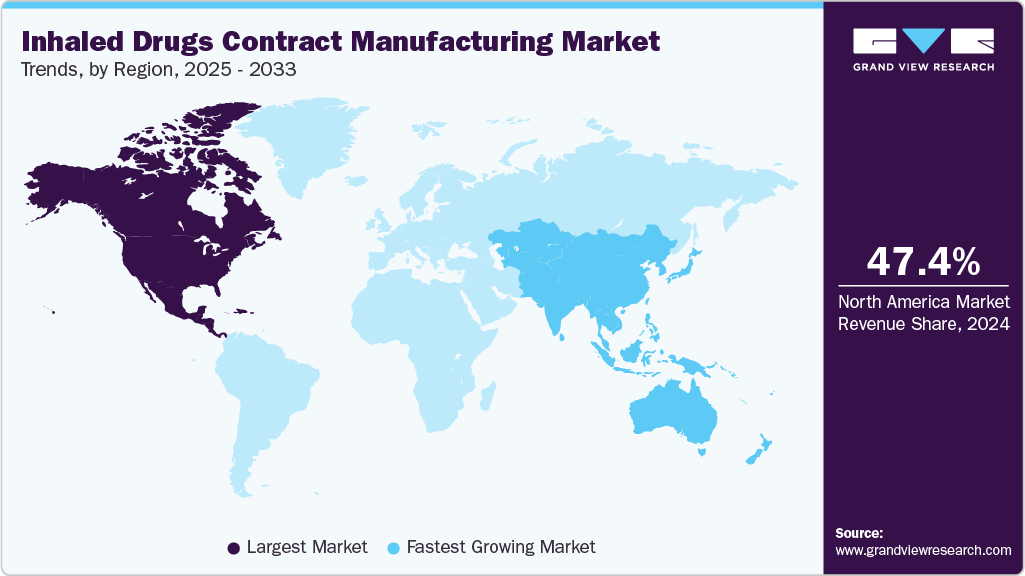
- The North America inhaled drugs contract manufacturing market held the largest global revenue share of 47.40% in 2024.
- The inhaled drugs contract manufacturing industry in the U.S. held the largest share in 2024.
- By service, the commercial manufacturing segment led the market with the largest revenue share of 63.91% in 2024.
- By product, the Active Pharmaceutical Ingredient (API) segment led the market with the largest revenue share in 2024.
- By end-use, the commercial segment held the largest revenue share of the global inhaled drugs contract manufacturing industry in 2024.
Market Size & Forecast
- 2024 Market Size: USD 5.47 Billion
- 2033 Projected Market Size: USD 12.56 Billion
- CAGR (2025-2033): 9.82%
- North America: Largest market in 2024
- Asia Pacific: Fastest growing market
The growing clinical need, coupled with the expanding adoption of advanced inhalation therapies, is prompting pharmaceutical and biopharmaceutical companies to increasingly outsource drug development and manufacturing processes to specialized contract manufacturing, which is contributing to the growth of the inhaled drugs contract manufacturing industry.In addition, the rising patient population suffering from chronic respiratory conditions, including asthma, COPD, among others, has accelerated the demand for effective inhalation therapies, including dry powder inhalers (DPIs), metered-dose inhalers (MDIs), and nebulizers. This growing therapeutic need is pushing pharmaceutical companies to expand their product pipelines and bring novel inhaled formulations to market faster. Developing these therapies requires specialized expertise in formulation, particle size optimization, and device compatibility, along with precision in dose delivery. Therefore, pharmaceutical companies are increasingly partnering with contract manufacturers organizations (CMOs) to leverage their technical capabilities, advanced infrastructure, and regulatory experience to enable faster development timelines and reduced capital expenditure. Thus, the aforementioned factors are driving the expansion of the inhaled drugs contract manufacturing market.
Furthermore, the need for cost optimization and operational efficiency is further accelerating the demand for contract manufacturing services in this market. Inhaled drug manufacturing involves high capital investment in device integration, cleanroom facilities, and aerosol testing, which can strain in-house resources. CDMOs with end-to-end capabilities, including formulation development, device engineering, and commercial-scale manufacturing, enable drug developers to minimize capital expenditure and accelerate time-to-market. This strategic outsourcing allows pharmaceutical companies to focus on core competencies such as research and marketing. At the same time, contract manufacturers benefit from long-term partnerships and recurring production contracts, contributing to sustained market growth.
Opportunity Analysis
The inhaled drugs contract manufacturing market presents significant growth opportunities due to increasing adoption of advanced inhalation technologies, including smart inhalers and combination drug-device products. Pharmaceutical companies are seeking cost-effective outsourcing solutions to reduce capital expenditure and accelerate time-to-market. Stringent regulatory requirements for dose accuracy, safety, and quality are further increasing the demand for CMOs. Moreover, expanding healthcare access and growing disposable income in emerging markets are creating new opportunities for market penetration.
Technological Advancements
Service Insights
The commercial manufacturing segment accounted for the largest revenue share of the inhaled drugs contract manufacturing industry with 63.91% in 2024. The segment's growth is due to increasing demand for large-scale production of inhaled therapies, rising outsourcing trends among pharmaceutical companies, and the need for cost-efficient, high-quality manufacturing solutions. Contract manufacturers with advanced facilities and expertise in scaling up formulations are increasingly relied upon to meet commercial supply requirements, ensuring consistent product quality and timely market availability. Thus, these factors are contributing to the segment’s market growth.
The packaging & labeling segment is anticipated to grow at the fastest CAGR during the forecast period. The segment growth is driven due to the increasing regulatory requirements for accurate labeling, serialization, and traceability of inhaled drug products. Rising demand for patient-centric packaging, including user-friendly inhaler devices and tamper-evident solutions, further supports market expansion.
Product Insights
The Active Pharmaceutical Ingredient (API) segment held the largest share of the inhaled drugs contract manufacturing market in 2024. The segment growth can be attributed to the increasing demand for high-quality, specialized APIs required for inhaled therapies, including complex formulations for asthma, COPD, and other respiratory disorders. Pharmaceutical companies are increasingly outsourcing API production to contract manufacturers to leverage their technical expertise, advanced facilities, and compliance with stringent regulatory standards. This enables faster development timelines, consistent product quality, and scalability which is further contributing to the segments market growth.
The finished drug product segment is anticipated to grow at a considerable CAGR during the forecast period. The segment’s growth is due to the rising demand for ready-to-use inhaled therapies, increasing preference for outsourcing end-to-end production, and the need for efficient scale-up from clinical to commercial supply. Contract manufacturers offering comprehensive capabilities in formulation, device integration, and quality control enable pharmaceutical companies to ensure timely product availability, regulatory compliance, and consistent therapeutic performance. These factors are contributing to the steady expansion of the finished drug product segment.
End-use Insights
The commercial segment held the largest revenue share of the global inhaled drugs contract manufacturing industry in 2024, owing to the growing demand for large-scale production of inhaled therapies to meet the growing needs of patient. Pharmaceutical companies are increasingly outsourcing commercial manufacturing to contract manufacturers with advanced capabilities in formulation, device integration, and regulatory compliance. This ensures consistent product quality, efficient scale-up, and timely market supply. Thus, the aforementioned factors are contributing to market growth.
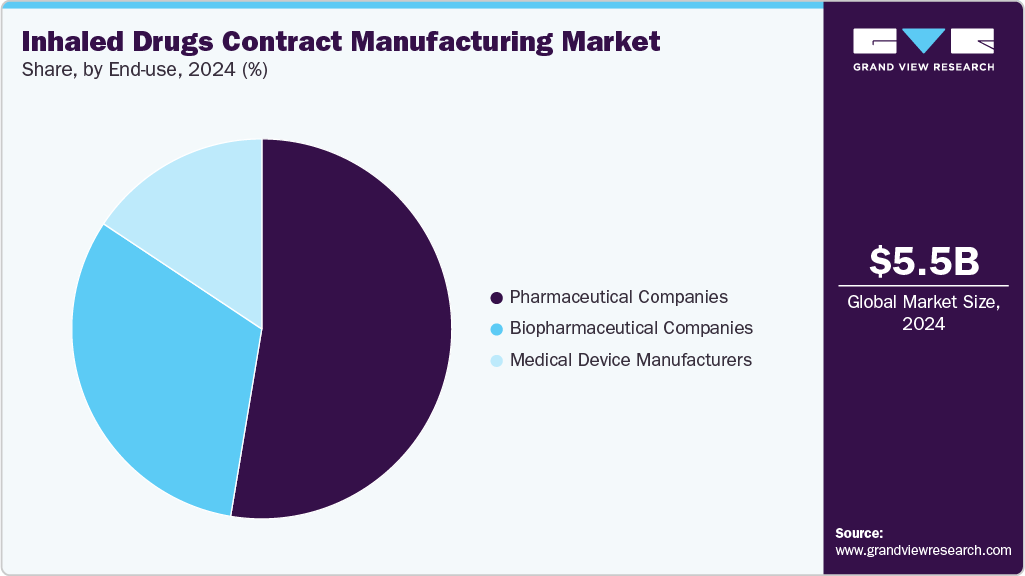
Biopharmaceutical companies segment is anticipated to grow at the fastest CAGR over the forecast period. The segment growth is driven due to the rising development of inhalable biologics, including protein- and peptide-based therapies, which require specialized manufacturing expertise. Contract manufacturers with advanced capabilities in aseptic processing, formulation development, and regulatory compliance are increasingly being relied upon to support the production of these complex therapies. This trend enables biopharmaceutical companies to accelerate time-to-market, ensure consistent product quality, and scale up efficiently, further contributing to the market growth.
Regional Insights
The North America inhaled drugs contract manufacturing market accounted for the largest revenue share of 47.40% in 2024. This is attributed to the regions advanced manufacturing facilities, higher concentration of clinical trials, and growing spending in research and development activities by the pharmaceutical companies.
U.S. Inhaled Drugs Contract Manufacturing Market Trends
The inhaled drugs contract manufacturing industry in the U.S. held the largest share in 2024. The country’s growth is due to high R&D spending, strong venture funding, and a well-established pharmaceutical and biotechnology infrastructure that supports innovation in inhaled therapies. In addition, favorable government policies, robust healthcare infrastructure, and a growing patient population with respiratory disorders is further contributing to the country’s dominance.
Europe Inhaled Drugs Contract Manufacturing Market Trends
The inhaled drugs contract manufacturing industry in Europe is expected to grow significantly due to increasing investments in pharmaceutical R&D, the presence of advanced manufacturing infrastructure, and stringent regulatory standards which is driving the demand for specialized contract manufacturing services.
The inhaled drugs contract manufacturing market in Germany held the significant share in 2024, owing to the country’s well-established pharmaceutical and biotechnology sector, strong focus on innovation, and advanced manufacturing infrastructure. Moreover, high R&D investments in respiratory therapies, coupled with strict regulatory standards for quality and safety, drive pharmaceutical companies to partner with experienced contract manufacturers.
The UK inhaled drugs contract manufacturing market held the significant share in 2024. The growth is due to the presence of leading contract manufacturing organizations with expertise in complex inhalation therapies, strong biopharmaceutical research clusters, and strategic partnerships between CMOs and pharmaceutical companies for innovative respiratory treatments. The U.K.’s emphasis on precision medicine and ongoing investment in advanced inhalation technology development further supports the expansion of the market.
Asia Pacific Inhaled Drugs Contract Manufacturing Market
The Asia Pacific inhaled drugs contract manufacturing industry is anticipated to witness the fastest CAGR over the estimated timeline. The regional growth is due to the increasing prevalence of respiratory diseases, increasing demand for advanced inhalation therapies, and expanding pharmaceutical R&D and manufacturing capabilities in countries such as China, India, and Japan. In addition, cost-efficient manufacturing, favorable government policies, and growing adoption of contract manufacturing services by local and multinational pharmaceutical companies are fueling the regional market expansion.
The inhaled drugs contract manufacturing market in China held the largest revenue share in 2024. The growth is due to the country’s robust pharmaceutical manufacturing infrastructure, increasing demand for inhaled therapies, and rising investments in advanced R&D for respiratory treatments. Moreover, supportive government initiatives, a large patient population, and the presence of cost-competitive contract manufacturers is also one of the factors contributing to the country’s leading position in the regional market.
The Japan inhaled drugs contract manufacturing market is expected to grow over the forecast period. The country’s growth is due toincreasing focus on advanced drug delivery systems, rising prevalence of asthma and COPD, and strong collaboration between domestic pharmaceutical companies and specialized contract manufacturers. Japan’s well-established regulatory framework, high adoption of innovative inhalation technologies, and continuous investment in precision medicine further support the market’s expansion.
The inhaled drugs contract manufacturing market in India is anticipated to grow at the lucrative CAGR over the forecast period. The country’s market growth is due to the lower operational costs, skilled scientific workforce, and growing R&D investments from both domestic and multinational companies.
Key Inhaled Drugs Contract Manufacturing Company Insights
The major players operating across the market are focused on adopting inorganic strategic initiatives such as mergers, partnerships, acquisitions, etc. Moreover, companies focus on technological innovations to augment their market position.
Key Inhaled Drugs Contract Manufacturing Companies:
The following are the leading companies in the inhaled drugs contract manufacturing market. These companies collectively hold the largest market share and dictate industry trends.
- Lonza Group
- Catalent Inc.
- Recipharm AB
- Hovione
- Kindeva Drug Delivery
- AptarGroup Inc.
- Vectura Group Ltd.
- Siegfried Holding AG
- CordenPharma
- PCI Pharma Services
Recent Developments
-
In April 2025, Lonza entered into a collaboration agreement with Ethris, a clinical-stage biotechnology company to develop a room-temperature stable, spray-dried formulations of mRNA-based vaccine candidates. This approach holds significant promise for combating respiratory diseases.
-
In September 2025, Kindeva announced to expand its service offerings by opening a new facility in UK. This expansion aims to test and develop next-generation propellants (NGP), aligning with the industry’s transition toward sustainable inhalation products. This strategic expansion strengthens Kindeva’s position in the market by meeting the growing demand for eco-friendly inhalation solutions.
Inhaled Drugs Contract Manufacturing Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 5.94 billion
Revenue forecast in 2033
USD 12.56 billion
Growth rate
CAGR of 9.82% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Service, product, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Norway; Denmark; Sweden; Japan; China; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; Kuwait; UAE; Oman; Qatar
Key companies profiled
Lonza Group; Catalent Inc.; Recipharm AB; Hovione; Kindeva Drug Delivery; AptarGroup Inc.; Vectura Group Ltd.; Siegfried Holding AG; CordenPharma; PCI Pharma Services
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Inhaled Drugs Contract Manufacturing Market Report Segmentation
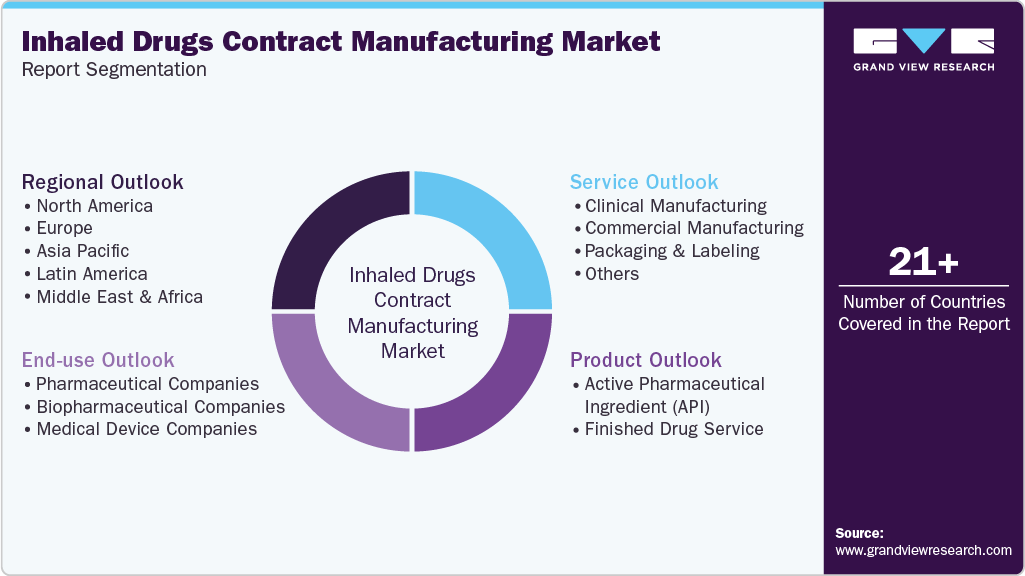
This report forecasts revenue growth at the global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global inhaled drugs contract manufacturing market report based on service, product, end-use, and region:
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Clinical Manufacturing
-
Commercial Manufacturing
-
Packaging & Labeling
-
Others
-
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Active Pharmaceutical Ingredient (API)
-
Finished Drug Service
-
Metered- dose inhalers (MDIs)
-
Dry Powder Inhalers
-
Nebulizers
-
Others
-
-
End-use Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical Companies
-
Biopharmaceutical Companies
-
Medical Device Companies
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Norway
-
Sweden
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
Oman
-
Qatar
-
-
Frequently Asked Questions About This Report
b. The global inhaled drugs contract manufacturing market size was estimated at USD 5.47 billion in 2024 and is expected to reach USD 5.94 billion in 2025.
b. The global inhaled drugs contract manufacturing market is expected to grow at a compound annual growth rate of 9.82% from 2025 to 2033 to reach USD 12.56 billion by 2033.
b. North America dominated the inhaled drugs contract manufacturing market, with a share of 47.40% in 2024. This is attributable to the region's advanced manufacturing facilities, higher concentration of clinical trials, and growing spending by pharmaceutical companies on research and development activities.
b. Some key players operating in the inhaled drugs contract manufacturing market include Lonza Group, Catalent Inc., Recipharm AB, Hovione, Kindeva Drug Delivery, AptarGroup Inc., Vectura Group Ltd., Siegfried Holding AG, CordenPharma, PCI Pharma Services.
b. Key factors driving the market growth include the rising prevalence of respiratory disorders such as asthma and chronic obstructive pulmonary disease (COPD) globally, coupled with the expanding adoption of advanced inhalation therapies.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










