- Home
- »
- Biotechnology
- »
-
Middle East Plasmid DNA Manufacturing Market Report, 2033GVR Report cover
![Middle East Plasmid DNA Manufacturing Market Size, Share & Trends Report]()
Middle East Plasmid DNA Manufacturing Market (2025 - 2033) Size, Share & Trends Analysis Report By Disease (Cancer, Infectious Diseases), By Grade (R&D, GMP), By Application, By Development Phase, By Country, And Segment Forecasts
- Report ID: GVR-4-68040-709-8
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
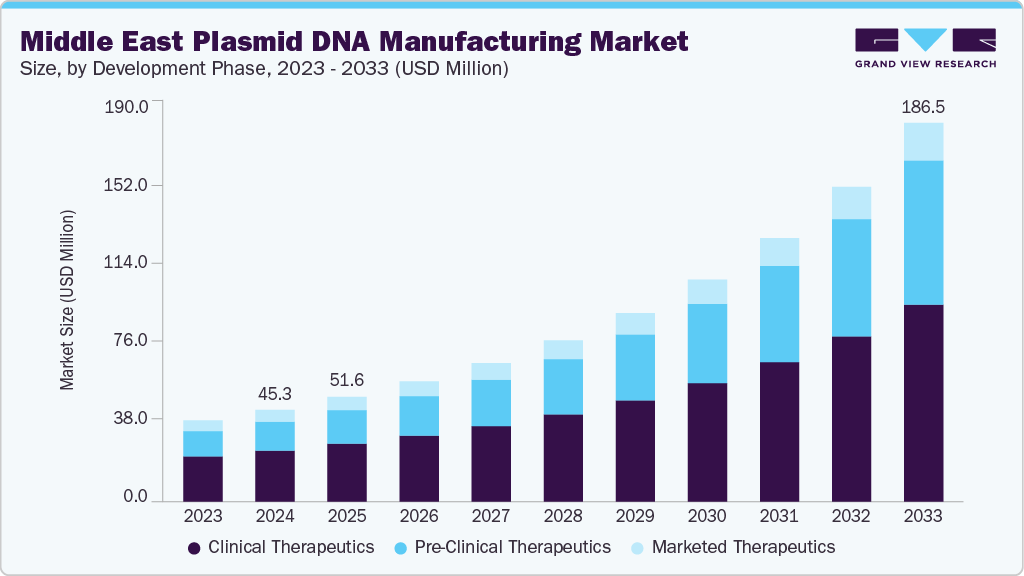
The Middle East plasmid DNA manufacturing market size was estimated at USD 45.3 million in 2024 and is expected to reach USD 186.5 million by 2033, expanding at a CAGR of 17.43% from 2025 to 2033. Growth is driven by increasing investments in biotechnology, the expansion of GMP-compliant facilities, and supportive government initiatives under national life sciences strategies. Strategic partnerships with global biopharma companies, coupled with advanced R&D infrastructure and strong logistics capabilities, are further enhancing the region’s position as a competitive hub for plasmid DNA production.
Increased Investment in Healthcare Transformation
Under Vision 2030, Saudi Arabia is committing over USD 65 billion to transform its healthcare system. The initiative aims to revamp infrastructure, establish 21 health clusters, scale up e-health services, and increase private sector participation from 40% to 65% by 2030. The Kingdom is also accelerating efforts to localize pharmaceutical production, reduce import dependence, and boost value creation in life sciences. These reforms are designed to enhance quality, accessibility, and efficiency while opening the market to global partnerships.
India, with its globally competitive pharmaceutical industry, is well-positioned to support Saudi Arabia’s diversification in healthcare. Accounting for about 20% of global generic medicine exports and being the world’s largest vaccine producer by volume, India offers affordable, high-quality healthcare solutions. Its strong track record in global vaccine supply and pharmaceutical manufacturing demonstrates reliability and the capacity to meet large-scale demand. During the COVID-19 pandemic, India supplied millions of vaccine doses to Saudi Arabia, reinforcing its role as a strategic healthcare partner.
To fully capitalize on this synergy, streamlined regulatory cooperation, technology transfer agreements, and localization incentives will be critical. Saudi procurement authorities are encouraging domestic manufacturing through offtake agreements and strategic partnerships. Several Indian pharmaceutical firms have already entered the Saudi market, secured local regulatory approvals and established a presence. Joint ventures and localized production facilities are expected to further strengthen bilateral trade, enhance Saudi self-sufficiency in essential medicines, and provide a gateway for Indian companies to access the broader MENA healthcare market.
Prevalence of Chronic & Rare Diseases
The growing incidence of cancer, genetic disorders, and infectious diseases is a key driver of demand in the Middle East plasmid DNA manufacturing market. The region is witnessing an increasing burden of oncology cases, particularly breast, colorectal, and hematological cancers, which are fueling the adoption of gene-based therapeutic approaches, including plasmid DNA vaccines, immunotherapies, and targeted gene delivery systems. Likewise, the rising diagnosis of rare genetic disorders, such as Duchenne muscular dystrophy, sickle cell anemia, and certain metabolic syndromes, is intensifying the need for innovative treatments leveraging plasmid-based gene therapy vectors. These therapies offer the potential to address conditions with limited or no existing cures, thereby driving investment in advanced manufacturing capabilities.
The prevalence of infectious diseases, including viral outbreaks such as Middle East Respiratory Syndrome (MERS), is further accelerating the region’s interest in DNA-based vaccine development. Plasmid DNA plays a critical role in the rapid design and scalable production of such vaccines, enabling faster response times to emerging health threats. Strong governmental initiatives, such as Saudi Arabia’s Vision 2030 and the UAE’s National Biotechnology Strategy, are promoting local R&D, clinical trial expansion, and biotech infrastructure development to address chronic and rare disease treatment needs. Combined with the entry of global biotech leaders through strategic partnerships, these factors are fostering a robust ecosystem for plasmid DNA manufacturing in the Middle East.
Market Concentration & Characteristics
The moderate degree of innovation in plasmid DNA manufacturing is a key driver of market growth in the Middle East, enabling cost-efficient, scalable, and high-quality production processes. Advancements in upstream and downstream processing, adoption of single-use technologies, and integration of automation are enhancing operational efficiency and reducing turnaround times. These innovations are supported by government-led initiatives, strategic industry-academia collaborations, and partnerships with biotechnology firms. As regional players invest in modernizing facilities and aligning with international regulatory standards, the Middle East is strengthening its competitive edge, attracting contract manufacturing opportunities, and positioning itself as a strategic hub for plasmid DNA supply in global markets.
M&A activity in the Middle East plasmid DNA manufacturing industry is moderate but growing, driven by the need to expand technological capabilities, enhance GMP-compliant production capacity, and access advanced bioprocessing expertise. Strategic acquisitions and partnerships enable regional players to strengthen their market position, diversify service offerings, and accelerate entry into high-growth segments such as gene therapy, cell-based research, and advanced biologics manufacturing.
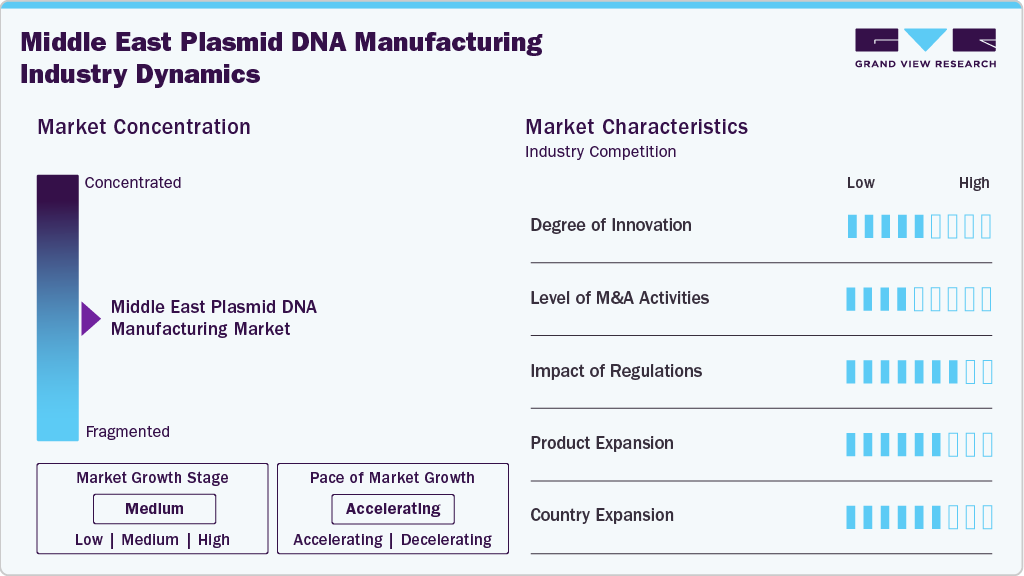
Regulations greatly impact the Middle East plasmid DNA manufacturing industry, influencing quality standards, production processes, and market entry requirements. Compliance with international GMP guidelines, biosafety protocols, and ethical frameworks ensures product safety and global acceptance. Government agencies are increasingly aligning regulatory frameworks with global best practices, fostering investor confidence, facilitating cross-border collaborations, and supporting the development of a robust and competitive plasmid DNA manufacturing ecosystem in the region.
Product expansion drives growth in the Middle East plasmid DNA manufacturing market as providers broaden their offerings to include high-yield vectors, GMP-grade plasmids, and customized production services, catering to increasing demand from gene therapy, vaccine development, and advanced research applications across regional and global markets.
The Middle East plasmid DNA manufacturing industry is expanding across countries like the UAE, Saudi Arabia, and Qatar, driven by significant government investments in biotechnology, the establishment of GMP-compliant facilities, and strategic collaborations with global biopharma firms. Growing demand for gene therapies, vaccines, and advanced research tools further accelerates regional market growth.
Grade Insights
The GMP grade segment dominated the Middle East plasmid DNA manufacturing market in 2024 and is anticipated to grow at the fastest CAGR over the forecast period. This is driven by growing demand for high-quality plasmids in gene therapy, vaccine development, and advanced clinical research. The expansion of GMP-certified facilities, particularly in the UAE and Saudi Arabia, alongside stringent regulatory compliance, has enhanced regional production capacity. Strategic collaborations with global biopharma firms and investments in advanced manufacturing technologies are further consolidating the segment’s leadership, positioning it for sustained growth throughout the forecast period.
The R&D grade segment is expected to witness a significant CAGR in the Middle East plasmid DNA manufacturing industry over the forecast period. This growth is fueled by increasing demand from academic institutions, biotech startups, and pharmaceutical companies for cost-effective, high-quality plasmids in preclinical research. Expanding research funding and supportive government initiatives are further accelerating adoption across diverse life sciences applications.
Development Phase Insights
The clinical therapeutics segment dominated the Middle East plasmid DNA manufacturing market in 2024 with a market share of 55.53%, driven by the rising adoption of plasmid-based vectors in gene therapy, cancer immunotherapy, and DNA vaccines. Growing investment in advanced therapeutics, coupled with expanding GMP-certified manufacturing capacity in countries like the UAE and Saudi Arabia, is strengthening the region’s role in supplying high-quality plasmids for clinical use, positioning the segment for continued leadership during the forecast period.
The pre-clinical therapeutics segment will witness the fastest CAGR of 19.92% in the Middle East plasmid DNA manufacturing industry over the forecast period. This growth is fueled by increasing demand for plasmids in early-stage drug development, rising research collaborations, and supportive funding for translational studies, accelerating innovation across gene therapy and advanced biologics pipelines.
Application Insights
The cell & gene therapy segment dominated the Middle East plasmid DNA manufacturing market with a revenue share of 55.84% in 2024, driven by the rising adoption of plasmid-based vectors in advanced therapies such as CAR-T cell treatments and gene editing. Strong government funding, the expansion of GMP-certified facilities, and strategic alliances with global biotech companies are bolstering regional production capacity. For instance, in February 2025, the Saudi Food and Drug Authority approved the Kingdom’s first gene therapy, marking a significant step toward strengthening the therapeutic and manufacturing ecosystem in the forecast period.
The DNA vaccines segment is projected to witness the fastest CAGR of 19.97% over the forecast period. This is driven by growing adoption of plasmid DNA platforms for infectious diseases and cancer immunotherapy. Advancements in delivery technologies, increased regional biotech investments, and government-backed health initiatives are accelerating development. For instance, in August 2021, INOVIO began a Phase 2 trial for its DNA vaccine against MERS, underscoring the region’s expanding role in plasmid DNA-based vaccine innovation.
Disease Insights
The cancer segment dominated the Middle East plasmid DNA manufacturing market in 2024 accounting for a market share of 38.43% and is anticipated to grow at the fastest CAGR over the forecast period. This is driven by the rising incidence of cancer, growing adoption of personalized medicine, and increasing investments in advanced cell and gene therapies. For instance, in May 2025, ImmunityBio, in collaboration with Saudi Arabia’s Ministry of Investment, King Faisal Specialist Hospital & Research Centre, and KAIMRC, announced the launch of the Cancer BioShield platform, enhancing regional capabilities in plasmid DNA-based cancer therapeutics, clinical research, and large-scale manufacturing.
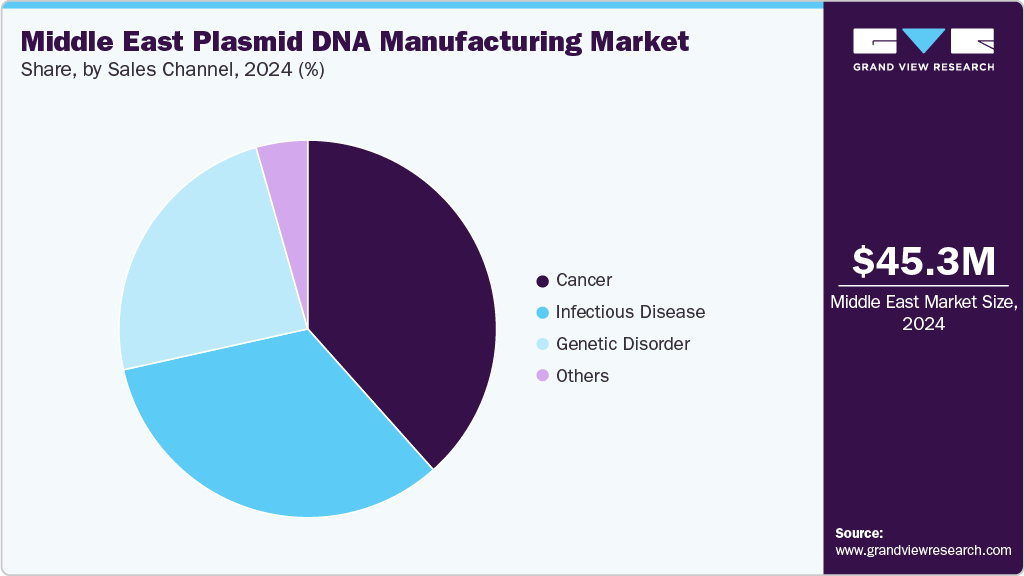
The genetic disorder segment is estimated to witness a significant CAGR of 16.91% in the Middle East plasmid DNA manufacturing industry throughout the forecast period. This growth is driven by rising demand for advanced gene therapies, increasing clinical research initiatives, and expanding access to innovative treatments. For instance, in April 2025, Fakeeh Care Group announced a strategic partnership with Fosun Pharma, a global innovation-driven healthcare leader, to accelerate healthcare innovation in Saudi Arabia and support Saudi Vision 2030, bolstering regional capabilities in plasmid-based gene therapy development.
Country Insights
Kuwait Plasmid DNA Manufacturing Market Trends
Kuwait's plasmid DNA manufacturing market is gradually growing, supported by increasing investments in biotechnology infrastructure, rising demand for advanced therapeutics, and growth in genetic research capabilities. Government support, along with partnerships between local institutions and global biotech firms, is fostering innovation and technology transfer. Increasing adoption of plasmid-based applications in vaccines, cell & gene therapies, and precision medicine is expected to strengthen Kuwait’s position in the Middle East plasmid DNA manufacturing industry over the forecast period.
UAE Plasmid DNA Manufacturing Market Trends
The UAE is a regional leader in plasmid DNA manufacturing, driven by strong government initiatives, advanced biotech infrastructure, and strategic investments in cell & gene therapies. Collaboration between global biotech firms and local research institutions, coupled with expanding GMP-certified facilities, is enhancing production capabilities. Rising demand for plasmid-based vaccines and therapeutics further strengthens the UAE’s market leadership over the forecast period.
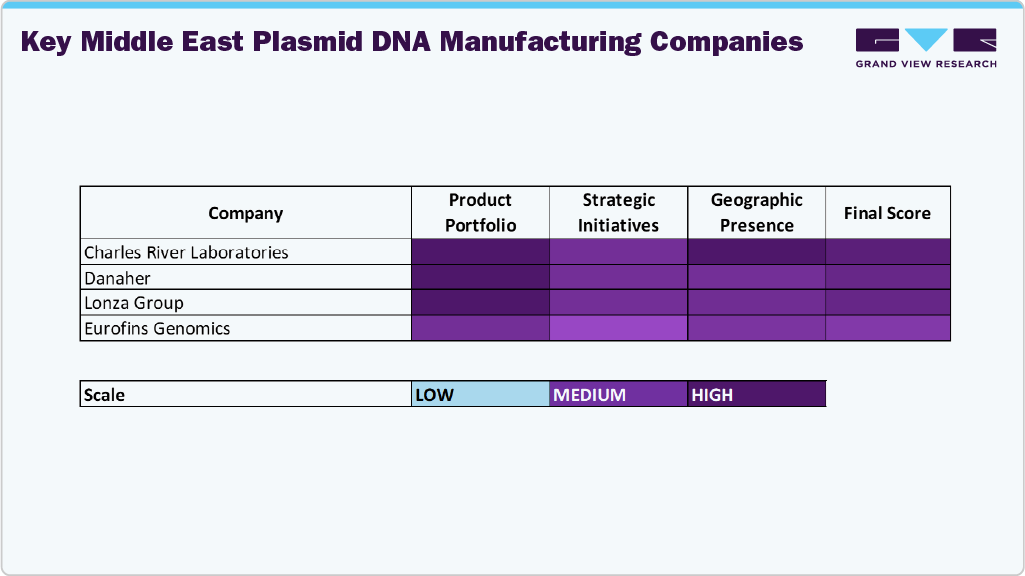
Key Middle East Plasmid DNA Manufacturing Company Insights
Key players operating in the Middle East plasmid DNA manufacturing market are undertaking various initiatives to strengthen their presence and increase the reach of their products and services. Strategies such as expansion activities and partnerships are key in propelling the market growth.
Key Middle East Plasmid DNA Manufacturing Companies:
- Charles River Laboratories
- Danaher (Aldevron)
- Lonza Group
- VGXI, Inc.
- Kaneka Corp.
- Eurofins Genomics
- Luminous BioSciences, LLC
- Cell & Gene Therapy Catapult
- Nature Technology
- Akron Biotech
Recent Developments
-
In August 2025, Saudi Arabia’s Ministry of Industry and Mineral Resources signed an MoU with Vertex Pharmaceuticals, backed by a USD 50 million investment, to localize gene therapy manufacturing, boosting regional demand for GMP-grade plasmid DNA.
-
In December 2024, Saudi Arabia’s Ministry of Industry and Mineral Resources signed an MoU with Vertex Pharmaceuticals to localize gene therapy manufacturing, boosting demand for high-quality plasmid DNA in the region’s advanced therapy sector.
-
In August 2021, INOVIO dosed the first participant in its Phase 2 trial for a DNA vaccine targeting Middle East Respiratory Syndrome (MERS), a coronavirus disease, highlighting the growing application of plasmid DNA in vaccine development.
Middle East Plasmid DNA Manufacturing Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 51.6 million
Revenue forecast in 2033
USD 186.5 million
Growth rate
CAGR of 17.43% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Grade, development phase, application, disease, and country
Country scope
Saudi Arabia; UAE; Kuwait;Qatar; Oman; Rest of ME
Key companies profiled
Charles River Laboratories; Danaher (Aldevron); Lonza Group; VGXI, Inc.; Kaneka Corp.; Eurofins Genomics; Luminous BioSciences, LLC; Cell & Gene Therapy Catapult; Nature Technology; Akron Biotech
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Middle East Plasmid DNA Manufacturing Market Report Segmentation
This report forecasts revenue growth at the regional and country levels and provides an analysis on the latest trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the Middle East plasmid DNA manufacturing market report on the basis of grade, development phase, application, disease, and country:
-
Grade Outlook (Revenue, USD Million, 2021 - 2033)
-
R&D Grade
-
Viral Vector Development
-
AAV
-
Lentivirus
-
Adenovirus
-
Retrovirus
-
Others
-
-
mRNA Development
-
Antibody Development
-
DNA Vaccine Development
-
Others
-
-
GMP Grade
-
-
Development Phase Outlook (Revenue, USD Million, 2021 - 2033)
-
Pre-Clinical Therapeutics
-
Clinical Therapeutics
-
Marketed Therapeutics
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
DNA Vaccines
-
Cell & Gene Therapy
-
Immunotherapy
-
Others
-
-
Disease Outlook (Revenue, USD Million, 2021 - 2033)
-
Infectious Disease
-
Cancer
-
Genetic Disorder
-
Others
-
-
Country Outlook (Revenue, USD Million, 2021 - 2033)
-
Saudi Arabia
-
UAE
-
Kuwait
-
Qatar
-
Oman
-
Rest of Middle East
-
Frequently Asked Questions About This Report
b. The Middle East plasmid DNA manufacturing market size was estimated at USD 45.3 million in 2024 and is expected to reach USD 51.6 million in 2025.
b. The Middle East plasmid DNA manufacturing market is expected to grow at a compound annual growth rate of 17.43% from 2025 to 2033 to reach USD 186.5 million by 2033.
b. The clinical therapeutics segment dominated the Middle East plasmid DNA manufacturing market with a share of 55.53% in 2024. This is attributable to the rising demand for plasmid DNA in advanced therapies and late-stage clinical trials.
b. Some key players operating in the Middle East plasmid DNA manufacturing market include Charles River Laboratories; Danaher (Aldevron); Lonza Group; VGXI, Inc.; Kaneka Corp.; Eurofins Genomics; Luminous BioSciences, LLC; Cell & Gene Therapy Catapult; Nature Technology; Akron Biotech.
b. Key factors driving the Middle East plasmid DNA manufacturing market growth include rising prevalence of chronic diseases, expanding cell and gene therapy adoption, increasing government investments, and advancements in biomanufacturing capabilities.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










