- Home
- »
- Clinical Diagnostics
- »
-
Middle East Hereditary Cancer Testing Market Report, 2033GVR Report cover
![Middle East Hereditary Cancer Testing Market Size, Share & Trends Report]()
Middle East Hereditary Cancer Testing Market (2025 - 2033) Size, Share & Trends Analysis Report By Cancer, By Technology (Cytogenetic, Biochemical, Molecular Testing), By Test Type (Predictive Testing, Diagnostic Testing), By End-use, By Country, And Segment Forecasts
- Report ID: GVR-4-68040-724-3
- Number of Report Pages: 320
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
The Middle East hereditary cancer testing market size was estimated at USD 66.25 million in 2024 and is projected to reach USD 169.10 million by 2033, growing at a CAGR of 11.28% from 2025 to 2033. The market is expanding rapidly, driven by high cancer prevalence, consanguinity, and national precision medicine initiatives. In the UAE, partnerships such as Viafet Genomics with Euformatics are strengthening NGS-based hereditary cancer testing, supported by the National Cancer Control Plan. Qatar, through the Qatar Genome Programme and NCCCR, has pioneered BRCA testing and large-scale genome sequencing, positioning itself as a leader in precision oncology. Kuwait aligns with global hereditary cancer rates (10%) and is advancing genetic services, IVF success, and early cancer detection through KCCC-led initiatives.
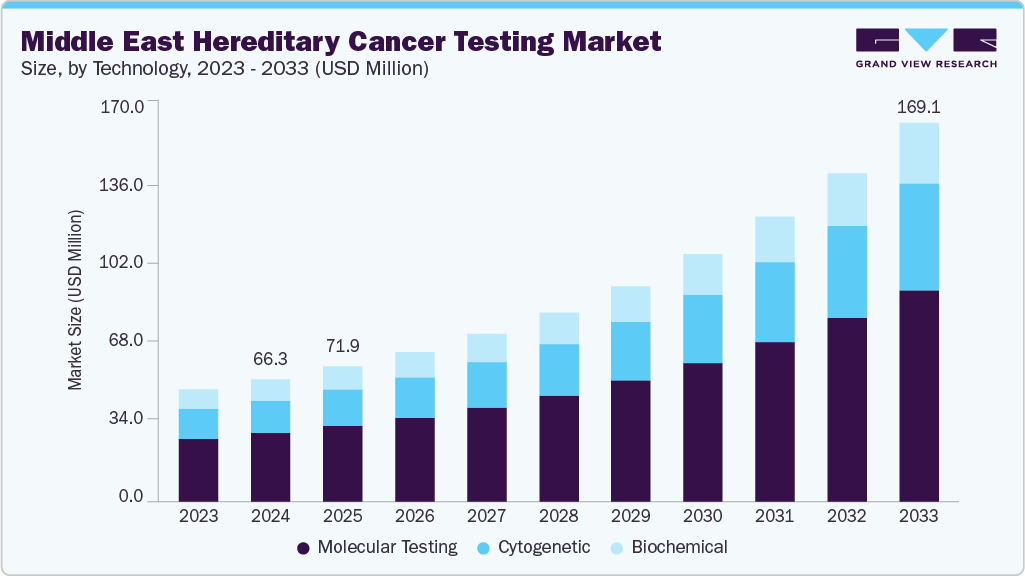
The Middle East hereditary cancer testing market is evolving rapidly, driven by high cancer incidence rates, unique demographic structures, and strong government-led precision medicine initiatives. Countries such as the UAE, Qatar, and Kuwait are spearheading regional adoption by embedding hereditary testing into cancer control frameworks, expanding laboratory capabilities, and integrating genomics into public health programs. Collectively, these factors are positioning the region as a fast-growing market for hereditary and predictive oncology diagnostics.
The UAE has emerged as a frontrunner in precision oncology. In 2021, the country reported 5,830 new cancer cases, with a crude incidence rate of 60.5 per 100,000 and an age-standardized rate of 107.8 per 100,000. Breast cancer was the leading malignancy, accounting for 20.3% of all cancers, followed by thyroid (10.6%) and colorectal cancers (9.5%). Emiratis represented 25.6% of cases, while expatriates-who form the majority population-accounted for 74.4%, underscoring the need for inclusive testing strategies. Despite clear hereditary risk, screening uptake remains low-only around 25% for both breast and colorectal cancer. This gap creates strong potential for hereditary testing to complement screening and identify high-risk individuals earlier. Hospitals are adopting NGS-based multi-gene panels, BRCA1/2, PALB2, and Lynch syndrome testing, as well as liquid biopsies. Policy frameworks such as the National Cancer Control Plan (2022-2026) and updated National Cancer Screening Guidelines mandate structured programs, integrate molecular diagnostics, and expand insurance coverage.
The UAE’s ecosystem is also being shaped by private-sector innovation and global partnerships. A notable example is the 2022 collaboration between Viafet Genomics Laboratory in Dubai and Euformatics, aimed at expanding clinical genetics services across the Gulf. Viafet has deployed Euformatics’ OmnomicsNGS bioinformatics platform in its data center to support a wide range of next-generation sequencing (NGS) applications, from whole exome analysis to carrier screening and hereditary cancer testing. The flexibility of the platform to manage both single-patient and multi-patient pipelines, combined with integrated quality control and validation, strengthens confidence in clinical-grade genetic data. By tailoring reporting frameworks and optimizing workflows, the partnership has enhanced Viafet’s capacity to deliver high-quality hereditary and cancer genomics services in the UAE. Together with national-level precision medicine policies, these advancements underscore why the UAE is set to grow at a double-digit CAGR, positioning it as a cornerstone of precision oncology in the region.
Qatar represents one of the region’s most advanced hereditary cancer testing ecosystems, shaped by its high consanguinity rate of nearly 55%, large family structures, and strong state-backed genomics programs. Breast cancer accounts for 36% of female cancers, with early-onset disease prevalent among Qatari women due to BRCA1/2 mutations and founder effects. The Hereditary Breast and Ovarian Cancer and High-Risk Clinic, launched in 2013 at the National Center for Cancer Care and Research (NCCCR), pioneered regional hereditary testing. By 2016, it had evaluated 697 patients, with 38% testing BRCA-positive, and such cases were strongly linked to young-onset, triple-negative breast cancer. In 2015, in-house CAP-accredited BRCA testing at Hamad Medical Corporation removed cost barriers and increased uptake. National-scale initiatives such as the Qatar Genome Programme and Qatar Biobank have already sequenced more than 29,000 genomes, targeting 100,000 by 2025, and developed tools such as QChipPMv2 for variant detection. Coupled with translational research at Sidra Medicine and QBRI, Qatar has built a globally competitive infrastructure. Furthermore, the launch of a genetic counseling master’s program in 2018 ensures culturally sensitive expertise, positioning Qatar as both a domestic leader and regional model for hereditary cancer testing and precision oncology.
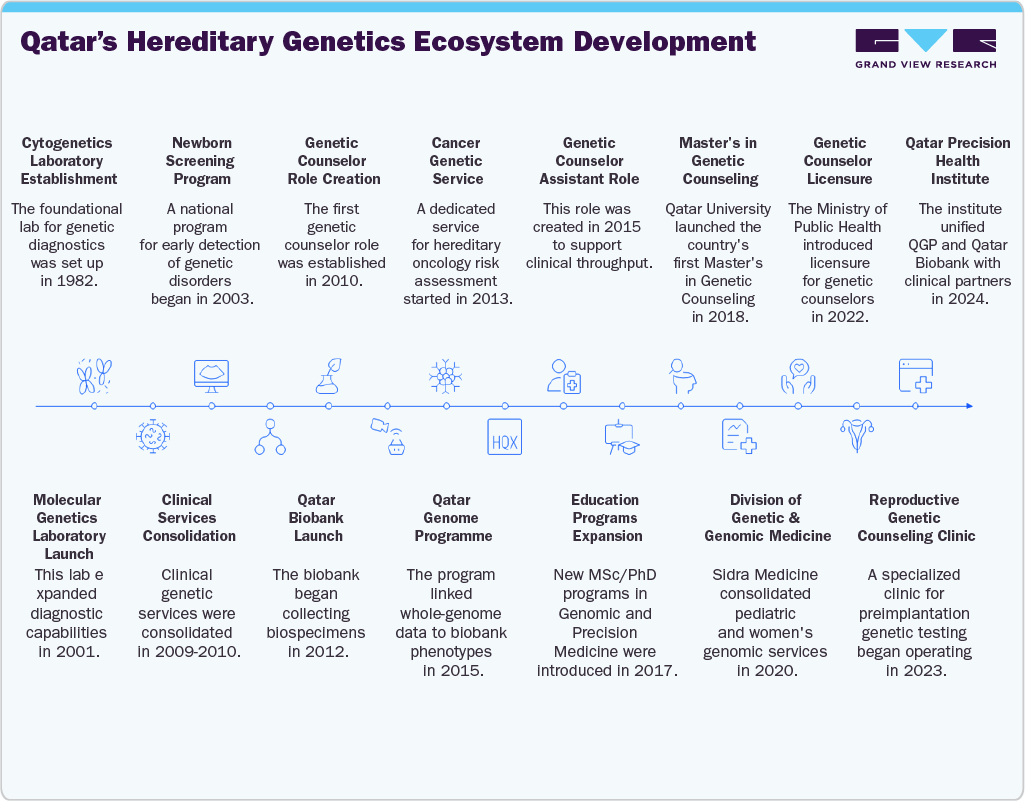
The timeline shows a deliberately sequenced build: lab capacity → national screening → dedicated clinics → population genomics → workforce training → regulation → subspecialty expansion → precision-health integration. For hereditary cancer specifically, this architecture means faster identification of high-risk families (BRCA/Lynch and beyond), earlier intervention via tailored surveillance and prophylaxis, streamlined access to multigene NGS testing, and a steady pipeline of licensed counselors to deliver culturally sensitive, evidence-based care.
In Kuwait, the hereditary cancer rate is estimated at 10%, aligning with global averages. The Kuwait Cancer Control Center plays a central role in advancing hereditary testing, supported by both public and private sector collaboration. The country’s most common cancers include breast, colon, ovarian, and uterine cancers, while leukemia, lymphoma, and brain cancers also show hereditary links. Significant progress has been made in medical genetics, from examinations to diagnostics and preventive medicine. The establishment of specialized genetic testing departments, such as at Ghanim Al-Ghanim Center, has expanded local capabilities. The application of pre-implantation embryo testing has reduced the percentage of genetic diseases, while the success rates of IVF treatment improved from 32% to 76%, demonstrating how genetic testing directly impacts health outcomes. Conferences such as the Genome Day Conference, organized in collaboration with KCCC and global partners such as Thermo Fisher, further emphasize Kuwait’s role in fostering research, awareness, and professional training.
Across the Middle East, hereditary cancer testing is moving from a niche service to a strategic pillar of cancer care. The combination of high prevalence of hereditary-linked cancers, strong government-backed genomics programs, growing investment in NGS and molecular diagnostics infrastructure, and workforce development in genetic counseling and oncology is creating a robust foundation for market expansion. Moreover, the integration of hereditary testing into premarital, prenatal, and cancer-specific care pathways reflects a preventive health model tailored to the region’s demographic and cultural needs. Looking ahead, the market is expected to grow at double-digit rates, with the UAE and Qatar leading adoption, while Kuwait and other GCC countries steadily expand capacity. By bridging conventional screening gaps, reducing late-stage diagnoses, and supporting precision medicine, hereditary cancer testing is becoming central to the region’s fight against cancer and long-term healthcare transformation.
Market Concentration & Characteristics
The Middle East hereditary cancer testing market demonstrates a notably high degree of innovation, especially in Qatar and the UAE. Qatar has progressed from cytogenetics to NGS, whole-genome sequencing, and population-scale programs such as Qatar Genome and Biobank, supported by academic programs in genomic medicine and genetic counseling. Specialized services from hereditary cancer clinics to preimplantation genetic testing reflect deep integration of precision care. In the UAE, partnerships such as Viafet-Euformatics have brought automated NGS bioinformatics into clinical practice. Across the region, innovation is transformative, enabling personalized oncology, preventive strategies, and rapid technological adoption that elevate the standard of genetic testing.
The Middle East hereditary cancer testing market has seen a measured but rising level of M&A activities, primarily shaped by the region’s unique healthcare landscape. Unlike the West, where consolidation is driven by scale, in the Middle East M&A is more focused on filling capability gaps-such as acquiring advanced sequencing platforms, bioinformatics expertise, or licensing proprietary assays. Cross-border investments are also emerging, with Gulf-based groups increasingly exploring equity stakes in European or U.S. genomics companies to localize cutting-edge hereditary testing solutions. This selective, capability-driven approach defines the region’s M&A trajectory.
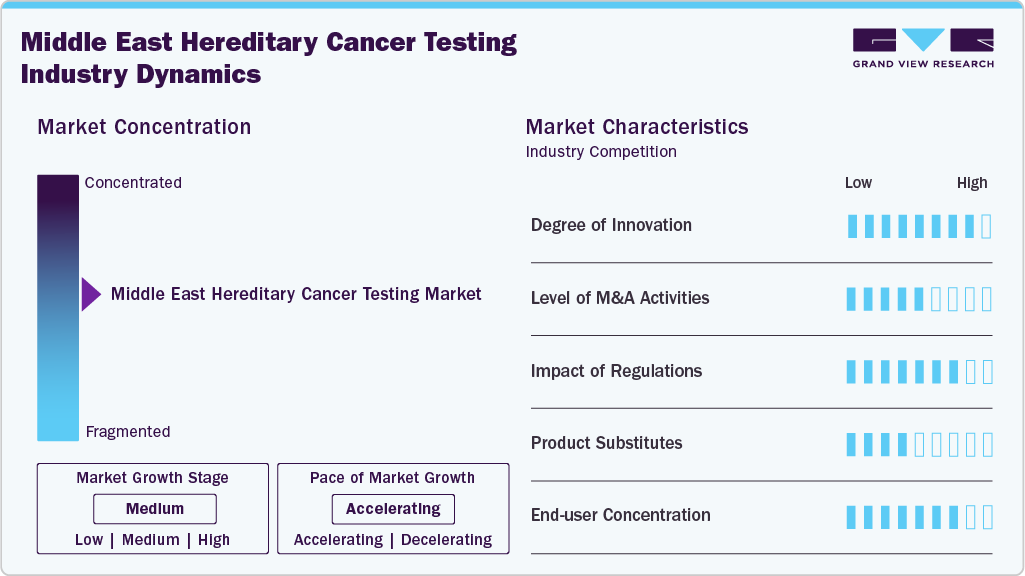
The regulatory environment is playing a decisive role in shaping the hereditary cancer testing market across the Middle East. Countries such as the UAE and Qatar have integrated genetic testing into national cancer control strategies, mandating structured screening for high-risk cancers such as breast and colorectal. Regulatory frameworks are increasingly aligned with international standards (CAP, CLIA, ISO), which ensures quality assurance and builds trust among physicians and patients. At the same time, governments are tightening rules on data security, genomic storage, and cross-border sample transfers, which affects how multinational labs operate in the region. Insurance-linked regulations, particularly in the UAE, are also driving adoption by requiring reimbursement pathways for NGS-based hereditary tests. Overall, regulation is acting as both a growth enabler and a compliance challenge, accelerating adoption while demanding higher operational rigor from labs and diagnostic players.
Product expansion in the Middle East hereditary cancer testing market is being driven by both local innovation and global partnerships. Diagnostic providers are moving beyond single-gene tests to multi-gene NGS panels that cover BRCA1/2, PALB2, Lynch syndrome genes, and other hereditary cancer markers. For example, in the UAE, Viafet Genomics Laboratory expanded its menu to include carrier screening, whole exome sequencing, and hereditary cancer testing through its partnership with Euformatics, strengthening in-house genomic capabilities. Qatar’s National Center for Cancer Care and Research (NCCCR) and Sidra Medicine are also broadening their offerings with liquid biopsy and exome-based risk panels, making testing less invasive and more comprehensive. In parallel, international companies are launching region-specific solutions such as Devyser’s BRCA PALB2 kit-while local labs customize reporting frameworks to reflect consanguinity-linked risk variants prevalent in the Gulf. This expansion ensures broader clinical utility, supports precision medicine initiatives, and makes hereditary testing accessible to a wider population base.
Regional expansion in the Middle East hereditary cancer testing market is being driven by population-scale genomics programs and cross-border healthcare strategies. In the UAE, expansion is supported by advanced oncology centers and government-backed insurance coverage, with hospitals integrating NGS-based hereditary testing into routine care. Qatar has built one of the region’s strongest precision health infrastructures, leveraging the Qatar Genome Programme and Biobank to expand clinical genetics and predictive testing nationwide. Saudi Arabia, through Vision 2030, is rapidly scaling genomic centers and hereditary testing as part of its broader cancer care transformation, with pilot projects already extending services into tertiary hospitals. Kuwait and Oman are gradually expanding through collaborations with international diagnostics companies and regional academic institutions, while also strengthening premarital and population screening programs. Collectively, these initiatives reflect a regional push to not only meet rising domestic demand but also to position the Middle East as a leader in hereditary cancer genomics across MENA.
Cancer Insights
The breast cancer dominated the market with largest revenue share of 11.15% and was amongst the fastest growing in this segment with a CAGR of 11.82% in 2024. The hereditary cancer testing market driven by increasing awareness of genetic risk factors, particularly mutations in BRCA1/2, which significantly elevate breast cancer risk. With growing demand for early detection and personalized treatment, genetic testing enables tailored preventive strategies, such as targeted therapies and prophylactic surgeries, improving patient outcomes.
Another key driving factor in this segment are the ongoing advancements such as next-generation sequencing (NGS) have revolutionized testing, enabling comprehensive analysis of multiple cancer-related genes, while AI and machine learning enhance data interpretation for more accurate risk assessments. Liquid biopsy technology under molecular testing also offers a non-invasive alternative for detecting genetic mutations, making testing more accessible. Transformations in research for identifying genetic mutations has broadened the scope of hereditary cancer testing, making it more accurate and accessible.
Increasing awareness of genetic factors contributing to breast cancer, such as BRCA1 and BRCA2 mutations, is driving more people to seek hereditary cancer testing. The rising demand for early detection and risk assessment is helping detect high-risk individuals to combat this deadly disease.
Technology Insights
The molecular testing segment held the highest position accounting for the largest share of 56.48% in 2024. Molecular testing in the hereditary cancer testing market is expediting, driven by the need for early detection and personalized treatment. One of the primary drivers is the increasing recognition of genetic mutations as key risk factors for various cancers. For identifying genetic mutations molecular testing is first choice in the market that enables its association with various cancers, allowing for individual assessments for prevention strategies and treatment plans. As individuals become more aware of the importance of genetics in cancer prevention and treatment, the demand for molecular testing is rising. Furthermore, the growing emphasis on precision medicine is accelerating the adoption of molecular tests, which enable healthcare providers to provide treatments based on an individual's genetic makeup. Advancements in molecular testing technologies, such as next-generation sequencing (NGS), are transforming the landscape.
In addition, cytogenetic segment is the fastest-growing in the hereditary cancer testing market, driven by increasing understanding of how chromosomal alterations, such as translocations, deletions, and duplications, contribute to the development of hereditary cancers. It also enables more accurate genetic counseling for families at risk of passing on cancer-related mutations, which is crucial for preventive strategies. Advancements in cytogenetics have led to improved testing methods, such as fluorescence in situ hybridization (FISH) and array comparative genomic hybridization (aCGH), which allow for the detection of chromosomal abnormalities at a more granular level. These technologies have enhanced the precision of hereditary cancer risk assessments. As healthcare increasingly adopts genomic innovations, cytogenetic systems will continue to lead medical advancements, providing unmatched insights into the genetic foundations of diseases and revolutionizing patient care.
Test Type Insights
The predictive test accounted for the largest share of 66.60% and was among the fastest growing test in the segment for the year 2024. The expansion of this segment is driven by the heightened focus on genetic counseling services and the growing incidence of genetic diseases. The significance of predictive testing lies in its ability to identify individuals at high genetic risk before cancer develops, allowing for early preventive measures that can save lives. These predictive tests allow for better-informed decisions regarding lifestyle changes, enhanced screening, and therapeutic interventions.
Advancements in predictive testing technologies have played a critical role in expanding the scope and accessibility of genetic testing. Next-generation sequencing (NGS) has made it possible to analyze multiple genes simultaneously, offering a more comprehensive risk assessment for various cancers. Liquid biopsy, a non-invasive alternative, is also emerging as a promising tool for predictive testing, offering greater convenience and accessibility for patients. These advancements are making predictive tests an essential part of modern cancer care.
End Use Insights
The hospitals dominated the end use segments with the largest market share of 66.60% in 2024 and were the fastest growing among the other segments over the period. Hospitals are increasingly adopting hereditary cancer testing as part of personalized treatment strategies. By understanding an individual's genetic predisposition, healthcare providers can tailor prevention, diagnosis, and treatment plans, leading to improved patient outcomes.
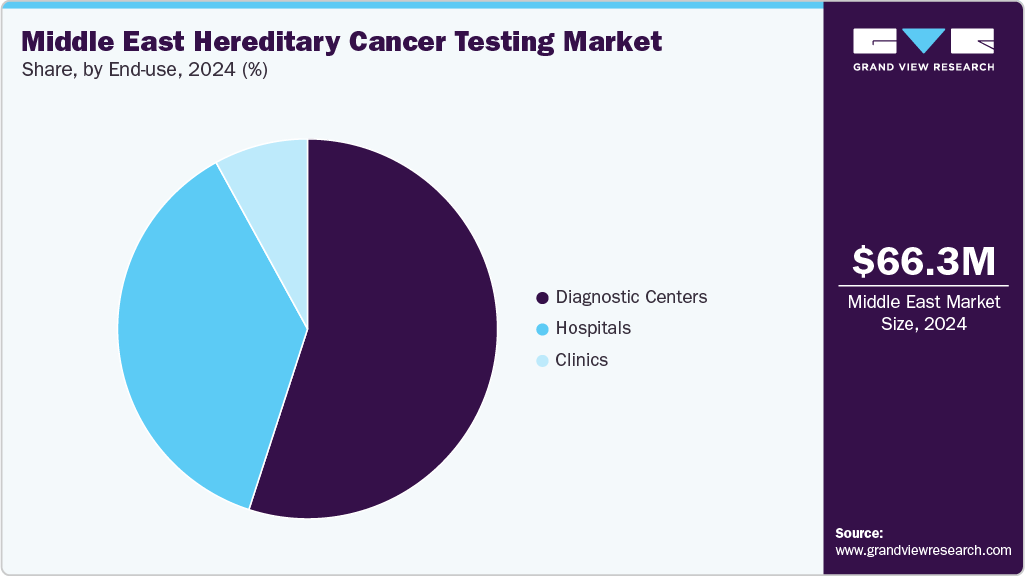
Government programs and policies promoting genetic testing and cancer screening are facilitating the adoption of hereditary cancer testing in hospitals. For instance, the Centers for Medicare & Medicaid Services (CMS) in the U.S. have expanded coverage for next-generation sequencing tests for patients with advanced cancer, encouraging hospitals to integrate these services.
Country Insights
The Middle East hereditary cancer testing market is undergoing a period of accelerated growth, driven by rising cancer incidence, strong government backing, and increasing adoption of precision medicine. Countries such as the UAE and Qatar are at the forefront, integrating hereditary testing into national cancer frameworks, expanding genomic databases, and offering insurance-backed NGS testing. High rates of consanguinity across the region amplify the prevalence of hereditary cancers, strengthening the demand for predictive and molecular testing. Regional players are collaborating with global technology providers, while population-scale genome programs in Qatar, Saudi Arabia, and the UAE are establishing the foundation for large-scale adoption. Hospitals remain the primary hub for testing, supported by initiatives such as premarital and newborn screening programs, genetic counseling workforce development, and international partnerships. Together, these trends highlight a market shifting from niche, high-cost services to mainstream adoption, positioning the Middle East as a rising hub for hereditary cancer diagnostics and precision oncology.
Saudi Arabia Hereditary Cancer Testing Market Trends
Saudi Arabia is witnessing rapid growth in hereditary cancer testing, fueled by high consanguinity rates, rising cancer incidence, and strong government initiatives under Vision 2030. Breast and colorectal cancers are leading priorities, with hereditary factors playing a major role. Hospitals and genomic centers such as King Faisal Specialist Hospital and Research Centre are expanding NGS-based panels and predictive testing. Public awareness campaigns, premarital screening programs, and collaborations with international genomics firms are driving adoption, while insurance reforms increasingly support access to advanced hereditary testing services.
UAE Hereditary Cancer Testing Market Trends
The UAE market is expanding steadily, supported by the National Cancer Control Plan (2022-2026), strong investment in genomic medicine, and advanced oncology centers. Breast and colorectal cancers dominate incidence, creating demand for BRCA and Lynch syndrome testing. Hospitals and private labs are rapidly adopting multi-gene NGS panels and liquid biopsy. Collaborations, such as Viafet Genomics with Euformatics, highlight ongoing product innovation and data-driven integration. Government-backed cancer screening guidelines and insurance coverage expansions are accelerating adoption, positioning the UAE as a regional hub for precision oncology and hereditary cancer diagnostics.
Kuwait Hereditary Cancer Testing Market Trends
In Kuwait, hereditary cancer testing is gaining traction due to rising cancer incidence, particularly breast, colorectal, and ovarian cancers. The country’s high prevalence of lifestyle-related cancers, coupled with genetic predispositions, is prompting demand for predictive and molecular diagnostics. Government healthcare spending and the integration of hereditary testing in public hospitals are facilitating adoption. Kuwait Cancer Control Centre and other tertiary hospitals are beginning to incorporate BRCA and multi-gene panel testing. Growing awareness among clinicians and patients, along with regional collaborations, is driving market growth and expanding precision medicine adoption.
Qatar Hereditary Cancer Testing Market Trends
Qatar is leading the region with strong genomic infrastructure, including the Qatar Genome Programme and Biobank, which support population-scale risk prediction. High consanguinity rates and elevated BRCA1/2 prevalence, especially among younger women with breast cancer, are driving testing demand. Hamad Medical Corporation and Sidra Medicine have integrated hereditary testing across oncology and genetic counseling services. Local CAP-accredited BRCA testing and advanced NGS platforms are reducing cost and turnaround times. Academic and clinical integration, workforce development in genetic counseling, and national cancer strategies are positioning Qatar as a precision oncology pioneer in the Middle East.
Oman Hereditary Cancer Testing Market Trends
Oman is gradually strengthening its hereditary cancer testing ecosystem, with breast and gastrointestinal cancers being key areas of focus. The country’s high consanguinity rates and family clustering of cancers underscore the need for predictive genetic testing. The Ministry of Health has been enhancing cancer registries, promoting screening awareness, and collaborating with international organizations to expand genetic services. Hospitals in Muscat are adopting NGS-based hereditary panels and predictive tests, though access remains limited compared to GCC peers. Growing public awareness, government-backed initiatives, and regional partnerships are expected to drive future expansion of hereditary testing services.
Key Middle East Hereditary Cancer Testing Company Insights
Some of the key players operating in the market include Myriad Genetics, Inc., Invitae Corporation, Illumina, Inc., Bio-Rad Laboratories, Qiagen NV, F. Hoffmann-La Roche Ltd., EUROFINS SCIENTIFIC and Thermo Fisher Scientific, Inc. New disease type launch, mergers and acquisitions, and collaborations are some of the major strategies adopted by these players to counter the stiff competition.
Key Middle East Hereditary Cancer Testing Companies:
- Myriad Genetics
- Invitae Corporation
- Bio-Rad Laboratories
- CSL Ltd
- Qiagen NV
- Danaher Corporation
- Thermo Fisher Scientific
- Abbott Laboratories
- EUROFINS SCIENTIFIC
- F. HOFFMANN-LA ROCHE LTD
- Illumina, Inc.
Recent Developments
- In February 2025, BGI Genomics, through its joint venture Genalive with Saudi partner Tibbiyah Holding, has secured a landmark three-year contract valued at approximately USD 131 million from the Saudi National Unified Procurement Company (NUPCO), the largest outsourced testing services tender in the Kingdom’s history. Covering 930,000 testing services across 83 public hospitals, the initiative spans advanced methodologies including NGS, pathology, cytogenetics, and Sanger sequencing. Genalive’s portfolio will deliver WGS, WES, hereditary cancer screening, PGT, and NIPT. The collaboration significantly strengthens Saudi Arabia’s precision medicine infrastructure and supports Vision 2030 by integrating AI-enabled genomics with national healthcare systems to enhance early detection, prevention, and personalized care.
Middle East Hereditary Cancer Testing Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 71.91 million
Revenue forecast in 2033
USD 169.10 million
Growth rate
CAGR of 11.28% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD billion/million and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Cancer, test type, technology, end use, country
Regional scope
Middle East
Country scope
UAE; Kuwait; Oman; Qatar
Key companies profiled
Myriad Genetics; Invitae Corporation; Bio-Rad Laboratories; CSL Ltd; Qiagen NV; Danaher Corporation; Thermo Fisher Scientific; Abbott Laboratories; EUROFINS SCIENTIFIC; F. HOFFMANN-LA ROCHE LTD; Illumina, Inc.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Middle East Hereditary Cancer Testing Market Report Segmentation
This report forecasts revenue growth at regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For the purpose of this study, Grand View Research has segmented the Middle East hereditary cancer testing market report on the basis of cancer, test type, technology, end use, and country:
-
Cancer Outlook (Revenue, USD Million, 2021 - 2033)
-
Lung Cancer
-
Breast Cancer
-
Colorectal Cancer
-
Cervical Cancer
-
Ovarian Cancer
-
Prostate Cancer
-
Stomach/Gastric Cancer
-
Melanoma
-
Sarcoma
-
Uterine Cancer
-
Pancreatic Cancer
-
Others
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Cytogenetic
-
Biochemical
-
Molecular Testing
-
-
Test Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Predictive Testing
-
Diagnostic Testing
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Diagnostic Centers
-
Hospitals
-
Clinics
-
-
Country Outlook (Revenue in USD Million, 2021 - 2033)
-
Middle East
-
UAE
-
Kuwait
-
Oman
-
Qatar
-
-
Frequently Asked Questions About This Report
b. The Middle East hereditary cancer testing market size was estimated at USD 66.25 million in 2024 and is expected to reach USD 71.91 million in 2025.
b. The Middle East hereditary cancer testing market is expected to witness a compound annual growth rate of 11.28% from 2025 to 2033 to reach USD 169.10 million in 2033.
b. The breast cancer dominated the market with largest revenue share of 11.15% and was amongst the fastest growing in this segment with a CAGR of 11.82% in 2024. The hereditary cancer testing market driven by increasing awareness of genetic risk factors, particularly mutations in BRCA1/2, which significantly elevate breast cancer risk. With growing demand for early detection and personalized treatment, genetic testing enables tailored preventive strategies, such as targeted therapies and prophylactic surgeries, improving patient outcomes
b. Some key players operating in the Middle East hereditary cancer testing market include Myriad Genetics, Invitae Corporation, Bio-Rad Laboratories, CSL Ltd, Qiagen NV, Danaher Corporation, Thermo Fisher Scientific, Abbott Laboratories, EUROFINS SCIENTIFIC, F. HOFFMANN-LA ROCHE LTD, Illumina, Inc.
b. The Middle East hereditary cancer testing market is expanding rapidly, driven by high cancer prevalence, consanguinity, and national precision medicine initiatives. In the UAE, partnerships like Viafet Genomics with Euformatics are strengthening NGS-based hereditary cancer testing, supported by the National Cancer Control Plan. Qatar, through the Qatar Genome Programme and NCCCR, has pioneered BRCA testing and large-scale genome sequencing, positioning itself as a leader in precision oncology. Kuwait aligns with global hereditary cancer rates (10%) and is advancing genetic services, IVF success, and early cancer detection through KCCC-led initiatives.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










