- Home
- »
- Clinical Diagnostics
- »
-
U.S. Prostate Cancer Biomarkers Market Size Report, 2033GVR Report cover
![U.S. Prostate Cancer Biomarkers Market Size, Share & Trends Report]()
U.S. Prostate Cancer Biomarkers Market (2025 - 2033) Size, Share & Trends Analysis Report By Type (Protein Biomarkers, Metabolomic Biomarkers), By Application (Screening & Early Detection, Companion Diagnostics), By End-use, And Segment Forecasts
- Report ID: GVR-4-68040-673-5
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2023
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
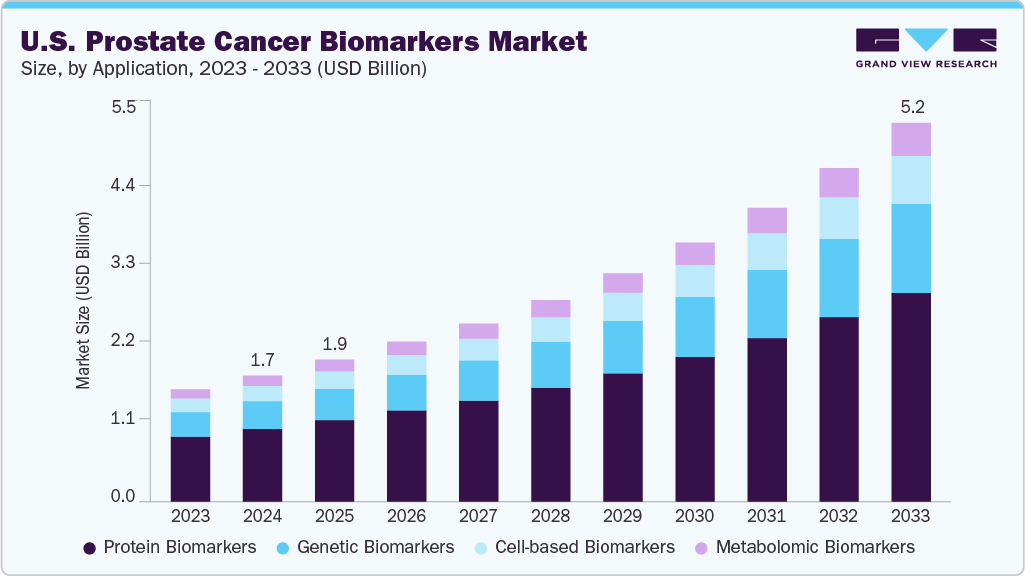
Market Size & Trends
The U.S. prostate cancer biomarkers market size was estimated at USD 1.7 billion in 2024 and is expected to grow at a CAGR of 14.74% from 2025 to 2033. The market growth is driven by the rising prevalence of prostate cancer, awareness around early detection, and adoption of precision diagnostics. Advancements in liquid biopsy, genomics, and non-invasive biomarker technologies are enhancing screening accuracy and patient stratification. Additionally, the rising demand for personalized treatment planning, along with favorable reimbursement frameworks for diagnostic tests such as PSA, PCA3, and gene-expression panels (e.g., Decipher, Oncotype DX), is further accelerating market growth. Strategic collaborations and continuous R&D investment are also expanding the biomarker pipeline and commercial availability.
Prostate cancer is one of the most prevalent malignancies affecting men in the United States. According to the American Cancer Society, 1 in 8 men will be diagnosed with the disease in their lifetime. In 2022 alone, an estimated 268,500 new cases were reported, with approximately 34,500 deaths, making it the second leading cause of cancer death among American men. Today, over 3.1 million men in the U.S. are living with prostate cancer. This substantial patient population, along with limitations in traditional diagnostics such as PSA testing, which lacks specificity to distinguish between benign and malignant conditions, has created strong demand for novel biomarkers that can enable earlier detection, improved risk stratification, and treatment personalization.
Genomic Risk Assessment & Market Entry: OncoAssure
On April 28, 2025, OncoAssure, an Ireland-based medtech company, launched its OncoAssure Prostate Test in the U.S. during the American Urological Association (AUA) 2025 Annual Meeting. This next-generation prognostic test is designed for men diagnosed with localized prostate cancer and can be used post-biopsy or post-surgery. The test integrates genomic analysis of “Master Driver” genes with clinical parameters, including the CAPRA score, providing a precise estimate of disease aggressiveness and recurrence risk. This approach helps clinicians distinguish between indolent and aggressive cancers, ultimately reducing unnecessary overtreatment.
OncoAssure has partnered with Diacarta Inc., which will conduct testing through its ISO and CAP/CLIA-certified lab in Pleasanton, California. The company also announced its plans for an investment round in 2025 to support commercialization and pipeline expansion, including the development of a melanoma prognostic assay.
AI-Enabled Imaging Biomarkers: Quibim’s QP-Prostate® CAD
In March 2025, Quibim secured FDA 510(k) clearance for its QP-Prostate CAD, an AI-powered platform designed to improve lesion detection on prostate MRI scans. Trained using actual biopsy-confirmed pathology data, the platform enhances diagnostic accuracy, reduces interpretation variability, and generates 3D lesion maps for improved visualization.
QP-Prostate has already been approved in the EU (CE mark), UK (UKCA), and Australia (TGA) and is being used in leading institutions like Hospices Civils de Lyon and Hospital Universitario Ramón y Cajal. With rising demand for prostate MRI and increasing radiologist workloads in the U.S., Quibim’s solution addresses a critical bottleneck by accelerating image interpretation and supporting early intervention.
Multiplex Biomarker Panels: Randox Biochip Technology
Randox Laboratories is advancing prostate cancer diagnostics through its proprietary Biochip Array Technology, which enables simultaneous analysis of multiple biomarkers. Their panel includes novel PSA isoforms such as pro-PSA, 2proPSA, and Nicked PSA, in addition to inflammatory markers including IL-8, EGF, and MCP-1. Studies demonstrate that this multi-marker panel significantly improves diagnostic accuracy compared to total PSA alone, offering better discrimination between benign prostatic hyperplasia and malignancy.
AI-Driven Biomarker Discovery: Lantheus & PCCTC Partnership
In January 2022, Lantheus Holdings, through its subsidiary EXINI Diagnostics, partnered with the Prostate Cancer Clinical Trials Consortium (PCCTC) to integrate its AI platforms into clinical research. These platforms include:
-
aPROMISE: a PSMA PET/CT imaging solution (FDA cleared in 2021) that quantifies tumor burden.
-
aBSI: a bone scan index system (FDA cleared in 2019) that detects and quantifies skeletal metastases.
These platforms will be used in early-phase PCCTC trials to evaluate response to therapy and assist in patient selection, signaling the growing role of imaging biomarkers in therapeutic decision-making.
Molecular Imaging Breakthroughs: Clarity’s 64Cu-SAR-bisPSMA
Clarity Pharmaceuticals is advancing precision diagnostics with its PSMA-targeted PET imaging agent, 64Cu-SAR-bisPSMA. The company received Fast Track Designation (FTD) from the FDA in August 2024 for patients with suspected metastasis and a second FTD in January 2025 for patients with biochemical recurrence (BCR) following initial therapy.
The designations are based on data from the Phase 1/2 COBRA trial (NCT05249127), which demonstrated:
-
Lesion detection in up to 80% of patients who had negative or inconclusive standard imaging
-
A change in clinical treatment plan in 48% of patients
Clarity plans to initiate its pivotal Phase 3 AMPLIFY trial in October 2024, enrolling 220 patients with rising PSA levels post-treatment to confirm diagnostic performance and support FDA approval.
Market Concentration & Characteristics
The degree of innovation in prostate cancer biomarkers is advancing rapidly, driving improvements in early detection, risk assessment, and personalized treatment strategies. Emerging biomarkers such as PCA3, TMPRSS2-ERG fusion, and the Prostate Health Index (PHI) enhance diagnostic accuracy beyond traditional PSA testing. Innovations in liquid biopsy technologies, such as circulating tumor cells (CTCs) and extracellular vesicles, provide non-invasive options for real-time monitoring of disease progression. In addition, artificial intelligence (AI)-driven biomarker analysis is refining predictive models, enabling more precise identification of aggressive versus indolent prostate cancer. These advancements are transforming clinical decision-making, optimizing therapeutic approaches, and improving patient outcomes while reducing unnecessary interventions.
The level of mergers and acquisitions (M&A) in the U.S. prostate cancer biomarkers industry is moderate but steadily rising as companies seek to expand their diagnostic capabilities and strengthen their biomarker portfolios. Several diagnostic and biotech firms engage in strategic acquisitions to access novel biomarker technologies, proprietary assays, and advanced molecular platforms. Collaborations between diagnostic and precision medicine firms are also shaping the competitive landscape, reflecting a growing interest in integrated biomarker solutions for prostate cancer. While large-scale acquisitions remain limited, targeted M&A efforts increasingly focus on enhancing expertise in genomics, liquid biopsy, and AI-based analytics for prostate cancer diagnostics.
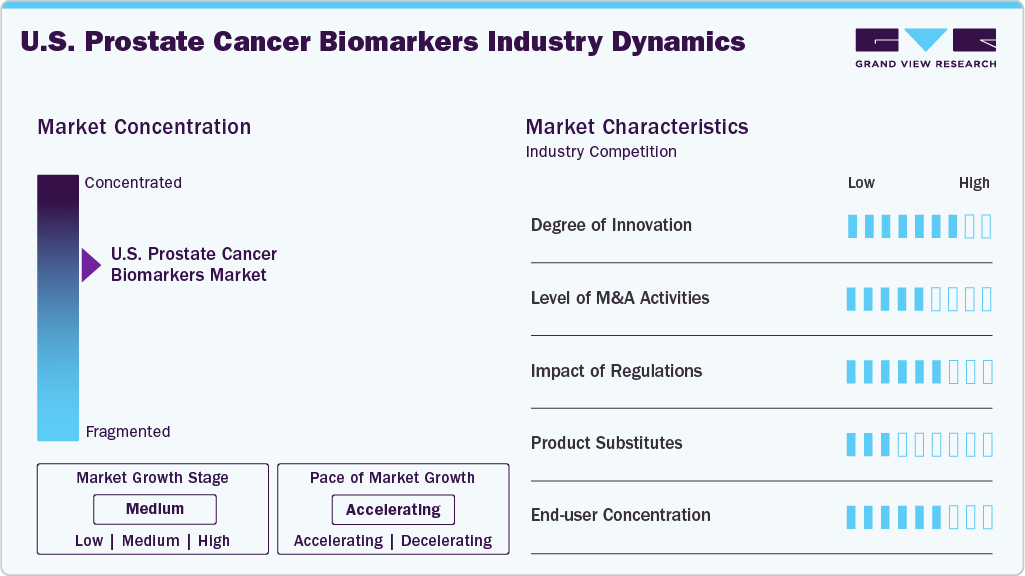
Regulatory frameworks significantly influence the development, approval, and commercialization of prostate cancer biomarkers. Authorities such as the FDA (U.S.), EMA (Europe), and PMDA (Japan) enforce rigorous standards to ensure biomarker accuracy, analytical reliability, and clinical relevance. The FDA’s Biomarker Qualification Program offers a structured path for biomarker acceptance but requires robust evidence, which can extend the time to market.
The U.S. prostate cancer biomarkers market faces competition from several conventional and emerging diagnostic approaches. Traditional methods such as digital rectal examination (DRE), prostate-specific antigen (PSA) testing, and multiparametric MRI (mpMRI) continue to serve as primary tools in prostate cancer screening and diagnosis. These techniques, while accessible and cost-effective, often lack the precision of newer biomarker-based tests. In addition, tissue biopsies and genomic profiling, when used without biomarker integration, can offer diagnostic insights but may not fully support risk stratification or treatment personalization. In cost-sensitive healthcare systems, the reliance on these established tools may limit the adoption of newer biomarker technologies, particularly where reimbursement frameworks are not well aligned with advanced diagnostics.
The U.S. prostate cancer biomarkers industry is expanding geographically due to rising investments in cancer diagnostics, increasing awareness of early detection, and ongoing research efforts. In North America, the use of biomarker-based tests is supported by established screening programs, access to advanced technologies, and regulatory initiatives. Europe is witnessing steady growth through government-backed cancer research and public health strategies. In Asia-Pacific, awareness and adoption of these tests are growing as healthcare systems improve and research activities increase in countries such as China, Japan, and India. Latin America and the Middle East are gradually adopting biomarker diagnostics with support from improvements in healthcare infrastructure and focused efforts to enhance cancer detection, contributing to broader market growth across regions.
Type Insights
The protein biomarkers segment dominated the market, accounting for a share of 57.60% in 2024. Protein biomarkers play a critical role in prostate cancer diagnostics by facilitating early detection and tracking disease progression through the identification of abnormal protein expression patterns. The growing emphasis on personalized medicine and targeted treatment strategies is accelerating the adoption of protein-based diagnostic solutions. Advances in technologies such as mass spectrometry and immunoassays have significantly enhanced the precision and throughput of protein analysis in biological fluids, including blood and urine. These innovations are driving wider clinical integration and research use, further solidifying the segment’s impact within the prostate cancer biomarker market.
The metabolomic biomarkers segment is expected to grow at the fastest CAGR of 15.79% over the forecast period. Rising interest in the metabolic alterations linked to prostate cancer is fueling the segment’s growth. These biomarkers offer valuable insights into disease mechanisms by profiling small molecules in biofluids such as urine, serum, and plasma. They hold promise for early detection and for differentiating between aggressive and indolent cancer types. Technological advancements in analytical platforms, including NMR spectroscopy and mass spectrometry, have enhanced the sensitivity and accuracy of metabolic profiling. Additionally, increased investment in precision oncology and the expanding application of metabolomics in research and clinical trials are further driving this segment’s growth.
Application Insights
The screening and early detection segment dominated the market, accounting for a share of 45.01% in 2024. The growth is driven by the increasing need for early detection of prostate cancer to enhance treatment outcomes and reduce overall disease burden. Biomarkers are crucial in identifying cancer at an asymptomatic stage, enabling timely and less invasive therapeutic interventions. The integration of biomarker-based tests into routine health check-ups and screening programs is becoming more widespread across hospitals and diagnostic laboratories. Additionally, advancements in non-invasive technologies, including liquid biopsies and multiplex assays, are improving diagnostic accuracy and patient compliance. These innovations are accelerating the adoption of biomarkers in prostate cancer screening and early diagnosis.
The companion diagnostics segment is expected to grow at the fastest CAGR of 14.5% over the forecast period. This segment is gaining momentum as the demand for personalized treatment approaches in prostate cancer management continues to rise. Companion diagnostics enable the identification of patients most likely to respond to specific targeted therapies by analyzing distinct biomarker profiles. These tools enhance treatment precision and minimize the risk of adverse effects by informing therapeutic choices. Ongoing advancements in molecular diagnostics, coupled with supportive regulatory frameworks for precision medicine, are accelerating the development and approval of companion diagnostic assays. As the pipeline of targeted therapies expands, the need for reliable biomarker-driven companion diagnostics is expected to grow, fueling this segment’s continued growth.
End-use Insights
The hospitals and diagnostic laboratories segment dominated the U.S. prostate cancer biomarkers industry, accounting for a share of 63.11% in 2024. Hospitals and diagnostic laboratories play a pivotal role in the adoption and implementation of prostate cancer biomarkers, managing high volumes of diagnostic testing and patient monitoring. Their involvement in early detection, treatment planning, and ongoing disease surveillance is driving increased utilization of biomarker-based diagnostics. Advances in clinical workflows and access to more sophisticated testing platforms have enhanced the speed and accuracy of biomarker analysis. Moreover, the growing emphasis on personalized care and the integration of molecular diagnostics into standard clinical practice continue to reinforce the significance of this segment.
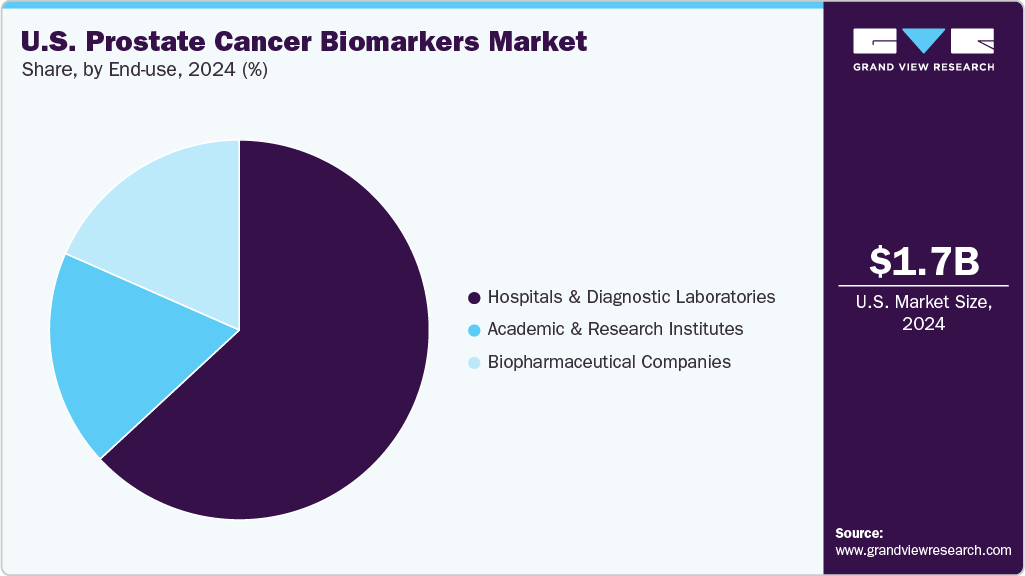
The academic and research institutes segment is expected to grow at the fastest CAGR of 15.4% over the forecast period. Academic and research institutions play a critical role in the discovery and validation of prostate cancer biomarkers, driving progress in translational research and clinical innovation. Leveraging robust academic networks and dedicated research funding, these institutions lead collaborative studies and clinical trials that advance biomarker development. The growing emphasis on personalized medicine and precision diagnostics is accelerating the integration of novel biomarkers into academic research frameworks. Additionally, strategic partnerships with healthcare providers and biotechnology companies are facilitating the translation of research findings into clinically applicable diagnostic solutions, contributing to the sustained growth of this segment.
Country Insights
The U.S. accounted for a substantial share of the North America prostate cancer biomarkers market in 2024, driven by advanced healthcare infrastructure, significant research funding, and the increasing adoption of biomarker-based diagnostics. The market is bolstered by high awareness and institutional support for early detection and precision medicine in prostate cancer care. Research organizations and academic institutions actively engage in studies to validate novel biomarkers, enhancing clinical confidence in these diagnostic tools.
For instance, in February 2022, Datar Cancer Genetics Inc. received the U.S. Food and Drug Administration's Breakthrough Device Designation for its 'TriNetra-Prostate' blood test, designed to detect early-stage prostate cancer. This test demonstrated high accuracy (>99%) without false positives and requires only a 5 mL blood sample. It is indicated for males aged 55-69 years with serum PSA of 3 ng/mL or higher. The test identifies prostate adenocarcinoma-specific Circulating Tumor Cells (CTCs) in the blood, potentially reducing the need for invasive biopsies among individuals with benign prostate conditions and improving detection rates among those with prostate cancer.
Key U.S. Prostate Cancer Biomarkers Company Insights
Major market players engage in various strategies, such as distribution agreements, mergers and acquisitions, and expansions. Most crucially, they exhibit a high degree of innovation in product research and development to improve their market penetration.
Key U.S. Prostate Cancer Biomarkers Companies:
- Exact Sciences Corporation
- Myriad Genetics, Inc.
- Bio-Techne
- ExoDx
- OPKO Health, Inc.
- mdxhealth
- Veracyte, Inc.
- Beckman Coulter, Inc.
- Nucleix
- DiaCarta
Recent Developments
-
In February 2025, Myriad Genetics partnered with PATHOMIQ to exclusively license PATHOMIQ_PRAD, an AI-driven technology platform for prostate cancer, in the U.S. This partnership integrates AI-enabled diagnostics into Myriad's oncology portfolio, supporting more informed treatment decisions before and after prostate cancer therapy. By leveraging advanced artificial intelligence, the collaboration aims to strengthen diagnostic precision in prostate cancer care, aligning with evolving needs in the prostate cancer biomarker market.
-
In February 2024, DiaCarta entered a strategic collaboration with OncoAssure Ltd to commercialize a prostate cancer prognostic test. This 6-gene expression assay evaluates the risk of aggressive disease following diagnosis and estimates the probability of biochemical recurrence within five years post-surgery. The partnership utilizes DiaCarta's clinical diagnostic capabilities to support validation and market expansion of the test, contributing to more tailored prostate cancer management through biomarker-based risk stratification.
U.S. Prostate Cancer Biomarkers Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 1.95 billion
Revenue forecast in 2033
USD 5.21 billion
Growth rate
CAGR of 14.74% from 2025 to 2033
Base year for estimation
2024
Historical data
2021 - 2023
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, application, end-use
Key companies profiled
Exact Sciences Corporation; Myriad Genetics, Inc.; Bio-Techne; ExoDx; OPKO Health, Inc.; mdxhealth; Veracyte, Inc.; Beckman Coulter, Inc.; Nucleix; DiaCarta
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Prostate Cancer Biomarkers Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest industry trends in each of the sub-markets from 2021 to 2033. For this study, Grand View Research has segmented the U.S. prostate cancer biomarkers market report based on type, application, and end-use:
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Genetic Biomarkers
-
Protein Biomarkers
-
Cell-based Biomarkers
-
Metabolomic Biomarkers
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Screening and Early Detection
-
Diagnosis and Risk Stratification
-
Prognosis and Treatment Monitoring
-
Companion Diagnostics
-
-
End-use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals and Diagnostic Laboratories
-
Academic and Research Institutes
-
Biopharmaceutical Companies
-
Frequently Asked Questions About This Report
b. The U.S. prostate cancer biomarker market size was estimated at USD 1.73 billion in 2024 and is expected to reach USD 1.95 billion in 2025.
b. The U.S. prostate cancer biomarker market is expected to grow at a compound annual growth rate of 12.8% from 2025 to 2033 to reach USD 5.21 billion by 2033.
b. The protein biomarkers segment dominated the U.S. prostate cancer biomarker market and accounted for the largest revenue share of 57.60% in 2024.
b. Some key players operating in the prostate cancer biomarker market include Exact Sciences Corporation; Myriad Genetics, Inc.; Bio-Techne; ExoDx; OPKO Health, Inc.; mdxhealth; Veracyte, Inc.; and Beckman Coulter, Inc
b. Key factors that are driving the prostate cancer biomarker market growth include rising incidence of prostate cancer and increasing awareness among physicians & patients regarding early detection. Furthermore, advancements in biomarker technologies and growing research efforts for prostate cancer diagnosis & treatment are further propelling the overall market.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










