- Home
- »
- Medical Devices
- »
-
Diabetes Clinical Trials Market Size, Industry Report, 2033GVR Report cover
![Diabetes Clinical Trials Market Size, Share & Trends Report]()
Diabetes Clinical Trials Market (2026 - 2033) Size, Share & Trends Analysis Report By Phase (Phase I, Phase II, Phase III, Phase IV), By Study Design, By Indication, Indication By Study Design, By Service (Protocol Designing, Site Identification), By Sponsor, By Region, And Segment Forecasts
- Report ID: GVR-4-68040-910-5
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
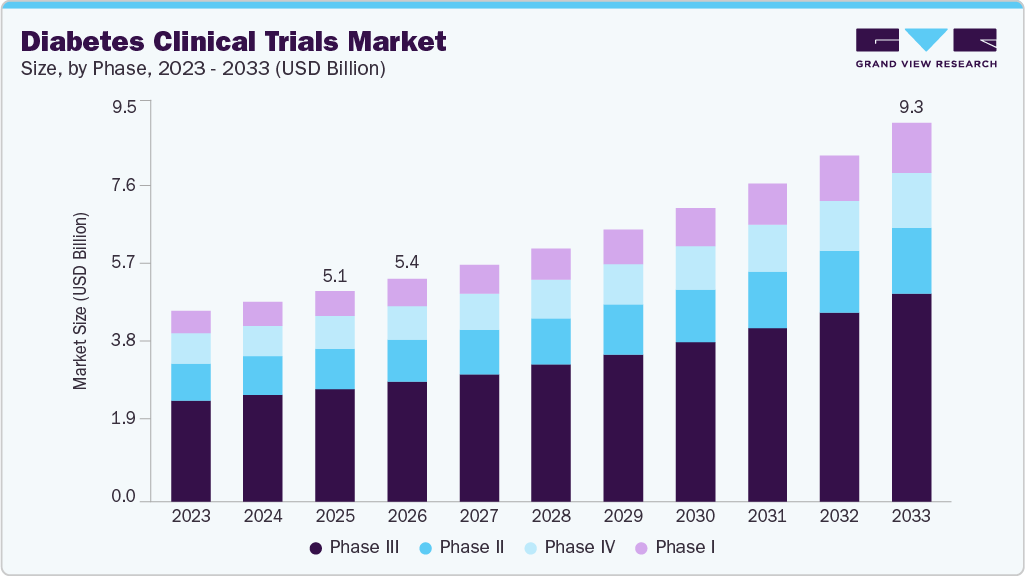
Diabetes Clinical Trials Market Summary
The global diabetes clinical trials market size was estimated at USD 5.1 billion in 2025 and is projected to reach USD 9.3 billion by 2033, growing at a CAGR of 7.9% from 2026 to 2033. The market is driven by the increasing prevalence of diabetes and its related complications, the growing expansion of clinical trials, and rising R&D investments by pharmaceutical and biotech companies.
Key Market Trends & Insights
- North America diabetes clinical trials market held the largest share of 50.8% of the global market in 2025.
- The diabetes clinical trials industry in the U.S. accounted for the highest market share in North America.
- By phase, the phase III segment held the largest market share of 53.6% in 2025.
- Based on the study design, the interventional trials segment held the largest market share in 2025.
- By indication, the type 2 diabetes mellitus (T2DM) segment held the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 5.1 Billion
- 2033 Projected Market Size: USD 9.3 Billion
- CAGR (2026-2033): 7.9%
- North America: Largest market in 2025
- Asia Pacific:Fastest growing market
Some other factors contributing to market growth include sedentary lifestyles, an aging population, and changes in dietary habits, all of which have escalated the demand for innovative therapeutic options. This has led to increased investment in clinical research aimed at enhancing glycemic control, minimizing complications, and improving patient outcomes.The market growth is attributed to the rising demand for more advanced diabetic treatments, particularly in areas such as personalized medicine, combination therapies, and long-acting insulin formulations. The industry is also shifting towards a more patient-centric approach to treatment, with an increased focus on managing comorbidities, such as cardiovascular and renal issues, further boosting investments in clinical trials. In addition, support from regulatory bodies and increased funding from pharmaceutical and biotechnology companies are aiding in the market's growth.
Moreover, shifting trend towards decentralized and hybrid clinical trial models that enhance patient recruitment and retention rates. The incorporation of digital health technologies, such as wearable glucose monitors and remote patient monitoring systems, is improving data accuracy and overall trial efficiency. Emerging markets are also becoming attractive locations for clinical trials due to their cost-effectiveness and diverse patient populations.
Furthermore, technological advancements are expected to drive the market, with the integration of artificial intelligence, big data analytics, and real-world evidence significantly influencing trial design and execution. Innovations such as continuous glucose monitoring systems, closed-loop insulin delivery, and new drug classes, such as GLP-1 receptor agonists and SGLT2 inhibitors, are transforming treatment approaches. In addition, companies are increasingly forming collaborations, partnerships, and mergers to enhance their clinical pipelines and expand their global reach. The focus on innovation strategies aims to accelerate time-to-market, optimize trial protocols, and leverage precision medicine. Thus, the diabetes clinical trials market is expected to see new growth opportunities driven by technological innovation, evolving treatment paradigms, and a strong commitment to improving long-term patient outcomes. Such factors are expected to drive the market.
Opportunity Analysis
The diabetes clinical trials market is experiencing new growth opportunities attributed to the increasing global prevalence of both type 1 and type 2 diabetes, along with rising complications like cardiovascular and renal disorders. Besides, expanding patient pools in emerging economies creates a strong demand for large-scale, diverse clinical trials. In addition, the shift toward precision medicine is paving the way for targeted therapeutics, especially in genetically stratified populations.
Biopharmaceutical companies are increasingly outsourcing clinical trial activities to contract research organizations (CROs) to reduce costs and speed up timelines, creating opportunities for specialized service providers. The integration of digital health technologies, such as wearable devices and remote monitoring systems, enhances patient recruitment and retention, improving trial efficiency. In addition, a growing pipeline of novel therapeutics, including GLP-1 receptor agonists and cell-based treatments, is expanding the clinical research landscape.
Moreover, regulatory harmonization and adaptive trial designs present opportunities to expedite approvals and mitigate development risks. Strategic collaborations among pharmaceutical firms, academic institutions, and technology providers further boost innovation capabilities. Overall, the market is poised for strong expansion, supported by technological advancements, unmet clinical needs, and increasing investment in diabetes therapeutics development.
Impact of U.S. Tariffs on Diabetes Clinical Trials Market
U.S. tariffs on pharmaceutical inputs, medical devices, and laboratory equipment moderately impact the diabetes clinical trials market. Increased import costs for specialized equipment and trial materials raise operational expenses for sponsors and CROs, potentially leading to budget reallocations and delays in trial timelines. These tariffs also affect global supply chains, prompting companies to diversify sourcing strategies or shift operations to more cost-effective regions like the Asia-Pacific. This geographic shift may accelerate the globalization of diabetes clinical trials but introduces regulatory compliance and quality assurance challenges. Moreover, higher costs could deter smaller biotech firms from launching early-stage trials, impacting innovation pipelines. However, larger pharmaceutical companies may mitigate these challenges through strategic partnerships and long-term supplier agreements. Thus, CROs are optimizing operations and utilizing decentralized trial models to manage cost pressures effectively.
Technological Advancements
Technological advancements are transforming the diabetes clinical trials industry by enhancing efficiency, accuracy, and patient engagement. A significant innovation in this space is the use of artificial intelligence (AI) and machine learning, which streamline the patient recruitment process, optimize protocol design, and facilitate predictive analytics. These technologies help identify suitable patient populations, minimize trial failures, and expedite decision-making, ultimately reducing development timelines and costs. Besides, a prominent advancement is the adoption of decentralized clinical trials (DCTs), which allow for remote participation via telemedicine, mobile applications, and digital platforms. This model broadens access to diverse patient populations, increases recruitment rates, and improves patient retention by diminishing the need for frequent site visits. As a result, DCTs are becoming more prevalent in diabetes studies that require long-term monitoring.

Wearable devices and continuous glucose monitoring (CGM) systems have also revolutionized data collection by offering real-time, high-frequency patient data. These tools enhance the accuracy of clinical endpoints, improve patient adherence to protocols, and enable a more thorough evaluation of therapy outcomes. They also decrease dependence on in-clinic visits, reinforcing cost efficiency and convenience.
Moreover, big data analytics facilitates the integration of varied datasets, including electronic health records, genomic data, and real-world evidence. This integration leads to improved trial design, enhanced risk assessment, and more precise outcome predictions. Advanced biomarkers and precision medicine strategies are enabling targeted therapies that improve treatment efficacy and decrease adverse effects. Thus, these technological innovations are reshaping the landscape of diabetes clinical trials by fostering more adaptive, patient-centric, and data-driven methods, thus enhancing success rates and the overall efficiency of clinical development programs.
Pricing Model Analysis
The pricing models in the diabetes clinical trials market are structured to balance cost predictability, operational flexibility, and risk-sharing between sponsors and contract research organizations (CROs). The fixed price model is a common choice for trials with clearly defined protocols and timelines, providing sponsors with budget certainty and simplifying financial planning. However, it often lacks the flexibility needed to handle protocol amendments or unforeseen challenges during trial operations. On the other hand, the time-and-materials model allows sponsors to pay based on actual resources used and time spent, making it well-suited for more complex or adaptive diabetes trials. This approach offers greater flexibility but requires careful cost monitoring, as expenses can increase if the project's scope expands. This model is particularly relevant in early-stage or exploratory studies, where uncertainty is higher. Moreover, a milestone-based pricing model, in which payments are linked to the achievement of specific clinical or operational milestones, such as meeting patient recruitment targets or completing trial phases. This method promotes accountability and aligns the incentives of sponsors and CROs, ensuring that outcomes are delivered on time while maintaining financial oversight.
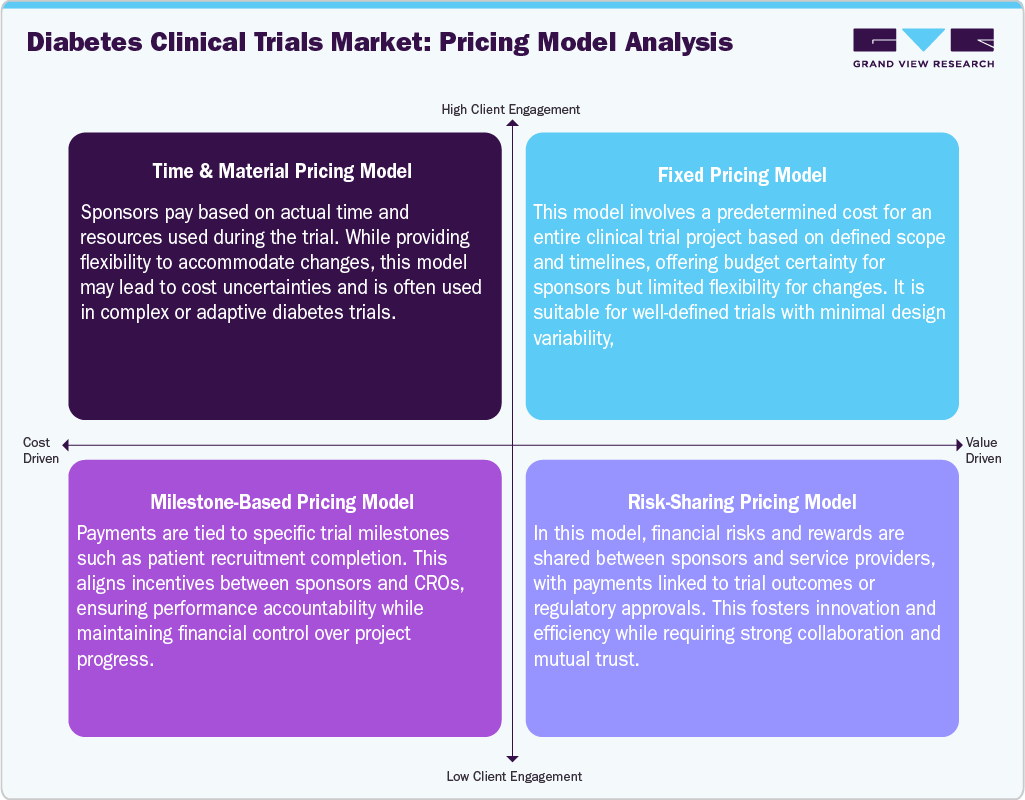
Furthermore, the risk-sharing model drives a collaborative approach, wherein both parties share the financial risks and rewards. In this model, payments may be contingent on trial success, regulatory approvals, or performance outcomes. It encourages innovation and efficiency, particularly in trials focused on novel therapies or those characterized by significant uncertainty. Thus, many organizations implement hybrid pricing strategies that integrate aspects of these models to enhance cost efficiency and flexibility. The selection of a suitable pricing model ultimately hinges on the complexity of the trial, the nature of therapeutic innovations, and the strategic objectives of stakeholders involved in the diabetes clinical trials market.
Market Concentration & Characteristics
The market growth stage is medium, and the pace of the market growth is accelerating. The diabetes clinical trials market is characterized by evolving technologies and therapeutics, regulatory considerations, growing need for advanced therapeutics, increasing demand for personalized medicine, and increasing outsourcing of development processes to leverage cost advantages and specialized capabilities.
The market showcases high innovation driven by advancements in drug development, including GLP-1 therapies and emerging therapies. AI, digital health tools, and real-world data integration enhance trial efficiency and personalize treatment approaches.
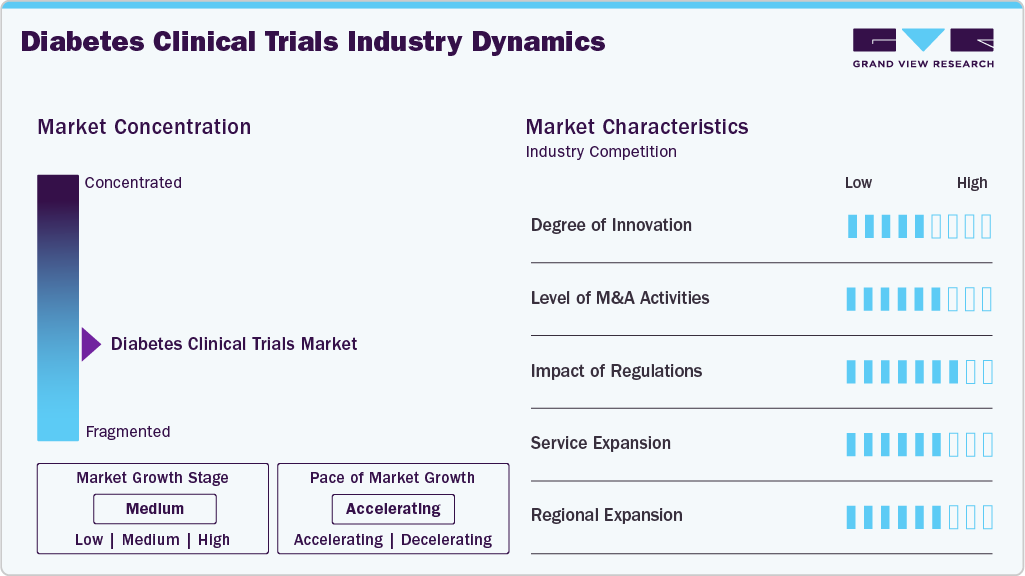
Stringent regulatory requirements regarding patient safety and trial transparency significantly impact operational processes, increasing compliance costs but ensuring high-quality outcomes. Companies with robust regulatory expertise gain competitive advantages.
Mergers and acquisitions are on the rise as companies seek to expand capabilities and access advanced technologies. Strategic collaborations and acquisitions of niche CROs and biotech firms bolster global presence and execution capabilities.
The market is moderately fragmented, with numerous global and regional CROs, pharmaceutical companies, and research institutions in competition. Specialized service providers focusing on diabetes therapeutics contribute to competitive intensity.
Key end users include pharmaceutical companies, biotech firms, medical device manufacturers, and academic institutions, whose growing demand for efficient and high-quality clinical trials significantly influences market dynamics and pricing strategies.
Phase Insights
The phase III segment held the largest market share, accounting for a revenue share of 53.6% in 2025. The segment is driven by the phase III critical role in confirming efficacy, monitoring adverse reactions, and supporting regulatory approvals. These large-scale, multicenter studies involve diverse patient populations, making them essential for commercialization strategies. The increasing number of late-stage pipeline drugs for diabetes management, particularly for Type 2 diabetes, is driving demand for Phase III trials. In addition, strong investments from pharmaceutical companies and the need for robust clinical evidence to meet stringent regulatory standards further drive the segment growth.
The phase I segment is expected to grow significantly during the forecast period. The segment's growth is driven by rising innovation in drug discovery and early-stage biologics. These trials primarily focus on safety, tolerability, and pharmacokinetics in small patient groups or healthy volunteers. Increasing investments in novel therapies such as gene therapies, peptide-based drugs, and advanced insulin formulations are accelerating early-phase research. Moreover, the growing presence of biotech startups and increased funding for experimental diabetes treatments are contributing to the rapid expansion of Phase I clinical trials globally. Thus, these factors are expected to increase over the estimated period.
Study Design Insights
On the basis of study design, the interventional trials segment accounted for the largest share in 2025. The segment is driven by the increased involve active testing of new drugs, devices, or treatment protocols. These studies are essential for evaluating the effectiveness and safety of therapeutic interventions under controlled conditions. The growing prevalence of diabetes and the need for innovative treatment options, including combination therapies and advanced insulin delivery systems, are fueling demand for interventional trials. In addition, regulatory requirements mandate extensive interventional data, further strengthening the segment growth over the forecast period.
The observational trials segment is expected to grow significantly during the forecast period. The growth is fueled by the increasing need for real-world evidence and long-term disease monitoring. These studies analyze patient outcomes without intervention, offering insights into treatment patterns, disease progression, and patient behavior. The growing adoption of digital health technologies, wearable devices, and electronic health records is enabling large-scale data collection. Pharmaceutical companies and research organizations are increasingly leveraging observational studies to complement clinical trial data, support post-marketing surveillance, and enhance decision-making in diabetes care.
Indication Insights
The type 2 diabetes mellitus (T2DM) segment dominated the market with the largest revenue share in 2025. The segment is driven by the higher prevalence of type 2 diabetes compared to other forms. Besides, the rising incidence linked to sedentary lifestyles, obesity, and aging populations has created a substantial demand for effective therapeutics. In addition, most pharmaceutical companies are increasingly investing in T2DM drug development, including GLP-1 receptor agonists and SGLT2 inhibitors. Moreover, a large patient pool and the ongoing need for improved treatment outcomes continue to drive segment growth in clinical research. Such factors are expected to drive segment growth.
On the other hand, the type 1 diabetes mellitus (T1DM) segment is projected to grow at a significant CAGR during the forecast period. The segment is driven by increasing research into innovative therapies such as immunotherapy, stem cell therapy, and artificial pancreas systems. Although T1DM has a smaller patient base, the unmet need for disease-modifying treatments is significant. Advancements in biotechnology and growing interest in precision medicine are accelerating clinical trials in this segment. In addition, supportive regulatory pathways and funding for rare and autoimmune diseases are contributing to the rapid expansion of T1DM-focused research initiatives. Such factors are expected to drive the market growth.
Service Insights
On the basis of service, the laboratory services segment accounted for the largest share in 2025 during the forecast period. The segment growth is attributed to fundamental trial execution, including biomarker analysis, sample testing, and data validation. These services ensure the accuracy and reliability of clinical outcomes, making them indispensable across all trial phases. Besides, the increasing complexity of clinical trials, along with the demand for advanced diagnostic techniques and centralized laboratory operations, is driving growth. Furthermore, the integration of automation and digital technologies is enhancing efficiency and scalability in laboratory services.
The patient recruitment segment is expected to grow significantly during the forecast period. The growth is attributed to the increasing complexity of clinical trials and the need for diverse patient populations. Recruiting eligible participants remains one of the most challenging aspects of diabetes trials, particularly for long-duration studies. The adoption of digital recruitment platforms, social media outreach, and AI-driven patient matching is transforming this segment. In addition, decentralized clinical trials and remote monitoring solutions are improving patient accessibility, thereby accelerating enrollment rates. Such factors are expected to drive new growth opportunities over the estimated period.
Sponsor Insights
The pharmaceutical & biopharmaceutical companies segment accounted for the largest share in 2025. The segment growth is driven by their strong focus on developing innovative diabetes therapies. Besides, increasing R&D investments, expanding drug pipelines, and strategic collaborations with contract research organizations are driving growth. These companies are actively exploring novel drug classes, biologics, and combination therapies to address unmet clinical needs. In addition, the competitive landscape and pressure to bring differentiated products to market are further accelerating their involvement in diabetes clinical trials globally. Thus, these factors are expected to drive the market.
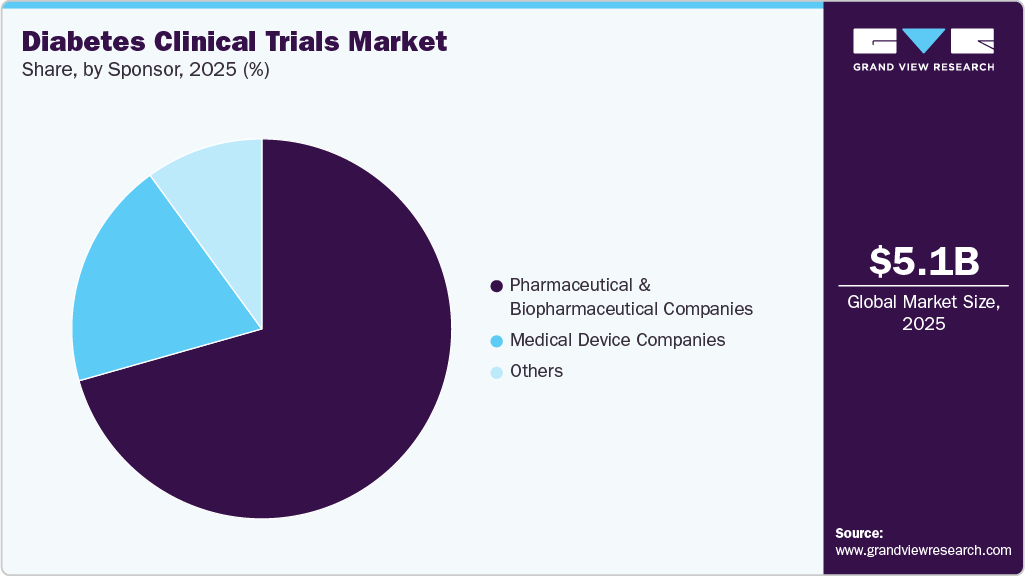
The medical device companies segment is expected to grow significantly during the forecast period. The growth is attributed to advancements in diabetes management technologies such as continuous glucose monitoring systems and insulin delivery devices. These companies are increasingly conducting clinical trials to validate the safety and effectiveness of innovative devices. The growing adoption of digital health solutions and integration with mobile applications is enhancing patient engagement and treatment outcomes. In addition, rising demand for minimally invasive and user-friendly devices is driving increased clinical research activity in this segment. Such factors are expected to drive new growth opportunities over the estimated period.
Regional Insights
The North America diabetes clinical trials market dominated the global market in 2025, accounting for a revenue share of 50.8%. The market growth is attributed to the presence of an advanced biopharmaceutical ecosystem, high R&D spending, and the strong presence of global pharmaceutical and biotechnology companies. Besides, the region benefits from a well-established clinical research infrastructure, streamlined regulatory pathways, and early adoption of advanced technologies such as decentralized trials and digital health integration. In addition, increasing outsourcing of complex biologics and rising demand for innovative diabetes therapies further support market expansion. Moreover, strong collaborations between CROs, sponsors, and research institutions enhance trial efficiency and accelerate drug development timelines. Such factors are expected to drive the market growth.
U.S. Diabetes Clinical Trials Market Trends
The diabetes clinical trials industry in the U.S. accounted for the highest market share in North America, owing to strong innovation capabilities, robust funding environment, and concentration of leading biopharma companies. Besides, high clinical trial activity is supported by advanced healthcare infrastructure, access to diverse patient populations, and rapid adoption of digital trial technologies. In addition, regulatory support from agencies such as the FDA enables faster approvals and commercialization. Moreover, increasing investments in precision medicine, biologics, and novel diabetes therapeutics continue to drive demand for clinical trials, reinforcing the country's leadership in global diabetes research. Such factors are expected to drive the U.S. market.
The Canada diabetes clinical trials market is expected to grow at a significant CAGR during the forecast period. The market is driven by the rapidly expanding biotechnology sector and favorable government initiatives. Strong funding programs and regulatory support encourage innovation and collaboration between academic institutions, CROs, and pharmaceutical companies. The country's emphasis on research-driven development and clinical excellence enhances its attractiveness for early-phase and late-stage trials. In addition, investments in advanced clinical infrastructure and increased participation in global multicenter trials are expected to drive demand for diabetes-related research over the estimated time period.
Europe Diabetes Clinical Trials Market Trends
The diabetes clinical trials market in Europe is driven by strong regulatory expertise, advanced healthcare systems, and a well-integrated biopharmaceutical industry. Countries such as Germany, the UK, and France play a pivotal role in driving innovation and clinical research. The region benefits from harmonized regulatory frameworks and high standards of Good Manufacturing Practices (GMP), ensuring quality and compliance. Besides, the increasing demand for biologics, coupled with rising collaborations between research institutions and industry players, is accelerating clinical development and strengthening Europe's position in global diabetes trials.
The Germany diabetes clinical trials market held the largest share in 2025. Germany is one of the leading hubs for diabetes clinical trials, supported by a strong industrial base, advanced research infrastructure, and a highly skilled workforce. The country is a key hub for clinical research and biologics development, supported by significant investments in R&D and innovation. Besides, demand for advanced therapies, including monoclonal antibodies and next-generation diabetes treatments, drives clinical trial activity. In addition, stringent regulatory standards and GMP-compliant facilities attract global sponsors, while strategic collaborations between academia and industry further enhance its competitiveness in the clinical trials landscape.
The diabetes clinical trials market in the UK is expected to grow significantly over the forecast period. The country's growth is fueled by a strong biotechnology ecosystem, world-class academic institutions, and a supportive regulatory environment. Government funding initiatives and policies promoting clinical research contribute to sustained market growth. The increasing focus on innovative therapies, including cell and gene-based treatments for diabetes management, is boosting trial activity. In addition, the emphasis on digital health integration and patient-centric trial models enhances efficiency and recruitment, making it a preferred destination for conducting complex and large-scale clinical studies.
Asia Pacific Diabetes Clinical Trials Market Trends
The diabetes clinical trials market in the Asia Pacific is expected to grow at a significant CAGR over the forecast period. The market is driven by cost advantages, expanding healthcare infrastructure, and rising biopharmaceutical investments. Countries such as China, India, and Japan are strengthening regulatory frameworks and aligning with international standards to attract global sponsors. The region offers access to large and diverse patient populations, enabling efficient trial recruitment. The increasing availability of skilled professionals, improved clinical capabilities, and supportive government initiatives further the region as a key outsourcing hub for diabetes clinical research. Such factors are expected to drive the market.
The China diabetes clinical trials market is witnessing new growth opportunities driven by strong government support, expanding biotechnology capabilities, and rising R&D investments. The country's cost-effective clinical trial environment and large patient pool make it attractive for global pharmaceutical companies. Regulatory reforms aimed at accelerating drug approvals and improving transparency have further enhanced its competitiveness. In addition, the development of advanced research facilities and increased participation in global clinical programs are driving outsourcing, further accelerating it and supporting the market.
The diabetes clinical trials market in Japan is characterized by high-quality standards, advanced technological capabilities, and a well-established pharmaceutical industry. The country emphasizes precision medicine and innovative therapeutic approaches, supported by a robust regulatory framework. Strong collaboration between academia, research institutions, and industry players drives continuous innovation in diabetes treatment. Japan's expertise in clinical research and commitment to compliance ensure reliable trial outcomes, making it a preferred destination for specialized and high-value clinical trials in the region.
The India diabetes clinical trials market is experiencing a cost-efficient environment, a large patient population, and an expanding pharmaceutical sector. The availability of skilled investigators and improved regulatory alignment with global standards have enhanced the country's attractiveness for clinical research. Increasing investments in clinical infrastructure and rising participation in multinational trials are further supporting market growth. In addition, the country's ability to provide scalable and cost-effective trial solutions positions it as a preferred destination for outsourcing diabetes clinical studies.
Latin America Diabetes Clinical Trials Market Trends
The diabetes clinical trials market in Latin America is expected to grow significantly over the forecast period. The market is driven by improving healthcare infrastructure, favorable regulatory developments, and increasing investment in biopharmaceutical research. Countries such as Brazil and Argentina are strengthening domestic capabilities to reduce dependency on imports and enhance regional clinical research capacity. The region offers access to diverse patient populations and cost advantages, attracting global sponsors. Besides, government initiatives promoting local innovation and partnerships are further contributing to the expansion of diabetes clinical trials across the region.
The Brazil diabetes clinical trials market is driven by increasing healthcare investments, supportive government policies, and growing focus on domestic biopharmaceutical production. The country's large patient pool and expanding clinical research infrastructure make it attractive for global clinical trials. Rising demand for biologics and biosimilars further supports trial activity. In addition, Brazil's efforts to enhance regulatory efficiency and promote innovation are strengthening its position as a key regional hub for diabetes clinical research. Such factors are expected to drive the market over the estimated time period.
Middle East & Africa Diabetes Clinical Trials Market Trends
The diabetes clinical trials market in the MEA region is expected to grow steadily over the forecast period. The market is driven by increasing government investments, improving healthcare systems, and rising demand for advanced therapies. The region's strategic location and expanding clinical infrastructure are attracting global pharmaceutical companies. The growing focus on regulatory improvements and the adoption of international standards is enhancing trial quality and compliance. In addition, the increasing prevalence of diabetes and demand for innovative treatments are driving clinical research activities across the region.
The UAE diabetes clinical trials market is driven by strong government initiatives, investments in healthcare infrastructure, and efforts to establish a robust life sciences sector. The country's favorable business environment and evolving regulatory framework attract global biopharma companies and CROs. Its strategic location as a distribution and logistics hub further enhances its appeal. Besides, increasing focus on innovation, advanced clinical facilities, and partnerships with international organizations is expected to drive sustained growth in diabetes clinical trials in the UAE.
Key Diabetes Clinical Trials Company Insights
The key players operating across the market are adopting strategic initiatives such as service launches, mergers & acquisitions, partnerships & agreements, and expansions to gain a competitive edge in the market. For instance, in October 2025, Eli Lilly and Company initiated two new clinical studies to evaluate the potential of the drug Baricitinib in delaying the onset and slowing the progression of Type 1 Diabetes. These trial launches build on prior evidence generated from the BANDIT study conducted in Australia, which assessed the drug’s effectiveness in children and young adults recently diagnosed with the condition. The earlier research was supported by Breakthrough T1D through its Type 1 Diabetes Clinical Research Network. The new trials are expected to further validate baricitinib’s role as a disease-modifying therapy, marking a significant step toward early intervention strategies in Type 1 diabetes management.
Key Diabetes Clinical Trials Companies:
The following key companies have been profiled for this study on the diabetes clinical trials market.
- Syneos Health
- Medpace
- IQVIA
- ICON plc
- Fortrea
- Parexel
- ProSciento
- Profil Institut für Stoffwechselforschung
- Novo Nordisk
- Eli Lilly and Company
- Sanofi
- AstraZeneca
Diabetes Clinical Trials Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 5.4 billion
Revenue forecast in 2033
USD 9.3 billion
Growth rate
CAGR of 7.9% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends, clinical trials outlook, volume analysis
Segments covered
Phase, study design, indication, indication by study design, service, sponsor, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country Scope
U.S.; Canada; Mexico; UK; Germany; Italy; France; Spain; Denmark; Sweden; Norway; Japan; China; India; South Korea; Australia; Thailand; Brazil; Argentina; Colombia; South Africa; Saudi Arabia; UAE; Kuwait; Qatar; Oman
Key companies profiled
Syneos Health; Medpace; IQVIA; ICON plc; Fortrea; Parexel; ProSciento; Profil Institut für Stoffwechselforschung; Novo Nordisk; Eli Lilly and Company; Sanofi; AstraZeneca
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Diabetes Clinical Trials Market Report Segmentation
This report forecasts revenue growth at regional and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global diabetes clinical trials market report based on phase, study design, indication, indication by study design, service, sponsor, and region:
-
Phase Outlook (Revenue, USD Million, 2021 - 2033)
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Study Design Outlook (Revenue, USD Million, 2021 - 2033)
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Type 1 Diabetes Mellitus (T1DM)
-
Type 2 Diabetes Mellitus (T2DM)
-
Gestational Diabetes
-
Others
-
-
Indication by Study Design Outlook (Revenue, USD Million, 2021 - 2033)
-
Type 1 Diabetes Mellitus (T1DM)
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Type 2 Diabetes Mellitus (T2DM)
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Gestational Diabetes
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Others
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Protocol Designing
-
Site Identification
-
Patient Recruitment
-
Laboratory Services
-
Analytical Testing Services
-
Clinical Trial Data Management Services
-
Others
-
-
Sponsor Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical & Biopharmaceutical Companies
-
Medical Device Companies
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
Japan
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
Colombia
-
-
Middle East and Africa (MEA)
-
South Africa
-
UAE
-
Saudi Arabia
-
Kuwait
-
Qatar
-
Oman
-
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.









